Moleculin Announces Presentation of Positive Data Demonstrating High Anti-Cancer Activity of Annamycin and Non-Cardiotoxic Properties
Annamycin demonstrated to be a more potent inhibitor of topoisomerase II-alpha and II-beta while remaining inactive against established cardiomyocyte cultures
Results clearly aligned with lack of drug-related cardiotoxic events in patients treated with Annamycin in ongoing clinical trials; 100% of Annamycin subjects in multiple studies (N=82) continue to show no signs of cardiotoxicity during study
A prominent treatment for many adult cancers and approximately 50% of all pediatric cancer patients are treated with a cardiotoxic anthracycline
Annamycin composition of matter patent issued April 9th; Patent enables expansion into greater patient populations where cardiotoxicity remains an unmet need
HOUSTON, April 10, 2024 /PRNewswire/ -- Moleculin Biotech, Inc., (NASDAQ:MBRX) (Moleculin or the Company), a clinical stage pharmaceutical company with a broad portfolio of drug candidates targeting hard-to-treat tumors and viruses, today announced positive preclinical data regarding the Company's next-generation anthracycline, Annamycin, was presented at the American Association for Cancer Research (AACR) Annual Meeting, taking place April 5-10, 2024 in San Diego, CA.
The poster titled, Non-cardiotoxic Properties of Annamycin, a Clinically Evaluated Anthracycline and Potent Topoisomerase 2β Poison, was presented in the "Late-Breaking Research: Experimental and Molecular Therapeutics 2" session held on Monday, April 8th. The presented poster outlined results from the assessment and comparison of the potency of doxorubicin (a commonly prescribed anthracycline) and Annamycin, Moleculin's next-generation anthracycline, against topoisomerase II-alpha and II-beta and determine their impact on physiology of human cardiomyocytes demonstrating no pathologic changes in mice hearts following chronic in vivo exposure.
"Cardiotoxicity continues to be a significant side effect limiting the clinical use of anthracyclines, despite anthracyclines representing some of the most important treatments available for AML and Advanced STS. These data, documenting lack of cardiotoxicity of Annamycin and aligned with the data demonstrated to date in our ongoing clinical trials, bolster our confidence in Annamycin potential to offer patients a safe and effective treatment option. Interestingly, the presented data demonstrates that Annamycin appears to be a significantly more potent inhibitor of topoisomerase II-beta than doxorubicin providing validation for additional studies to reevaluate the role of topoisomerase II-beta in anthracycline induced cardiotoxicity," commented Walter Klemp, Chairman and Chief Executive Officer of Moleculin.
"Additionally, with our recently issued patent, which covers composition of matter protection across all indications, we have the potential to expand the development of Annamycin into greater patient populations in indications where cardiotoxicity remains a significant unmet need. We remain highly encouraged by Annamycin and committed to advancing its development," concluded Mr. Klemp.
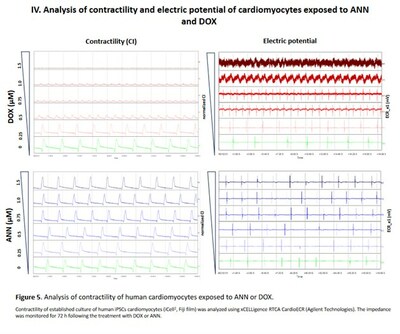
As part of the presented data, the potency of Annamycin and doxorubicin to inhibit topoisomerase II was tested in DNA relaxation assays using recombinant topoisomerase II-alpha and II-beta with kinetoplast as the DNA substrate. Annamycin and doxorubicin cytotoxicity was assessed in a panel of cancer cell lines cardiomyocytes (murine and human). In addition, the effects on cardiomyocyte physiology (beating rate, contraction, electric potential,) were assessed in human cardiomyocytes using the xCELLigence RTCA CardioECR. The pathophysiology of the heart after chronic exposure to the drugs was also evaluated in mice models.
Key Data Highlights:
- Annamycin demonstrated to be a more potent topoisomerase II-alpha and II-beta poison than doxorubicin.
- In contrast to doxorubicin:
- Annamycin does not affect viability of established culture of human iPSCc cardiomyocytes (tested up to 1.5 μM)
- Annamycin does not affect contractility or the electric potential of the cardiomyocytes
- Rat cardiomyocytes (H9c2) appear to be resistant to ANN while sensitive to DOX
- Annamycin is well tolerated by the animals even at schedules exceeding the therapeutic dosage while ex vivo pathology examination of the mice confirmed no toxicity to the heart/myocardium.
- These data are clearly aligned with the lack of drug-related cardiotoxic events in Annamycin-treated patients in ongoing clinical trials
- The role of topoisomerase II-beta in cardiotoxicity of anthracyclines should be further investigated
- A video on the comparison of cardiotoxic effects of Annamycin and doxorubicin on human cardiomyocytes can be accessed here
The Company also announced that the United States Patent and Trademark Office (USPTO) formally issued U.S. Patent number 11,951,118 titled, "Preparation of Preliposomal Annamycin Lyophilizate" (the '118 patent') to Moleculin and The University of Texas System Board of Regents.
Annamycin is currently being evaluated in ongoing clinical trials for the treatment of relapsed or refractory acute myeloid leukemia (AML) and soft tissue sarcoma (STS) lung metastases. For more information about the ongoing trials, please visit clinicaltrialsregister.eu: EudraCT 2020-005493-10 or clinicaltrials.gov: NCT05319587; and clinicaltrials.gov: NCT04887298, respectively.
About Moleculin Biotech, Inc.
Moleculin Biotech, Inc. is a clinical stage pharmaceutical company with a growing pipeline, including Phase 2 clinical programs, for hard-to-treat tumors and viruses. The Company's lead program, Annamycin is a next-generation anthracycline designed to avoid multidrug resistance mechanisms with little to no cardiotoxicity. Annamycin is currently in development for the treatment of relapsed or refractory acute myeloid leukemia (AML) and soft tissue sarcoma (STS) lung metastases.
Additionally, the Company is developing WP1066, an Immune/Transcription Modulator capable of inhibiting p-STAT3 and other oncogenic transcription factors while also stimulating a natural immune response, targeting brain tumors, pancreatic and other cancers, and WP1220, an analog to WP1066, for the topical treatment of cutaneous T-cell lymphoma. Moleculin is also engaged in the development of a portfolio of antimetabolites, including WP1122 for the potential treatment of viruses, as well as cancer indications including brain tumors, pancreatic and other cancers.
For more information about the Company, please visit www.moleculin.com and connect on Twitter, LinkedIn and Facebook.
Forward-Looking Statements
Some of the statements in this release are forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, Section 21E of the Securities Exchange Act of 1934 and the Private Securities Litigation Reform Act of 1995, which involve risks and uncertainties. Forward-looking statements in this press release include, without limitation, the expected issuance of the patent discussed above, the pace of enrollment in Moleculin's clinical trials, the timing of Moleculin's ability to report topline data from its studies, the timing of the commencement of investigator-sponsored and/or externally funded clinical trials which are outside the control of Moleculin, and whether the results of Moleculin's preclinical animal models can be replicated in human trials. Although Moleculin believes that the expectations reflected in such forward-looking statements are reasonable as of the date made, expectations may prove to have been materially different from the results expressed or implied by such forward-looking statements. Moleculin has attempted to identify forward-looking statements by terminology including 'believes,' 'estimates,' 'anticipates,' 'expects,' 'plans,' 'projects,' 'intends,' 'potential,' 'may,' 'could,' 'might,' 'will,' 'should,' 'approximately' or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. These statements are only predictions and involve known and unknown risks, uncertainties, and other factors, including those discussed under Item 1A. "Risk Factors" in our most recently filed Form 10-K filed with the Securities and Exchange Commission (SEC) and updated from time to time in our Form 10-Q filings and in our other public filings with the SEC. Any forward-looking statements contained in this release speak only as of its date. We undertake no obligation to update any forward-looking statements contained in this release to reflect events or circumstances occurring after its date or to reflect the occurrence of unanticipated events.
Investor Contact:
JTC Team, LLC
Jenene Thomas
(833) 475-8247
[email protected]
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/moleculin-announces-presentation-of-positive-data-demonstrating-high-anti-cancer-activity-of-annamycin-and-non-cardiotoxic-properties-302112418.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/moleculin-announces-presentation-of-positive-data-demonstrating-high-anti-cancer-activity-of-annamycin-and-non-cardiotoxic-properties-302112418.html
SOURCE Moleculin Biotech, Inc.