PolyPid Announces Positive Topline Results from Phase 3 SHIELD II Trial: D-PLEX₁₀₀ Demonstrated Significant Reduction in Surgical Site Infections and Successfully Met Primary and All Key Secondary Endpoints
- D-PLEX100 successfully met the primary efficacy endpoint, with statistically significant results (p<0.005) in 798 patients with large abdominal surgery incisions.
- The trial successfully met all key secondary efficacy endpoints, including a 58% reduction in the rate of surgical site infections ("SSI") in patients treated with D-PLEX100 arm versus standard of care ("SoC") arm (p<0.005).
- The Company expects to submit a New Drug Application ("NDA") to the U.S. Food and Drug Administration ("FDA") in early 2026, with a Marketing Authorization Application ("MAA") in the E.U. to follow shortly thereafter.
- PolyPid will conduct a conference call and webcast at 8:30 A.M. ET today.
PETACH TIKVA, Israel, June 09, 2025 (GLOBE NEWSWIRE) -- PolyPid Ltd. (NASDAQ:PYPD) ("PolyPid" or the "Company"), a late-stage biopharma company aiming to improve surgical outcomes, today announced positive topline results from its pivotal SHIELD II Phase 3 trial of D-PLEX100 for the prevention of SSIs in patients undergoing abdominal colorectal surgery with large incisions, which has received Fast Track designation from the FDA.
"The positive results of our Phase 3 pivotal trial provide clear and compelling evidence of D-PLEX100's ability to significantly reduce SSIs, a major burden on patients and healthcare systems, while improving clinical outcomes for both patients and physicians," said Dikla Czaczkes Akselbrad, Chief Executive Officer of PolyPid. "D-PLEX100 has the potential to meaningfully lower costs for hospitals and payers, and we believe it will be a valuable addition to the standard of care. Targeting a total addressable U.S. market of over 12 million annual surgeries and a significant unmet need, D-PLEX100 has the potential to make a substantial market impact. We remain on track for an NDA submission in early 2026 and believe these robust data will serve as a strong catalyst for advancing our global partnership discussions. We are deeply grateful to the patients, principal investigators, and study teams who made this milestone possible."
Key findings from SHIELD II study include:
- The primary efficacy endpoint of the study was successfully met, with a significantly lower proportion of primary endpoint events among patients who received D-PLEX100 plus SoC (n=405; 10.9%), compared to SoC alone (n=393; 18.1%), representing a 38% reduction (p<0.005). The primary endpoint is the combination of deep and superficial SSIs, all-cause mortality and surgical reinterventions at the same incision of the original index surgery.
- SHIELD II included three key secondary endpoints:
- The first key secondary endpoint was met, with a 58% reduction in deep and superficial SSI rates among patients who received D-PLEX100 plus SoC (3.8%) compared to those who received SoC alone (9.5%) (p<0.005).
- The second key secondary endpoint showed statistical significance in favor of D-PLEX100 plus SoC over SoC alone (p<0.005). This efficacy endpoint combined SSI, mortality and reintervention as evaluated in the primary endpoint. This endpoint was assessed in the overall study population of 975 patients with an incision ≥7 cm, including laparoscopic surgery patients enrolled prior to the 2023 trial protocol change.
- The third key secondary endpoint was met with a 62% reduction of patients with an ASEPSIS1 score >20 in D-PLEX100 plus SoC arm compared to SoC alone arm (p<0.05). The ASEPSIS score is a clinical tool used to objectively assess surgical wound infections.
- The first key secondary endpoint was met, with a 58% reduction in deep and superficial SSI rates among patients who received D-PLEX100 plus SoC (3.8%) compared to those who received SoC alone (9.5%) (p<0.005).
- There were no safety concerns raised by the independent Data Safety Monitoring Board in SHIELD II.
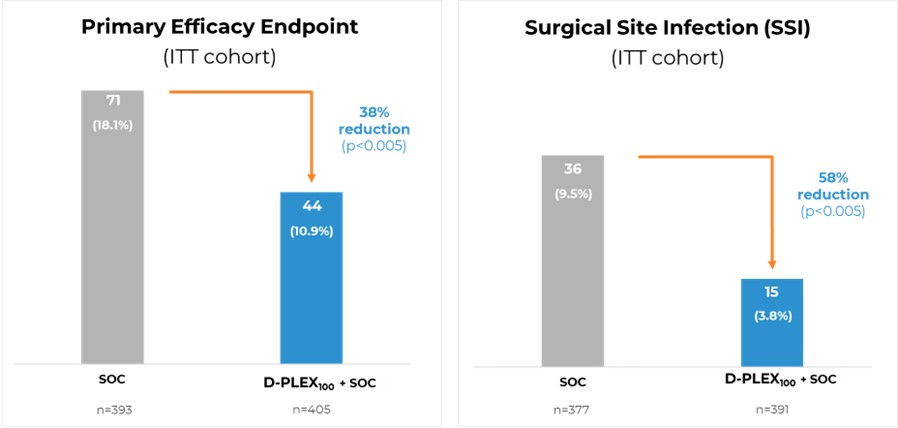
Image: SHIELD II Results on Primary Endpoint and First Key Secondary Endpoint
"The risk of surgical site infection in the colorectal patient population has recently been documented to approach 23%2 in patients with multiple comorbid risk factors," said Charles E. Edmiston, Jr., MS, PhD, CIC, FIDSA, FSIS, FAPIC Emeritus Professor of Surgery. "These topline results provide clinical practitioners with a novel, innovative strategy for reducing postoperative infection risk in patients undergoing colorectal surgery with large wound incisions. Based on the trial findings, particularly the significant risk-reduction benefit by reducing in-wound sepsis risk, I anticipate D-PLEX100 technology will be rapidly embraced as a critical component to the current evidence-based surgical care bundle for colorectal surgery following FDA approval."
Additional results from the SHIELD II trial will be presented at an upcoming medical conference.
Conference Call Dial-In & Webcast Information:
| Date: | Monday, June 9, 2025 |
| Time: | 8:30 A.M. Eastern Time |
| Conference Call: | https://register-conf.media-server.com/register/BIb5bb85aa1ffc4940b78f7191b278ca77 |
| Webcast: | https://edge.media-server.com/mmc/p/k6n5hrft |
A replay of the presentation will be available on the Company's Investor Relations Events and Presentations webpage following the event.
About SHIELD II
SHIELD II (Surgical site Hospital acquired Infection prEvention with Local D-PLEX) is a prospective, multinational, randomized, double blind Phase 3 trial designed to assess the efficacy and safety of D-PLEX100 administered concomitantly with standard of care ("SoC"), which includes prophylactic systemic antibiotics, compared to SoC alone arm, in the prevention of post abdominal-surgery incisional infection in patients undergoing abdominal colorectal surgeries with large incisions. The primary endpoint of the trial is measured by the proportion of subjects with either a surgical site infection ("SSI") event as determined by a blinded and independent adjudication committee, reintervention, or mortality for any reason within 30 days post-surgery. Patient safety will be monitored for an additional 30 days. The trial will enroll patients in centers in the United States, Europe and Israel.
About D-PLEX100
D-PLEX100, PolyPid's lead product candidate, is designed to provide local prolonged and controlled anti-bacterial activity directly at the surgical site to prevent SSIs. Following the administration of D-PLEX100 into the surgical site, the PLEX (Polymer-Lipid Encapsulation matriX) technology pairs with Active Pharmaceutical Ingredients, enabling a prolonged and continuous release of the broad-spectrum antibiotic doxycycline, resulting in a high local concentration of the drug for a period of 30 days for the prevention of SSIs, with additional potential to prevent SSIs caused by antibiotic-resistant bacteria at the surgical site. D-PLEX100 received Breakthrough Therapy designation from the U.S. Food and Drug Administration for the prevention of SSIs in patients undergoing elective colorectal surgery. D-PLEX100 is currently in Phase 3 SHIELD II trial for the prevention of surgical site infections in patients undergoing abdominal colorectal surgery with large incisions.
About PolyPid
PolyPid Ltd. (Nasdaq: PYPD) is a late-stage biopharma company aiming to improve surgical outcomes. Through locally administered, controlled, prolonged-release therapeutics, PolyPid's proprietary PLEX (Polymer-Lipid Encapsulation matriX) technology pairs with Active Pharmaceutical Ingredients (APIs), enabling precise delivery of drugs at optimal release rates over durations ranging from several days to months. PolyPid's lead product candidate D-PLEX100 is in a Phase 3 clinical trial for the prevention of abdominal colorectal surgical site infections. In addition, the Company is currently in preclinical stages to test the efficacy of OncoPLEX for the treatment of solid tumors, beginning with glioblastoma.
For additional Company information, please visit http://www.polypid.com and follow us on Twitter and LinkedIn.
Forward-looking Statements
This press release contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act and other securities laws. Words such as "expects," "anticipates," "intends," "plans," "believes," "seeks," "estimates" and similar expressions or variations of such words are intended to identify forward-looking statements. For example, the Company is using forward-looking statements when it discusses the safety, efficacy and benefits of D-PLEX100, D-PLEX100's potential to meaningfully lower costs for hospitals and payers, the expected submissions of an NDA to the FDA and MAA and the timing thereof; the Company's belief that the data results will serve as a strong catalyst for advancing its global partnership discussions; D-PLEX100 potential to make a substantial market impact; and the anticipation that D-PLEX100 technology will be rapidly embraced as a critical component to the current evidence-based surgical care bundle for colorectal surgery following FDA approval. Forward-looking statements are not historical facts, and are based upon management's current expectations, beliefs and projections, many of which, by their nature, are inherently uncertain. Such expectations, beliefs and projections are expressed in good faith. However, there can be no assurance that management's expectations, beliefs and projections will be achieved, and actual results may differ materially from what is expressed in or indicated by the forward-looking statements. Forward-looking statements are subject to risks and uncertainties that could cause actual performance or results to differ materially from those expressed in the forward-looking statements. For a more detailed description of the risks and uncertainties affecting the Company, reference is made to the Company's reports filed from time to time with the Securities and Exchange Commission, including, but not limited to, the risks detailed in the Company's Annual Report on Form 20-F filed on February 26, 2025. Forward-looking statements speak only as of the date the statements are made. The Company assumes no obligation to update forward-looking statements to reflect actual results, subsequent events or circumstances, changes in assumptions or changes in other factors affecting forward-looking information except to the extent required by applicable securities laws. If the Company does update one or more forward-looking statements, no inference should be drawn that the Company will make additional updates with respect thereto or with respect to other forward-looking statements.
References and links to websites have been provided as a convenience, and the information contained on such websites is not incorporated by reference into this press release. PolyPid is not responsible for the contents of third-party websites.
Contacts:
PolyPid Ltd.
Ori Warshavsky
COO – US
908-858-5995
[email protected]
Investors:
Brian Ritchie
LifeSci Advisors
212-915-2578
[email protected]
1 ASEPSIS is an acronym of wound assessment and treatment parameters, which provides a numerical score during an inspection of the surgical site. The final score is interpreted by the severity of wound appearance and the clinical consequences of the infection. Parameters include: serous exudate, erythema, purulent exudate, separation of deep tissue and also antibiotic therapy, drainage of pus under local/general anesthesia, isolation of pathogenic bacteria and hospital stay as inpatient.
2 David J. Leaper, D.Sc., M.D., Chantal E. Holy, M.Sc., Ph.D., Maureen Spencer, M.Ed., Abhishek Chitnis, Ph.D., Andrew Hogan, M.Sc., George W.J. Wright, Ph.D., Brian Po-Han Chen, Sc.M., Charles E. Edmiston, Jr, Ph.D. Assessment of the Risk and Economic Burden of Surgical Site Infection Following Colorectal Surgery Using a US Longitudinal Database: Is There a Role for Innovative Antimicrobial Wound Closure Technology to Reduce the Risk of Infection? Dis Colon Rectum 2020; 63:1628-1638.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/1a883ef3-5e5a-4ffc-8532-b9f87784ef42


