Fractyl Health Reports Positive 6-Month REVEAL-1 Open-Label Results Showing Sustained Post-GLP-1 Weight Maintenance After a Single Revita® Procedure
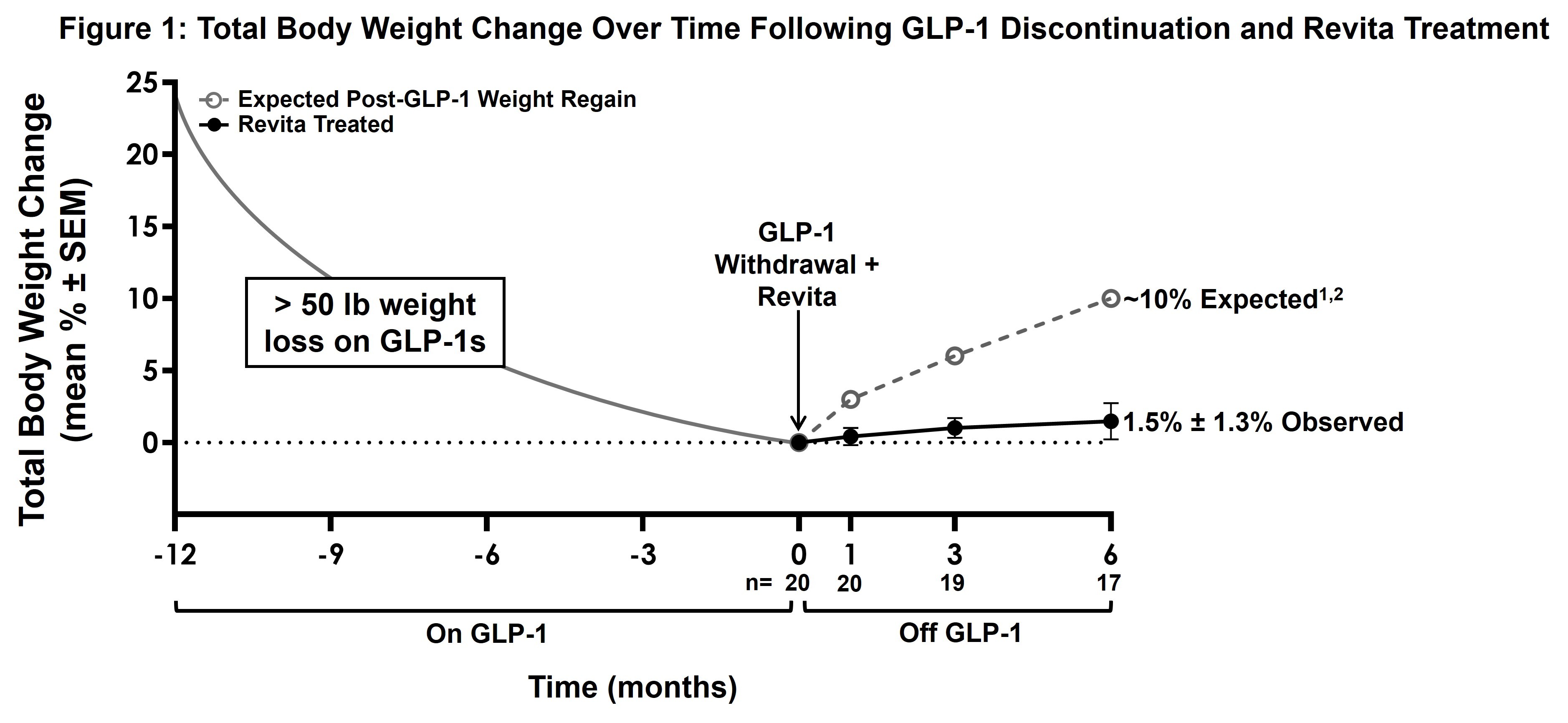
Participants who lost 24% total body weight (>50 lbs.) on GLP-1 drugs maintained stable weight 6 months after GLP-1 discontinuation and single Revita treatment
1.5% mean weight change observed with Revita (n=17); published third-party studies after GLP-1 withdrawal alone have shown ~10% weight regain at similar time points
Upcoming randomized 6-month REMAIN-1 Midpoint Cohort data expected in January 2026; topline 6-month Pivotal Cohort data and potential PMA filing anticipated in H2 2026
BURLINGTON, Mass., Dec. 02, 2025 (GLOBE NEWSWIRE) -- Fractyl Health, Inc. (NASDAQ:GUTS) (the Company or Fractyl), a metabolic therapeutics company focused on pioneering pattern-breaking approaches that treat root causes of obesity and type 2 diabetes (T2D), today announced positive 6-month results from its open-label REVEAL-1 Cohort. The REVEAL-1 Cohort is an open-label study in individuals with obesity who have lost at least 15% of their total body weight on a GLP-1 medication and who either need or choose to discontinue GLP-1 therapy. The REVEAL-1 data show that patients who underwent a single Revita procedure after discontinuing GLP-1 therapy maintained both stable body weight and glycemic control through 6 months. The new results highlight Revita's potential as a compelling first-in-class therapy for post-GLP-1 weight maintenance.
Key Findings:
- Hard-to-treat GLP-1 responder population: Participants lost 24% total body weight (>50 lbs.) on GLP-1 drugs prior to enrollment (duration on GLP-1 therapy ranging from 5 months to 5 years.) Safety and efficacy data through 6 months are available for 22 and 17 participants, respectively.
- Sustained weight maintenance: Participants maintained stable weight after a single Revita procedure, with a mean total body weight change of 1.5% ± 1.3% (SEM; n=17) at 6 months. Published third-party studies1,2 report ~10% weight regain by this time point after GLP-1 withdrawal alone (see Figure 1.)
- Sustained glycemic control: Minimal change in HbA1c levels was observed after the Revita procedure (0.04% ± 0.08%; SEM; n=17), compared to the ~0.4% increase in HbA1c seen post-GLP-1 discontinuation in the STEP-1 trial extension2 of GLP-1 withdrawal. These results indicate the potential for Revita to help stabilize cardiometabolic parameters beyond weight loss alone.
- Stable metabolic trajectory from 1 month to 3 months to 6 months: Mean body weight and HbA1c curves showed a stable and durable trajectory over time, consistent with prior Revita clinical study and real-world experience. These results are encouraging for potential longer-term durability of weight maintenance and metabolic control.
- Excellent tolerability: No procedure-related serious adverse events were observed; 8 of 22 participants (36%) experienced mild treatment-emergent adverse events which were transient and self-limited; all consistent with prior Revita experience and similar to routine upper endoscopy findings.
- Reproducible procedure performance: Average ablation length was approximately 16 cm (n=22), consistent with Revita European real-world experience and the ongoing REMAIN-1 Pivotal study, supporting the potential translatability of results and scalability of technique.
"Many patients desperately need an off-ramp from GLP-1 drugs. These six-month REVEAL-1 results are an important milestone in our understanding of the potential durability and trajectory of post-GLP-1 weight and metabolic maintenance with Revita," said Adarsh M. Thaker, M.D., Assistant Clinical Professor, Medicine, David Geffen School of Medicine at UCLA. "We know from experience that most patients begin regaining weight within weeks of stopping GLP-1 therapy, so seeing such sustained effects out to six months after a single Revita procedure is remarkable. This durability points to Revita's potential as an important new strategy for long-term metabolic health and a new path forward for patients seeking to stay off chronic drug therapy."
"These new data underscore Revita's potential to support weight maintenance after GLP-1 discontinuation," said Harith Rajagopalan, M.D., Ph.D., Co-Founder and Chief Executive Officer of Fractyl Health. "REVEAL-1 builds on the strong foundation we have established over years of development and gives us even greater confidence as we approach a pivotal year ahead. Revita has obtained FDA Breakthrough Device designation for post-GLP-1 weight maintenance, underscoring the urgency of the medical problem. We have seen rapid clinical enrollment in our pivotal REMAIN-1 study and have also observed an urgent need from patients, prescribers, and payers for a scalable, non-drug alternative in obesity. We are executing with focus and discipline, and the momentum across our programs continues to accelerate as we move closer to bringing this product candidate to patients who need it most."
Advancing Toward Value-Driving Catalysts in 2026
Fractyl is advancing toward multiple anticipated clinical readouts from the ongoing REMAIN weight maintenance program:
- January 2026: 6-month randomized data from the REMAIN-1 Midpoint Cohort
- Q2 2026: 1-year REVEAL-1 Cohort data
- Q3 2026: 1-year REMAIN-1 Midpoint Cohort data
- H2 2026: Topline 6-month randomized data from REMAIN-1 Pivotal Cohort
- H2 2026: Potential Revita PMA filing in post-GLP-1 weight maintenance
About REVEAL-1
The REVEAL-1 Cohort (n=22) is an open-label study in individuals living with obesity who have lost at least 15% of their total body weight on a GLP-1 medication and who either need or choose to discontinue GLP-1 therapy. After stopping the GLP-1 drug, participants receive the Revita treatment in an open-label setting. REVEAL-1 is designed to provide early, real-world insights on how Revita performs after GLP-1 discontinuation.
To date, 22 participants have been treated in the REVEAL-1 Cohort and are included in safety analyses, with 6-month follow-up efficacy data reported for 17. Three participants withdrew or were lost to follow up and two participants experienced protocol deviations. The profile of the REVEAL-1 Cohort closely mirrors that of the REMAIN-1 Midpoint and Pivotal Cohorts, with an average age of 50 and a gender distribution of 86% women. All 22 participants had previously been treated with a GLP-1 therapy for durations ranging from approximately 5 months to 5 years, with a mean total body weight loss of 24% while on therapy and a mean body weight of 80 kg ± 3 kg (SEM) at the time of intervention.
About Fractyl Health
Fractyl Health is a metabolic therapeutics company focused on pioneering new approaches to the treatment of metabolic diseases, including obesity and T2D. Despite advances in treatment over the last 50 years, obesity and T2D continue to be rapidly growing drivers of morbidity and mortality in the 21st century. Fractyl's goal is to transform metabolic disease treatment from chronic symptomatic management to durable disease-modifying therapies that target the organ-level root causes of disease. The Company has a robust and growing IP portfolio, with 35 granted U.S. patents and approximately 45 pending U.S. applications, along with numerous foreign issued patents and pending applications. Fractyl is based in Burlington, MA. For more information, visit www.fractyl.com.
About Revita®
Fractyl Health's lead product candidate, Revita, is based on the company's insights surrounding the potential role of the gut in obesity. Revita is designed to remodel the duodenal lining via hydrothermal ablation (i.e. duodenal mucosal resurfacing) to reverse damage to intestinal nutrient sensing and signaling mechanisms caused by chronic high-fat and high-sugar diets that are a root cause of metabolic disease. In the U.S., Revita is for investigational use only under U.S. law. Revita has U.S. FDA Breakthrough Device designation in weight maintenance for people with obesity who discontinue GLP-1 based drugs. A pivotal study of Revita in patients with obesity after discontinuation of GLP-1 based drugs, called REMAIN-1, was initiated in the third quarter of 2024 and has completed enrollment.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact are forward-looking statements. These statements may be identified by words such as "aims," "anticipates," "believes," "could," "estimates," "expects," "forecasts," "goal," "intends," "may," "plans," "possible," "potential," "seeks," "will" and variations of these words or similar expressions that are intended to identify forward-looking statements, although not all forward-looking statements contain these words. Forward-looking statements in this press release include, without limitation, statements regarding the promise and potential impact of our product candidates, including Revita's potential for preserving weight loss after GLP-1 drug discontinuation; the design, initiation, timing and results of clinical enrollment and any clinical studies or readouts, including readouts from the REVEAL-1 Cohort, REMAIN-1 Midpoint Cohort and REMAIN-1 Pivotal Cohort; the potential treatment population or benefits for any of our product candidates or products; our strategic and product development objectives and goals, including anticipated regulatory filings; and the timing of any of the foregoing. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause the Company's actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: the Company's limited operating history; the incurrence of significant net losses and the fact that the Company expects to continue to incur significant net losses for the foreseeable future; the Company's need for substantial additional financing; the Company's ability to continue as a going concern; the restrictive and financial covenants in the Company's credit agreement; the lengthy and unpredictable regulatory approval process for the Company's product candidates; uncertainty regarding its clinical studies; the fact that the Company's product candidates may cause serious adverse events or undesirable side effects or have other properties that may cause it to suspend or discontinue clinical studies, delay or prevent regulatory development, prevent their regulatory approval, limit the commercial profile, or result in significant negative consequences; the Company's reliance on third parties to conduct certain aspects of the Company's preclinical studies and clinical studies; the Company's reliance on third parties for the manufacture of sub-assembly components for Revita; the regulatory approval process of the FDA and comparable foreign regulatory authorities is lengthy, time-consuming and inherently unpredictable, and even if we complete the necessary clinical studies, we cannot predict when, or if, we will obtain regulatory approval or certification for any of our product candidates, and any such regulatory approval or certification may be for a more narrow indication than we seek; and the potential launch or commercialization of any of the Company's product candidates or products and our strategic and product development objectives and goals, and the other factors discussed under the caption "Risk Factors" in our Annual Report on Form 10-K for the year ended December 31, 2024 and Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, filed with the Securities and Exchange Commission on November 12, 2025 and in our other filings with the SEC. These forward-looking statements are based on management's current estimates and expectations. While the Company may elect to update such forward-looking statements at some point in the future, the Company disclaims any obligation to do so, even if subsequent events cause its views to change.
Contacts
Media Contact
Jessica Cotrone, Head of Corporate Communications
[email protected], 978.760.5622
Investor Contact
Brian Luque, Head of Investor Relations and Corporate Development
[email protected], 951.206.1200
1 Aronne et al. JAMA. 2024;331(1):38–48. doi:10.1001/jama.2023.24945
2 Wilding et al. Diabetes Obes Metab. 2022 Aug;24(8):1553-1564
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/c67dc7a4-491a-4ec3-b0be-64970ea07657


