Lantern Advances Drug Candidate LP-184 with IND Clearance for Phase 1b/2 Clinical Trial in Triple Negative Breast Cancer (TNBC)
- The FDA has cleared the investigational new drug (IND) application of LP-184 in triple negative breast cancer (TNBC) treatment – a novel small molecule advanced and developed with Lantern's AI platform, RADR®
- The phase 1b/2 clinical trial plans to evaluate LP-184 in recurrent TNBC patients as both monotherapy and in combination with olaparib.
- This achievement follows the FDA's Orphan Drug Designation for LP-184 in TNBC in 2023 and FDA Fast Track Designation for LP-184 in TNBC in 2024.
- The average survival for newly diagnosed, metastatic TNBC is estimated at 18 months and presents an annual market opportunity in excess of $4 billion USD.
Lantern Pharma Inc. (NASDAQ:LTRN), an artificial intelligence company developing targeted and transformative cancer therapies using its proprietary AI platform, RADR®, today announced that it has received clearance of its Investigational New Drug Application (IND) from the U.S. Food and Drug Administration (FDA) for a Phase 1b/2 clinical trial for LP-184 in Triple Negative Breast Cancer. This achievement builds on the previous regulatory momentum including Orphan Drug Designation in 2023 and Fast Track Designation in 20241.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250505715111/en/
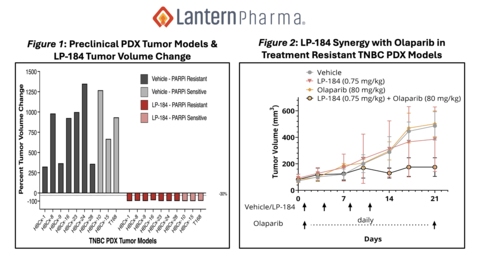
Figure 1: Preclinical PDX Tumor Models & LP-184 Tumor Volume Change. Figure 2: LP-184 Synergy with Olaparib in Treatment Resistant TNBC PDX Models.
Strategic Trial Design to Address Critical Treatment Gap in TNBC
The innovative dual-approach in the clinical trial is designed to evaluate LP-184 in recurrent, advanced-stage TNBC patients through:
- Monotherapy Arm: An open-label study involving approximately 30 patients with advanced-stage TNBC, focusing on dose optimization to evaluate, enhance and optimize safety and potential efficacy for TNBC patients.
- Combination Therapy: Evaluation of LP-184 in combination with olaparib in second-line settings for patients with advanced-stage TNBC harboring BRCA1 or BRCA2 alterations, with primary endpoints including safety and efficacy parameters that could potentially support a pathway to regulatory approval.
This strategic approach focuses on addressing a significant unmet medical need, with average survival for newly diagnosed metastatic TNBC estimated at 10 to 18 months, representing an annual market opportunity exceeding $4 billion USD.
LP-184 Background in TNBC & Mechanistic Rationale
LP-184 is a synthetically lethal small molecule that induces DNA double strand breaks upon bioactivation by the enzyme prostaglandin reductase 1 (PTGR1) in cancer cells. Preclinical studies and artificial intelligence-driven in silico modeling suggest that cancers with DDR gene alterations may preferentially respond to LP-184.2
Preclinical findings also suggest that LP-184 is particularly well positioned for TNBC with striking data from in vivo models including complete regression seen in several PARP resistant as well as PARP sensitive PDXs (see Figure 1).
Nearly 70% of TNBCs are noted to harbor deficiency in homologous recombination pathways, making them likely to be particularly sensitive tumors for targeting with drug-candidate LP-184. In addition, it is estimated that up to 46% percent of women with TNBC will develop brain metastasis3, and LP-184 has shown blood brain barrier (BBB) penetration, with evidence of activity in preclinical brain metastasis models.
LP-184 Phase 1b/2 TNBC Trial Overview - Monotherapy
The monotherapy phase 1b/2 trial is designed to evaluate LP-184 in patients with advanced-stage TNBC. The study is designed to focus on dose optimization to evaluate and enhance both safety and potential efficacy ultimately aiding in the potential determination of the best clinical position for LP-184 in advanced-stage TNBC patients. The design of the dose optimization phase, provides for evaluation of the safety, efficacy and pharmacokinetics of LP-184 using 2 dose levels in an open-label monotherapy study involving around 30 patients to be dosed with LP-184.
LP-184 Phase 1b/2 TNBC Trial Overview - Combination Therapy with PARP inhibitor
LP-184 has also been shown in preclinical studies to be highly potent in combination with the PARP inhibitor, olaparib, including in tumors that are resistant to PARP inhibitors. In preclinical studies, treatment of a HBCx-28 TNBC PDX model, with BRCA-1 LOH and an HRD score of 63 that was resistant to the PARP inhibitor, showed evidence of re-sensitization in combination with LP-184 (See Figure 2). These data, which were initially presented at the San Antonio Breast Cancer Symposium, support the clinical evaluation of LP-184 in combination with PARPi in an earlier line of treatment. Treating patients in an earlier clinical setting has the potential to reach more patients and potentially generate a more durable and deeper earlier control of the disease.
The design of the combination phase 1b/2 trial provides for LP-184 to be evaluated in a second-line setting in patients with advanced-stage TNBC whose primary tumor harbors alterations in BRCA1 or BRCA2. The primary end points of the study are expected to include safety and efficacy, with the aim of supporting a potential pathway to a regulatory approval process.
Multi-Region Clinical Strategy with Focus on High-Incidence Countries
The trials are planned to be conducted at select centers in the United States as well as academic cancer centers and institutions in India and Nigeria, where TNBC incidence rates are particularly high—comprising nearly 40% of initial breast cancer diagnoses. This strategic site selection is focused on leveraging established collaborative research networks that have a track record of successful collaborative cancer studies with US and European pharma companies. Lantern's planned objective will be to ensure proper local experience and support for this clinical trial while addressing regions with significant disease burden and high clinical demand.
"This IND clearance for LP-184 in a Phase 1b/2 study represents a pivotal advancement in our mission to bring precisely targeted, AI-developed medicines to patients with aggressive cancers and limited treatment options," said Panna Sharma, CEO and President of Lantern Pharma. "The strategic design of our clinical program reflects both the compelling mechanistic rationale and the encouraging data supporting LP-184's potential in TNBC."
Expanding Therapeutic Potential Across Multiple Indications
Beyond TNBC, LP-184 shows promise for the potential treatment of other cancers harboring DNA damage repair mutations, including lung, bladder, pancreatic, and ovarian cancers. Additional clinical trials in these targeted indications are in planning stages, with several being considered as Investigator Initiated Trials. LP-184 has received multiple Orphan Drug, Fast Track, and Rare Pediatric Disease Designations from the FDA across various solid tumor indications.
The global TNBC market is estimated at $3-5 billion USD annually, with over 300,000 new cases diagnosed worldwide each year. While homologous recombination deficient TNBCs are often initially treated with PARP inhibitors, resistance inevitably develops, underscoring the critical need for novel therapeutic approaches.
About Lantern Pharma
Lantern Pharma (NASDAQ:LTRN) is an AI company transforming the cost, pace, and timeline of oncology drug discovery and development. Our proprietary AI and machine learning (ML) platform, RADR®, leverages over 100 billion oncology-focused data points and a library of 200+ advanced ML algorithms to help solve billion-dollar, real-world problems in oncology drug development. By harnessing the power of AI and with input from world-class scientific advisors and collaborators, we have accelerated the development of our growing pipeline of drug-candidates that span multiple cancer indications, including both solid tumors and blood cancers and an antibody-drug conjugate (ADC) program. On average, our newly developed drug programs have been advanced from initial AI insights to first-in-human clinical trials in 2–3 years and at approximately $1.0 – $2.5 million per program.
Our lead development programs include a Phase 2 clinical program and multiple Phase 1 clinical trials. We have also established a wholly-owned subsidiary, Starlight Therapeutics, to focus exclusively on the clinical execution of our promising therapies for CNS and brain cancers, many of which have no effective treatment options. Our AI-driven pipeline of innovative product candidates is estimated to have a combined annual market potential of over $15 billion USD and have the potential to provide life-changing therapies to hundreds of thousands of cancer patients across the world.
Forward-Looking Statements:
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward-looking statements include, among other things, statements relating to: future events or our future financial performance; the potential advantages of our RADR® platform in identifying drug candidates and patient populations that are likely to respond to a drug candidate; our strategic plans to advance the development of our drug candidates and antibody drug conjugate (ADC) development program; estimates regarding the development timing for our drug candidates and ADC development program; expectations and estimates regarding clinical trial timing and patient enrollment; our research and development efforts of our internal drug discovery programs and the utilization of our RADR® platform to streamline the drug development process; our intention to leverage artificial intelligence, machine learning and genomic data to streamline and transform the pace, risk and cost of oncology drug discovery and development and to identify patient populations that would likely respond to a drug candidate; estimates regarding patient populations, potential markets and potential market sizes; sales estimates for our drug candidates and our plans to discover and develop drug candidates and to maximize their commercial potential by advancing such drug candidates ourselves or in collaboration with others. Any statements that are not statements of historical fact (including, without limitation, statements that use words such as "anticipate," "believe," "contemplate," "could," "estimate," "expect," "intend," "seek," "may," "might," "plan," "potential," "predict," "project," "target," "model," "objective," "aim," "upcoming," "should," "will," "would," or the negative of these words or other similar expressions) should be considered forward-looking statements. There are a number of important factors that could cause our actual results to differ materially from those indicated by the forward-looking statements, such as (i) the risk that we may not be able to secure sufficient future funding when needed and as required to advance and support our existing and planned clinical trials and operations, (ii) the risk that observations in preclinical studies and early or preliminary observations in clinical studies do not ensure that later observations, studies and development will be consistent or successful, (iii) the risk that our research and the research of our collaborators may not be successful, (iv) the risk that we may not be successful in licensing potential candidates or in completing potential partnerships and collaborations, (v) the risk that none of our product candidates has received FDA marketing approval, and we may not be able to successfully initiate, conduct, or conclude clinical testing for or obtain marketing approval for our product candidates, (vi) the risk that no drug product based on our proprietary RADR® AI platform has received FDA marketing approval or otherwise been incorporated into a commercial product, and (vii) those other factors set forth in the Risk Factors section in our Annual Report on Form 10-K for the year ended December 31, 2024, filed with the Securities and Exchange Commission on March 27, 2025. You may access our Annual Report on Form 10-K for the year ended December 31, 2024 under the investor SEC filings tab of our website at www.lanternpharma.com or on the SEC's website at www.sec.gov. Given these risks and uncertainties, we can give no assurances that our forward-looking statements will prove to be accurate, or that any other results or events projected or contemplated by our forward-looking statements will in fact occur, and we caution investors not to place undue reliance on these statements. All forward-looking statements in this press release represent our judgment as of the date hereof, and, except as otherwise required by law, we disclaim any obligation to update any forward-looking statements to conform the statement to actual results or changes in our expectations.
| _____________________________ |
2 https://ascopubs.org/doi/10.1200/JCO.2021.39.15_suppl.e15064 |
3 https://onlinelibrary.wiley.com/doi/full/10.1155/2024/8816102 |
View source version on businesswire.com: https://www.businesswire.com/news/home/20250505715111/en/
Investor Relations
[email protected]
(972) 277-1136
