Corvus Pharmaceuticals Announces Full Data from Cohort 3 of Placebo-Controlled Phase 1 Clinical Trial of Soquelitinib for Atopic Dermatitis
Cohort 3 demonstrates earlier and deeper responses compared to cohorts 1-2
All three cohorts show separation from placebo with statistically significant difference from placebo at day 28
Cohort 3 demonstrates clinically meaningful reduction in itch as early as day 8
Enrollment initiated in extension cohort study exploring the same cohort 3 dose (200 mg BID) for a longer 8-week treatment period
SOUTH SAN FRANCISCO, Calif., June 04, 2025 (GLOBE NEWSWIRE) -- Corvus Pharmaceuticals, Inc. (NASDAQ:CRVS), a clinical-stage biopharmaceutical company, today announced new interim data from the randomized, double-blind, placebo-controlled Phase 1 clinical trial evaluating soquelitinib in patients with moderate to severe atopic dermatitis. The data includes 28-day follow up results for all patients in cohort 3 and continues to show earlier and deeper responses in cohort 3 (200 mg twice per day, total daily dose 400 mg) compared to cohorts 1 and 2 (100 mg twice per day and 200 mg once per day, total daily dose 200 mg). Overall, data from cohorts 1-3 of the trial have demonstrated a favorable safety and efficacy profile, including a statistically significant improvement in Eczema Area and Severity Index (EASI) score for the soquelitinib treated patients compared to placebo at day 28 (p=0.036).
"The complete 28-day data from cohort 3 of our Phase 1 trial of soquelitinib in patients with atopic dermatitis is in-line with the data update we provided at the Society for Investigative Dermatology meeting last month," said Richard A. Miller, M.D., co-founder, president and chief executive officer of Corvus. "We are encouraged that results from cohort 3 continue to show earlier and deeper responses, along with a reduction in itch, which is an important factor for patients. We look forward to exploring the potential for further improvement in patient results with longer treatment duration that is being studied in our recently initiated extension cohort. Overall, the data to date is supportive of our view that ITK inhibition with soquelitinib has the potential to be a safe, effective and convenient new option for patients with atopic dermatitis and other immune diseases."
Dr. Miller will highlight the new interim data in a presentation at the Jefferies Global Healthcare Conference, which is scheduled for 9:20 am ET / 6:20 am PT on Thursday, June 5, 2025. The live webcast, which will include presentation slides, may be accessed via the investor relations section of the Corvus website. A replay of the webcast will be available on Corvus' website for 90 days.
Soquelitinib Interim Data from the Atopic Dermatitis Phase 1 Clinical Trial
As of May 28, 2025, enrollment in cohorts 1, 2 and 3 has been completed for a total of 48 patients and all patients (36 receiving soquelitinib and 12 placebos) had completed the 28-day treatment course. Patients in cohort 3 had more advanced disease with a higher mean baseline EASI score compared to patients in cohorts 1 and 2. At 28 days, the mean reduction in EASI for cohort 3 (n=12) was 64.8%, compared to 54.6% for cohort 1 and 2 combined (n=24) and 34.4% for placebo (n=12).
The graphs below (Figures 1 and 2) show the kinetics of response for each of the cohorts and for the combined cohorts 1, 2 and 3. The placebo patients (n=4 per cohort, total n=12) are combined in both graphs. Separation of the curves for patients receiving active drug began at day 15 and increased by day 28 for cohorts 1 and 2. Cohort 3 patients experienced earlier and deeper separation from placebo starting by day 8. EASI scores continue to improve further in treated patients from all cohorts out to day 58.
Figure 1: Percent Reduction in Mean EASI Score for Cohorts 1, 2 and 3. Mean percent change in EASI score over time is shown. Treatment beginning is designated "Baseline" and days post-baseline are shown. Screening to baseline data is shown and demonstrates relative disease stability. The study blinding remains in effect for the entire 58-day period. Numbers at the top of the graphs indicate numbers of patients evaluated at the various time points.
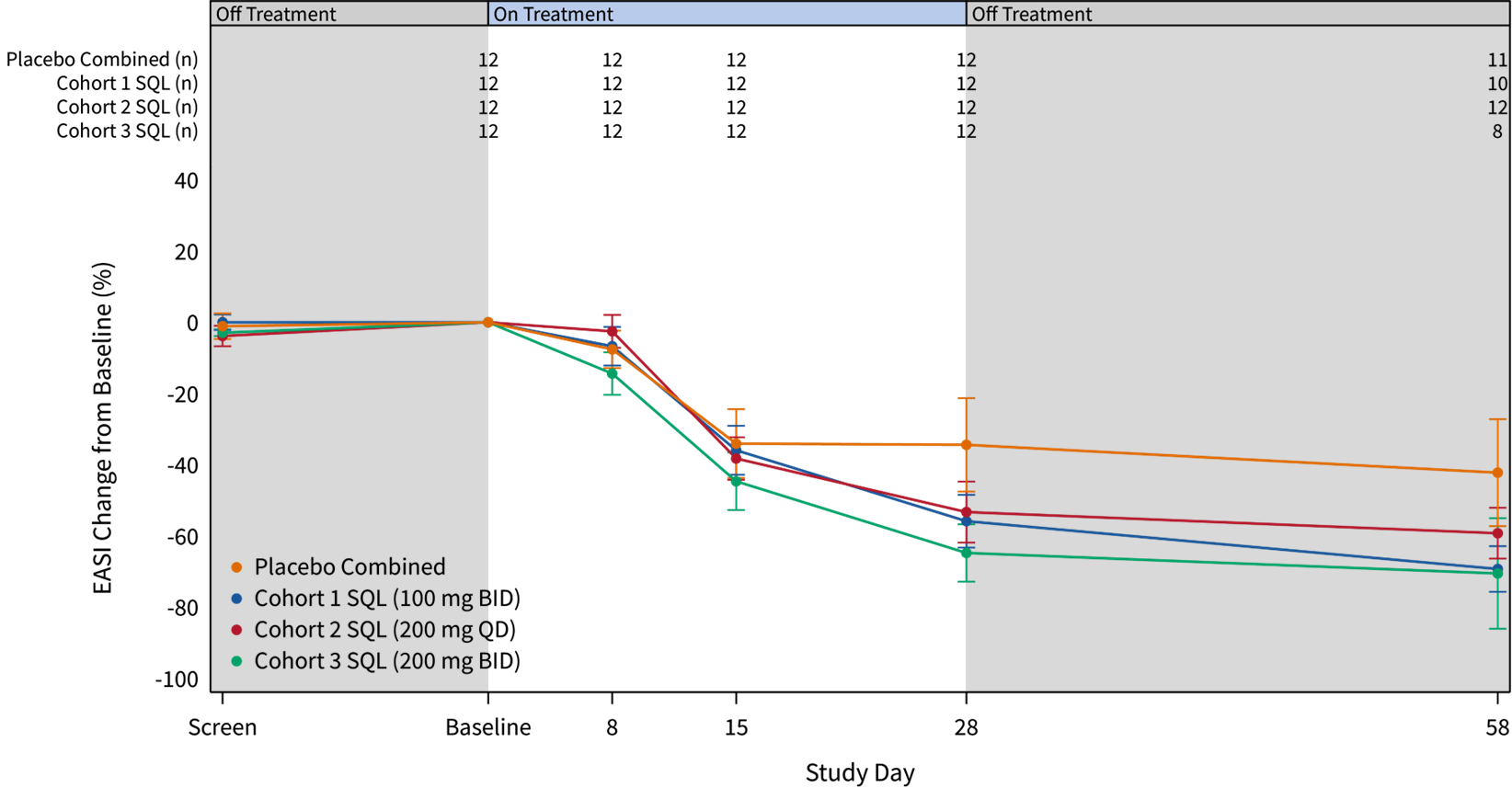
Figure 2: Percent Reduction in Mean EASI Score for Combined Cohorts 1, 2 and 3. The data is displayed below with cohorts combined.
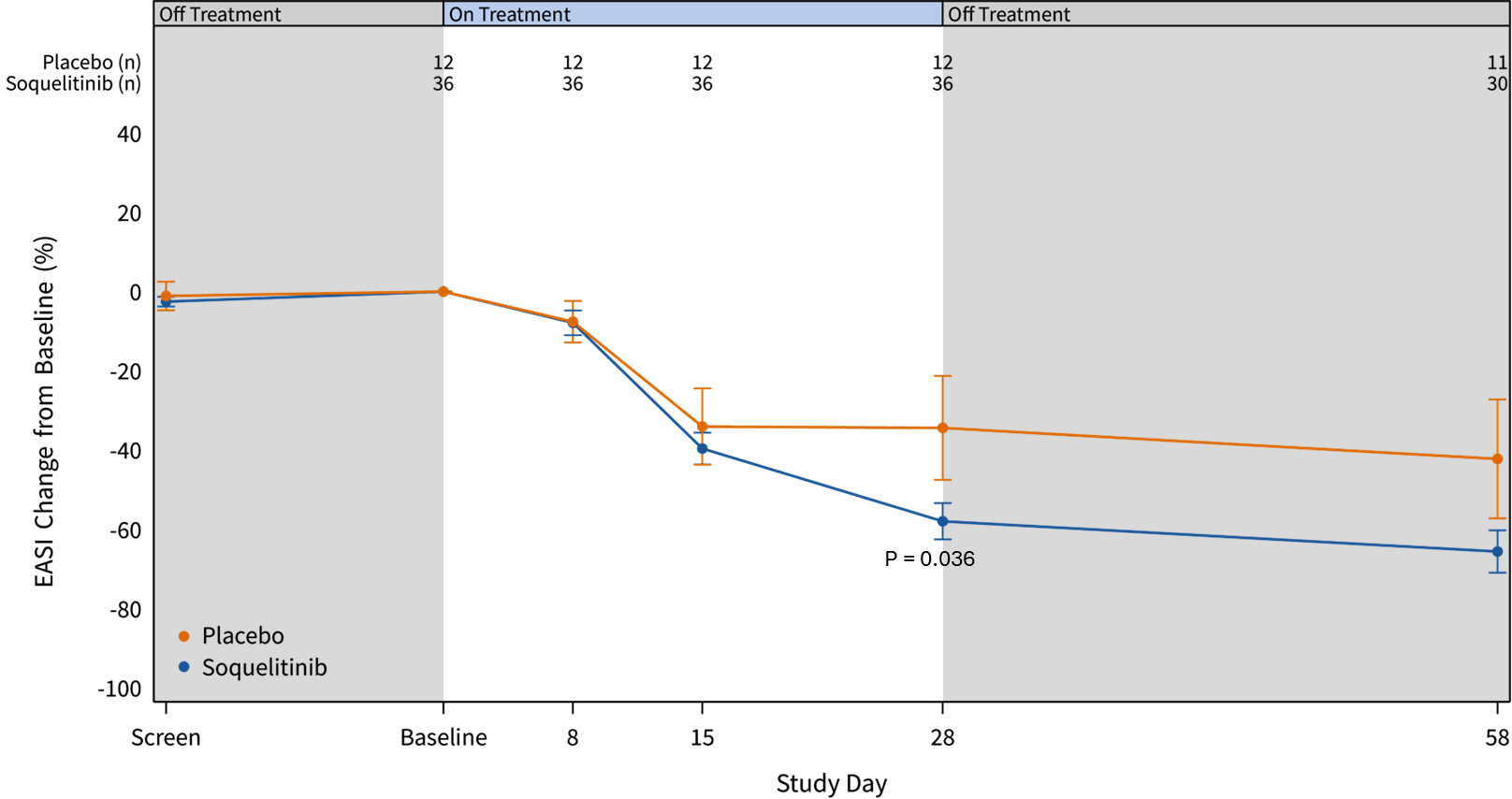
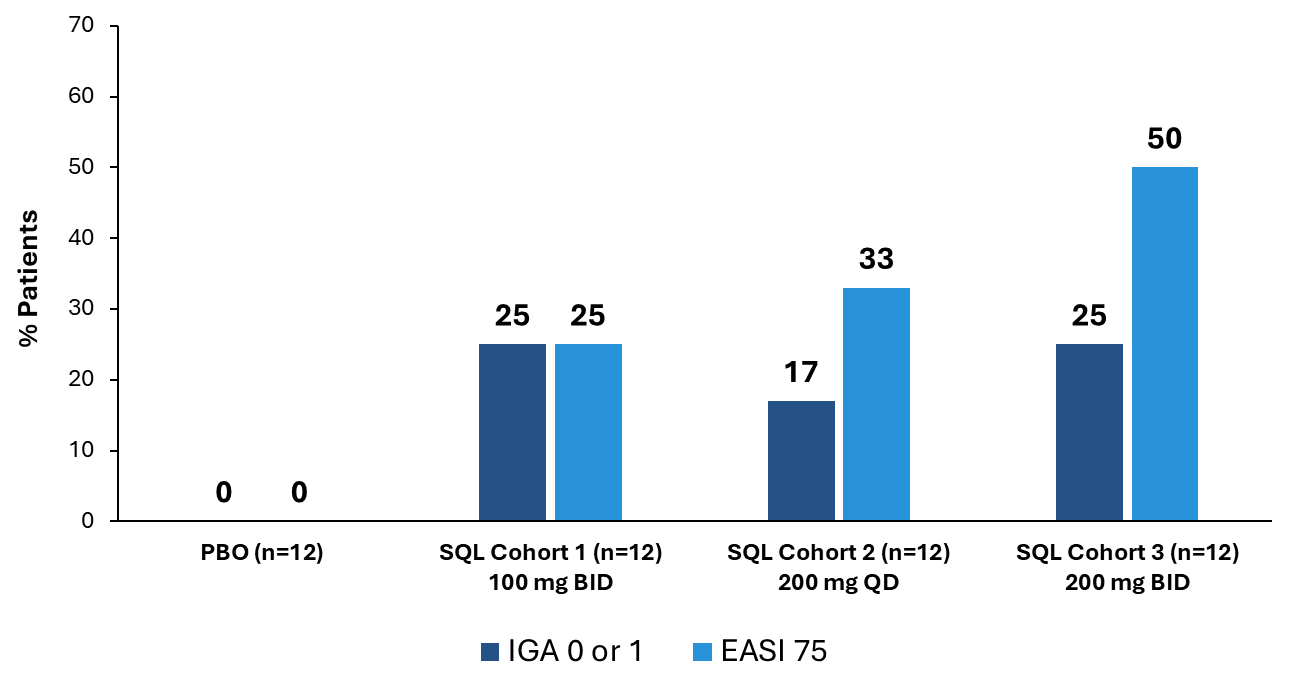
Figure 3 below shows the percent of patients that achieved IGA (Investigator Global Assessment) 0 or 1 or EASI 75 at day 28 of treatment. The placebo patients from cohort 1 (n=4), cohort 2 (n=4) and cohort 3 (n=4) are combined, with no placebo patients achieving IGA 0 or 1 or EASI 75. IGA 0 or 1 and EASI 75 have been determined by the U.S. Food and Drug Administration (FDA) to be clinically meaningful and approvable endpoints and have been the endpoints used in clinical trials for other FDA approved treatments for atopic dermatitis. Four additional patients in cohort 3 are now included in the results as compared to the data reported at the Society for Investigative Dermatology (SID) annual meeting in May 2025 (SID data was as of May 6; these four patients had not yet completed the 28-day treatment course). One of the four patients achieved EASI 75 (this patient experienced an 89% reduction in EASI score) and IGA 1 at day 28 of treatment.
Figure 3: Percent Patients Achieving Endpoints IGA 0 or 1, EASI 75 at Day 28 of Treatment
Patient Reported Reductions in Itch
Patients in the trial recorded the intensity of their pruritus, or itch, using the Peak Pruritus Numerical Rating Scale (PP-NRS), which rates the severity of itch on a scale from 0 (no itch) to 10 (the worst itch imaginable). A reduction of ≥4 points from baseline on the PP-NRS is considered to be a clinically meaningful result. In cohort 3, of the patients for whom adequate PP-NRS data was available, 4 of 8 (50%) had a ≥4 point reduction in PP-NRS score from baseline at day 28, with a reduction in itch seen as early as day 8. Of the remaining patients, two had baseline PP-NRS of less than 4 and two had incomplete PP-NRS data. 1 of 10 evaluable placebo patients (10%) experienced a ≥4 point reduction in PP-NRS score at Day 28.
Safety Data
As of May 28, 2025, no new safety signals have been observed. Soquelitinib was well tolerated, with no dose limiting toxicities (DLTs) and no clinically significant laboratory abnormalities observed in any of the cohorts. No interruption of drug dosing was seen in any of the cohorts. Grade 1/2 adverse events (treatment related and unrelated) were seen in 38.9% of patients receiving soquelitinib and 25% receiving placebo. Only one treatment related adverse event of grade 1 nausea was reported with soquelitinib treatment.
Serum Cytokine and Other Biomarker Studies
As reported previously, relationships between reductions in certain cytokines with improvement in EASI scores were observed. Reductions in serum cytokine levels were seen for IL-5, IL-9, IL-17, IL-31, IL-33, TSLP and TARC. Differences between responding and non-responding patients were found, while no such relationships were seen in the placebo group, and patients in cohort 3 had greater reductions in cytokines compared to cohorts 1 and 2. Increasing trends were seen in numbers of circulating T regulatory cells, consistent with the presumed mechanism of action of soquelitinib.
Soquelitinib Atopic Dermatitis Phase 1 Clinical Trial Extension Cohort
Corvus also announced that the first patient(s) has/have been enrolled in the recently announced extension cohort of the Phase 1 trial. This cohort is planned to enroll 24 patients randomized 1:1 between active and placebo, with patients in the treatment group receiving the same dose as cohort 3 – 200 mg orally twice per day. The treatment period for this group is 8 weeks, compared to 4 weeks in cohorts 1-3, with the same 30-day follow-up period with no treatment.
About Corvus Pharmaceuticals
Corvus Pharmaceuticals is a clinical-stage biopharmaceutical company pioneering the development of ITK inhibition as a new approach to immunotherapy for a broad range of cancer and immune diseases. The Company's lead product candidate is soquelitinib, an investigational, oral, small molecule drug that selectively inhibits ITK. Its other clinical-stage candidates are being developed for a variety of cancer indications. For more information, visit www.corvuspharma.com or follow the Company on LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements related to the potential of the Company's product candidates including soquelitinib and the potential for further improvement in patient results in the extension cohort of the Phase 1 trial of soquelitinib in patients with atopic dermatitis, the design and planned enrollment of the extension cohort, data in support of ITK inhibition with soquelitinib and its potential for patients, and continued advancement of the Company's clinical pipeline. All statements other than statements of historical fact contained in this press release are forward-looking statements. These statements often include words such as "believe," "expect," "anticipate," "intend," "plan," "estimate," "seek," "will," "may" or similar expressions. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company's control. The Company's actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to, risks detailed in the Company's Quarterly Report on Form 10-Q for the quarter ended March 31, 2025, filed with the Securities and Exchange Commission on May 8, 2025, as well as other documents that may be filed by the Company from time to time with the Securities and Exchange Commission. In particular, the following factors, among others, could cause results to differ materially from those expressed or implied by such forward-looking statements: the Company's ability to demonstrate sufficient evidence of efficacy and safety in its clinical trials of its product candidates; the accuracy of the Company's estimates relating to its ability to initiate and/or complete preclinical studies and clinical trials and release data from such studies and clinical trials; the results of preclinical studies and interim data from clinical trials not being predictive of future results; the Company's ability to enroll sufficient numbers of patients in its clinical trials; the unpredictability of the regulatory process; regulatory developments in the United States and foreign countries; the costs of clinical trials may exceed expectations; and the Company's ability to raise additional capital. Although the Company believes that the expectations reflected in the forward-looking statements are reasonable, it cannot guarantee that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and the timing of events and circumstances and actual results could differ materially from those projected in the forward-looking statements. Accordingly, you should not place undue reliance on these forward-looking statements. All such statements speak only as of the date made, and the Company undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise.
INVESTOR CONTACT:
Leiv Lea
Chief Financial Officer
Corvus Pharmaceuticals, Inc.
+1-650-900-4522
[email protected]
MEDIA CONTACT:
Sheryl Seapy
Real Chemistry
+1-949-903-4750
[email protected]
Figures accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/ad720d08-8c1d-4842-9c34-6772d9f1de2b
https://www.globenewswire.com/NewsRoom/AttachmentNg/db3a1681-831d-498a-85de-76b13d664563
https://www.globenewswire.com/NewsRoom/AttachmentNg/b9a3b3c5-b805-441c-bded-ac9cd240a54f

