EXCLUSIVE: GLP-1 Weight-Loss Drugs Could Become A Trillion-Dollar Market, Predicts Tema ETFs CEO
The GLP-1 drug market for obesity treatment is expected to skyrocket to $1 trillion or more, according to Maurits Pot, founder and CEO of Tema ETFs, who shared this optimistic forecast in an exclusive interview with Benzinga.
“Our view is that in the long term, this is not a two-horse race between Eli Lilly Company (NYSE:LLY) and Novo Nordisk A/S (NYSE:NVO),” Maurits stated, challenging the prevailing notion of a duopoly in the weight-loss market.
Pot predicts that rising demand for weight loss treatments and expanded applications for GLP-1 drugs will drive this trillion-dollar market opportunity.
In November 2023, Tema ETFs launched the Tema GLP-1, Obesity and Cardiometabolic ETF (NYSE:HRTS), the first ETF focused on weight loss. Since inception, the fund has risen 25%, slightly outpacing the performance of the S&P 500, as tracked by the SPDR S&P 500 ETF Trust (NYSE:SPY).
The ETF's major holdings include Eli Lilly and Company, Amgen Inc. (NASDAQ:AMGN), Novo Nordisk A/S, and Alnylam Pharmaceuticals, Inc. (NASDAQ:ALNY), each with about 5-6% weight. The HRTS portfolio also comprises over 40 other weight-loss related companies.
Chart: Tema GLP-1, Obesity And Cardiometabolic ETF Outperforms S&P 500 Since Inception
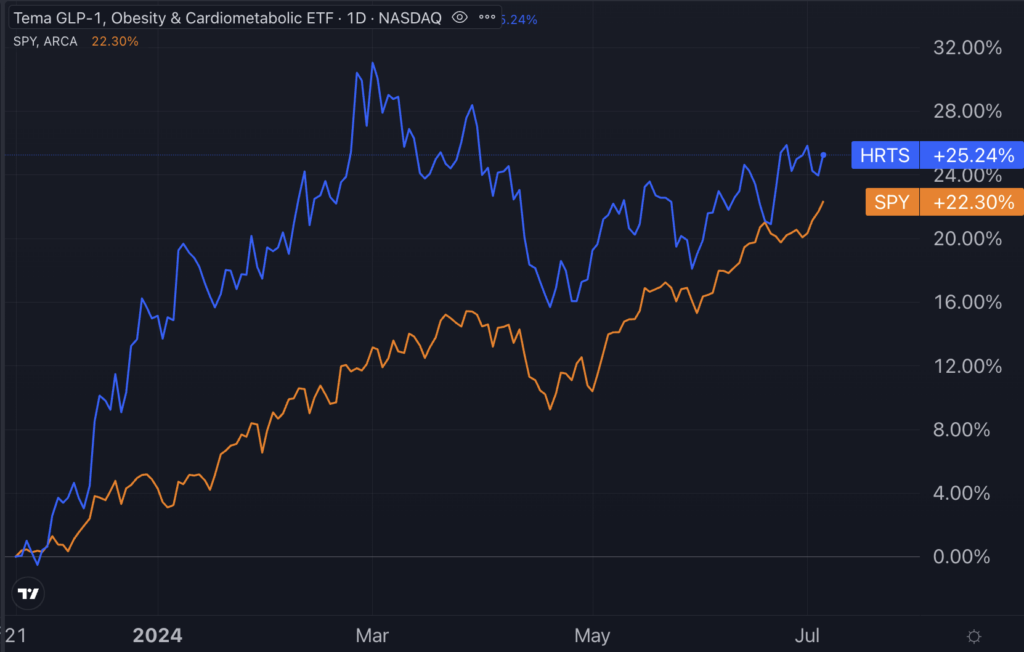
Chart: Benzinga Pro
Weight-Loss Treatments: Broadening Applications And Innovations
According to Pot, GLP-1 drugs may soon find applications beyond weight loss, first in diabetes, and heart, liver and kidney diseases, then potentially treating conditions such as cancer, Alzheimer's, Parkinson's and other neurogenic diseases.
The conventional injection method faces challenges from new approaches focusing on oral administration or reduced dosage frequencies. Innovations aim to make treatments less invasive, such as moving from weekly injections to potentially once every two months, or much easier to manufacture, such as producing oral tablets.
Obesity leads to various diseases like diabetes, heart disease and cancer. Addressing obesity could thus mitigate multiple health issues, amplifying the market potential for GLP-1 drugs.
This market is expected to extend beyond Eli Lilly and Novo Nordisk, addressing multiple diseases areas.
“We could see a world where half the world’s population takes GLP-1 or other weight loss drugs, but for different reasons including for primary prevention” Pot noted, citing uses for obesity, diabetes and potentially even Alzheimer’s or cancer.
Projections Behind The $1 Trillion Estimate
The global number of obese individuals is expected to double from 1 billion to 2 billion by 2035. Even without this growth, the existing 1 billion obese people represent a substantial market.
If long-term treatment reaches only 20% of today's obese population, similar to the current penetration rate for diabetes drugs, that equates to 200 million individuals. Assuming a price reduction from $12,000 to $6,000 per person annually, the market for GLP-1 drugs could surpass $1 trillion, Pot explained.
Goldman Sachs estimates the market at $100-130 billion, but Pot believes it could reach $1 trillion even before considering speculative applications for cancer, Alzheimer's and Parkinson's. The market's size is consistently underestimated, with estimates rising every few months.
Patent expirations will also shape the market. In the U.S., patents for Ozempic/Wegovy run out in 2032, in Europe in 2031, and in China in 2026.
“We think it's a trillion-dollar market before the patents run out,” Pot stated.
Focus On Small-, Mid-Caps
When asked why investors should focus on a diversified basket of weight-loss-related stocks instead of a few heavyweights, Pot explained “the impact of positive clinical results in this enormous market for small and med-cap biotechs is much higher” unlike larger firms such as Eli Lilly.
He underscored the importance of having mega caps as portfolio balance but notes that smaller companies may offer higher returns in the long term.
Pot highlighted that the biotech sector is ripe with mergers and acquisitions. Last year alone, there were 22 biotech M&A deals exceeding $1 billion U.S in value.
Major health care companies without an obesity business, like Merck & Company Inc. (NYSE:MRK), Pfizer Inc. (NYSE:PFE), and Abbvie Inc. (NYSE:ABBV), are likely to seek acquisitions.
Read Now:
Photo: Shutterstock
