Edgewise Therapeutics Announces Completion of the EDG-7500 CIRRUS-HCM Phase 2 Parts B and C and Favorable Interim Safety Results from the Ongoing Part D Study in Hypertrophic Cardiomyopathy
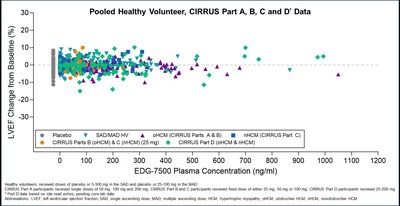
– In the CIRRUS-HCM trial, including interim safety results from Part D, EDG-7500 was generally well tolerated; no clinically meaningful reductions in LVEF or LVEF <50% –
– On track to deliver full 12-week Part D readout in 2Q 2026 and Phase 3 start in 4Q 2026 –
BOULDER, Colo., Dec. 24, 2025 /PRNewswire/ -- Edgewise Therapeutics, Inc., (NASDAQ:EWTX), today announced positive updates from the ongoing CIRRUS-HCM, Phase 2 clinical trial of EDG-7500, a novel oral, selective, cardiac sarcomere modulator, specifically designed to slow early contraction velocity and address impaired cardiac relaxation associated with hypertrophic cardiomyopathy (HCM) without impacting systolic function, two central clinical goals in the current management of HCM.
CIRRUS-HCM is a multi-part, open label trial of EDG-7500 in participants with obstructive and nonobstructive HCM. Earlier this year, the Company reported positive top-line results from the Part B (oHCM, n=17) and Part C (nHCM, n=12) 50 mg and 100 mg fixed dose cohorts, in which EDG-7500 administration led to improvements in key HCM disease markers, including N-terminal pro-B-type natriuretic peptide (NT-proBNP), Kansas City Cardiomyopathy Questionnaire (KCCQ), New York Heart Association (NYHA) class and left ventricular outflow tract gradient (LVOT-G) (in oHCM participants). In both Parts B and C, EDG-7500 administration led to KCCQ score improvements that appeared favorable relative to those reported in other cardiovascular trials, including those with cardiac myosin inhibitors (CMIs). EDG-7500 administration was also associated with measurable improvements in key diastolic parameters, including robust reductions in NT-proBNP, reduced left ventricular filling pressures, and improved relaxation metrics.
In addition to the 50 mg and 100 mg fixed doses previously reported, the Company evaluated a lower 25 mg fixed dose in 4 oHCM and 10 nHCM participants in CIRRUS-HCM Parts B and C, respectively. In these cohorts, evidence of clinical activity was observed across key HCM disease markers, including NT-proBNP, KCCQ, NYHA and LVOT-G (in oHCM participants), while maintaining a favorable safety profile, with no clinically meaningful changes in left ventricular ejection fraction (LVEF) or reductions in LVEF to below <50% and no events of atrial fibrillation (AF). The CIRRUS-HCM Part B and C cohorts have completed dosing, with a total of 43 participants enrolled across all dose groups.
CIRRUS-HCM Part D is designed to explore exposure-response correlations, assess biomarker-guided dose optimization to inform the design of Phase 3 trials, and support regulatory discussions. EDG-7500 is being evaluated over a 12-week active treatment period; participants will be provided the opportunity to continue into a long-term extension study. Dosing is guided by objective measures—LVOT-G in oHCM and NT-proBNP in nHCM—with participants dosed from 25 mg to 150 mg and eligible for further optimization up to 200 mg in the extension phase. With intra-patient dose optimization results from Part D, the Company aims to build on the robust clinical improvements observed in Parts B and C to further support EDG-7500's best-in-disease potential.
Screening at sites involved in Part D is complete, with more than 40 participants enrolled. As of the December 23, 2025 data cutoff, approximately 70% of participants had reached a dose of 100 mg or higher. Overall, patient demographics in Part D are generally consistent with those reported in Phase 3 studies of CMIs.
In this interim safety update, for the 20 participants who completed 12 weeks of dosing in Part D as of the data cutoff date (8 with oHCM and 12 with nHCM), EDG-7500 generally had a favorable safety profile and was well tolerated. Consistent with previous observations, no clinically significant changes in LVEF or reductions in LVEF to below 50% were observed, with EDG-7500 continuing to demonstrate a differentiated LVEF profile relative to CMIs. The safety profile observed with EDG-7500 to date, across both healthy volunteers and the CIRRUS-HCM trial, supports the potential use of EDG-7500 in diverse HCM patient populations. Unlike CMIs, which have a mechanism of action that has been associated with a risk of systolic dysfunction and an increased risk of heart failure, EDG-7500 administration continues to present no meaningful effect on LVEF. Preservation of systolic function has the potential to avoid the patient safety monitoring burden that could limit CMI adoption outside of HCM centers of excellence, which could expand prescriber reach and patient access, if approved.
Furthermore, in Part D, continuous active cardiac monitoring was conducted for four weeks during screening and for two weeks following each dose escalation. More than 300 ambulatory cardiac monitoring devices have been deployed to participants, capturing over 2,600 patient-days of active cardiac monitoring, during which no clinically detectable AF or atrial flutter has been observed. A single adverse event of new onset AF was reported; this event occurred in an oHCM participant who was not on active cardiac monitoring at the time and who had a history of arrhythmias, including supraventricular tachycardia. The investigator deemed the event as not related to study drug.
"I'm excited about the advances we've made in Part D of the CIRRUS-HCM trial, where we've exceeded our year-end enrollment goal, highlighting continued enthusiasm for the program from patients and physicians" said Kevin Koch, Ph.D., President and Chief Executive Officer. "I'm especially pleased with EDG-7500's safety profile to date, and the lack of clinically relevant drops in ejection fraction, or any ejection fraction drops below 50%. A major focus of 2026 will be refining our development strategy to deliver the best-in-disease therapeutic profile that EDG-7500 may offer to obstructive and nonobstructive HCM patients."
The Company remains on track to deliver comprehensive efficacy and safety data from Part D of the CIRRUS-HCM trial in the second quarter of 2026 and is advancing Phase 3 trial design in preparation for trial initiation by the end of 2026.
About Hypertrophic Cardiomyopathy
HCM is the most common form of genetic heart disease, affecting approximately one in 500 people, and is associated with reduced quality of life and an elevated risk of heart failure, abnormal heart rhythms, and sudden cardiac death. Individuals with HCM can become extremely limited in their functional capacity and ability to perform the activities of daily living. Commonly experienced symptoms include breathlessness, irregular heartbeats, chest pain, tiredness, dizziness, or even fainting. These symptoms are caused by excessive contraction and thickening (hypertrophy) of the left ventricular wall of the heart. Over time, the thickened muscle becomes stiff, making it difficult for the heart to relax and fill with blood (diastolic dysfunction). There are two major forms of HCM obstructive and nonobstructive. Despite advancements in treatment options for some patients with HCM, there remains a significant unmet need for additional therapeutic approaches for patients.
About Edgewise Therapeutics
Edgewise Therapeutics is a leading muscle disease biopharmaceutical company developing novel therapeutics for muscular dystrophies and serious cardiac conditions. The Company's deep expertise in muscle physiology is driving a new generation of novel therapeutics. EDG-7500 is a novel cardiac sarcomere modulator for the treatment of hypertrophic cardiomyopathy , currently in Phase 2 clinical development. Sevasemten is an orally administered first-in-class fast skeletal myosin inhibitor in late-stage clinical trials in Becker and Duchenne muscular dystrophies. EDG-15400 is a novel cardiac sarcomere modulator for the treatment of heart failure, currently in Phase 1 clinical development. The entire team at Edgewise is dedicated to our mission: changing the lives of patients and families affected by serious muscle diseases. To learn more, go to: www.edgewisetx.com or follow us on LinkedIn and X.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements as that term is defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Statements in this press release that are not purely historical are forward-looking statements. Such forward-looking statements include, among other things, statements regarding the benefits and potential of, and expectations regarding, EDG-7500; statements regarding the market opportunity for EDG-7500; statements regarding Edgewise's expectations relating to its clinical trials, including timing of reporting comprehensive efficacy and safety data from Part D of the CIRRUS-HCM trial and timing of the initiation of the Phase 3 trials of EDG-7500 ); clinical outcomes from trials of EDG-7500, which may materially change as more patient data become available; statements regarding the best-in-disease therapeutic profile of EDG-7500; and statements by Edgewise's President and Chief Executive Officer. Words such as "believes," "anticipates," "plans," "expects," "intends," "will," "goal," "potential" and similar expressions are intended to identify forward-looking statements. The forward-looking statements contained herein are based upon Edgewise's current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results could differ materially from those projected in any forward-looking statements due to numerous risks and uncertainties, including but not limited to: the potential for the results of the ongoing or any future clinical trial of EDG-7500 to differ from the results of Parts B and C and the interim results of Part D of the ongoing CIRRUS-HCM trial; the risk of delays in completing the ongoing Part D of the CIRRUS-HCM trial or initiating the planned Phase 3 trials of EDG-7500; risks associated with unexpected events during the remainder of Part D of the ongoing CIRRUS-HCM trial, including serious adverse events, toxicities or other undesirable side effects; the risk of difficulties in enrolling or maintaining patients in the clinical trials of EDG-7500; risks associated with Edgewise's limited operating history, its products being early in development and not having products approved for commercial sale; risks associated with Edgewise not having generated any revenue to date; Edgewise's ability to achieve objectives relating to the discovery, development and commercialization of its product candidates, if approved; Edgewise's need for substantial additional capital to finance its operations; Edgewise's substantial dependence on the success of sevasemten and EDG-7500; Edgewise's ability to develop and commercialize sevasemten, EDG-7500 and EDG-15400 risks related to Edgewise's clinical trials of its product candidates not demonstrating safety and efficacy; risks related to Edgewise's product candidates causing serious adverse events, toxicities or other undesirable side effects; the outcome of preclinical testing and early clinical trials not being predictive of the success of later clinical trials and the risks related to the results of Edgewise's clinical trials not satisfying the requirements of regulatory authorities; delays or difficulties in the enrollment and/or maintenance of patients in clinical trials; risks related to failure to capitalize on other indications or product candidates; risks related to competition; risks relating to interim, topline and preliminary data from Edgewise's clinical trials changing as more patient data becomes available; risks related to failure to develop a proprietary drug discovery platform; risks related to exposure to additional risk if we develop sevasemten and potential other programs in connection with other therapies; risks related to production of drugs by Edgewise's third-party manufacturers; risks related to changes in methods of product candidate manufacturing or formulation; risks related to not achieving adequate market acceptance; risks related to the patient population for our product candidates having a small patient population; risks related to the regulatory approval processes of domestic and foreign authorities being lengthy, time consuming and inherently unpredictable; risks relating to disruptions at the FDA, the SEC and other government agencies; risks relating to Edgewise's ability to attract and retain highly skilled executive officers and employees; Edgewise's ability to obtain and maintain intellectual property protection for its product candidates; Edgewise's reliance on third parties; risks related to future acquisitions or strategic partnerships; risks related to general economic and market conditions; and other risks. Information regarding the foregoing and additional risks may be found in the section entitled "Risk Factors" in documents that Edgewise files from time to time with the U.S. Securities and Exchange Commission. These forward-looking statements are made as of the date of this press release, and Edgewise assumes no obligation to update the forward-looking statements, or to update the reasons why actual results could differ from those projected in the forward-looking statements, except as required by law.
This press release contains hyperlinks to information that is not deemed to be incorporated by reference into this press release.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/edgewise-therapeutics-announces-completion-of-the-edg-7500-cirrus-hcm-phase-2-parts-b-and-c-and-favorable-interim-safety-results-from-the-ongoing-part-d-study-in-hypertrophic-cardiomyopathy-302649113.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/edgewise-therapeutics-announces-completion-of-the-edg-7500-cirrus-hcm-phase-2-parts-b-and-c-and-favorable-interim-safety-results-from-the-ongoing-part-d-study-in-hypertrophic-cardiomyopathy-302649113.html
SOURCE Edgewise Therapeutics