eigr-20231231falseFY000130525320230.033300013052532023-01-012023-12-3100013052532023-06-30iso4217:USD00013052532024-04-04xbrli:shares00013052532023-12-3100013052532022-12-31iso4217:USDxbrli:shares0001305253us-gaap:SubsequentEventMember2024-01-052024-01-05xbrli:pure0001305253us-gaap:ProductMember2023-01-012023-12-310001305253us-gaap:ProductMember2022-01-012022-12-310001305253eigr:OtherRevenueMember2023-01-012023-12-310001305253eigr:OtherRevenueMember2022-01-012022-12-3100013052532022-01-012022-12-310001305253us-gaap:CommonStockMember2021-12-310001305253us-gaap:AdditionalPaidInCapitalMember2021-12-310001305253us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-12-310001305253us-gaap:RetainedEarningsMember2021-12-3100013052532021-12-310001305253us-gaap:CommonStockMember2022-01-012022-12-310001305253us-gaap:AdditionalPaidInCapitalMember2022-01-012022-12-310001305253us-gaap:AccumulatedOtherComprehensiveIncomeMember2022-01-012022-12-310001305253us-gaap:RetainedEarningsMember2022-01-012022-12-310001305253us-gaap:CommonStockMember2022-12-310001305253us-gaap:AdditionalPaidInCapitalMember2022-12-310001305253us-gaap:AccumulatedOtherComprehensiveIncomeMember2022-12-310001305253us-gaap:RetainedEarningsMember2022-12-310001305253us-gaap:CommonStockMember2023-01-012023-12-310001305253us-gaap:AdditionalPaidInCapitalMember2023-01-012023-12-310001305253us-gaap:AccumulatedOtherComprehensiveIncomeMember2023-01-012023-12-310001305253us-gaap:RetainedEarningsMember2023-01-012023-12-310001305253us-gaap:CommonStockMember2023-12-310001305253us-gaap:AdditionalPaidInCapitalMember2023-12-310001305253us-gaap:AccumulatedOtherComprehensiveIncomeMember2023-12-310001305253us-gaap:RetainedEarningsMember2023-12-31eigr:segment0001305253us-gaap:CustomerConcentrationRiskMembereigr:CustomerAMemberus-gaap:FinanceReceivablesMember2023-01-012023-12-310001305253us-gaap:CustomerConcentrationRiskMemberus-gaap:FinanceReceivablesMembereigr:CustomerBMember2023-01-012023-12-310001305253us-gaap:CustomerConcentrationRiskMembereigr:CustomerCMemberus-gaap:FinanceReceivablesMember2023-01-012023-12-310001305253us-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMembereigr:CustomerAMember2023-01-012023-12-310001305253us-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMembereigr:CustomerBMember2023-01-012023-12-310001305253us-gaap:CustomerConcentrationRiskMembereigr:CustomerAMemberus-gaap:FinanceReceivablesMember2022-01-012022-12-310001305253us-gaap:CustomerConcentrationRiskMemberus-gaap:FinanceReceivablesMembereigr:CustomerBMember2022-01-012022-12-310001305253us-gaap:CustomerConcentrationRiskMemberus-gaap:SalesRevenueNetMembereigr:CustomerAMember2022-01-012022-12-310001305253eigr:LabEquipmentMember2023-12-310001305253us-gaap:FurnitureAndFixturesMember2023-12-310001305253us-gaap:LeaseholdImprovementsMember2023-12-310001305253eigr:ComputerEquipmentAndSoftwareMember2023-12-310001305253us-gaap:ProductMember2020-01-012020-12-3100013052532021-06-012021-06-300001305253eigr:AutorisationTemporaireDUtlisationMemberMember2023-01-012023-12-310001305253eigr:AutorisationTemporaireDUtlisationMemberMember2022-01-012022-12-310001305253us-gaap:StockOptionMember2023-01-012023-12-310001305253us-gaap:StockOptionMember2022-01-012022-12-310001305253us-gaap:EmployeeStockMember2023-01-012023-12-310001305253us-gaap:EmployeeStockMember2022-01-012022-12-310001305253us-gaap:RestrictedStockUnitsRSUMember2023-01-012023-12-310001305253us-gaap:RestrictedStockUnitsRSUMember2022-01-012022-12-310001305253us-gaap:FairValueInputsLevel3Member2023-12-310001305253us-gaap:FairValueInputsLevel3Member2022-12-310001305253us-gaap:FairValueInputsLevel3Member2022-01-012022-12-310001305253us-gaap:FairValueInputsLevel3Member2023-01-012023-12-310001305253us-gaap:FairValueInputsLevel1Memberus-gaap:MoneyMarketFundsMember2023-12-310001305253us-gaap:FairValueInputsLevel1Memberus-gaap:MoneyMarketFundsMember2022-12-310001305253us-gaap:CommercialPaperMemberus-gaap:FairValueInputsLevel2Member2022-12-310001305253us-gaap:FairValueInputsLevel2Membereigr:USGovernmentBondsMember2022-12-310001305253us-gaap:FairValueInputsLevel2Memberus-gaap:CorporateDebtSecuritiesMember2022-12-310001305253us-gaap:FairValueInputsLevel2Memberus-gaap:CommercialPaperMember2022-12-310001305253eigr:ComputerEquipmentAndSoftwareMember2022-12-310001305253us-gaap:ConstructionInProgressMember2023-12-310001305253us-gaap:ConstructionInProgressMember2022-12-310001305253eigr:LabEquipmentMember2022-12-310001305253us-gaap:FurnitureAndFixturesMember2022-12-310001305253us-gaap:LeaseholdImprovementsMember2022-12-310001305253eigr:UPennCHOPAgreementMember2019-01-012019-12-310001305253eigr:UPennCHOPAgreementMembersrt:MaximumMember2019-01-012019-12-310001305253eigr:CommercialSalesMembereigr:UPennCHOPAgreementMembersrt:MaximumMember2019-01-012019-12-310001305253eigr:CommercialSalesMembereigr:UPennCHOPAgreementMembersrt:MaximumMember2023-01-012023-12-310001305253eigr:ProductDevelopmentAgreementMember2018-08-112018-08-110001305253eigr:ProductDevelopmentAgreementMember2018-08-110001305253eigr:ProductDevelopmentAgreementMember2022-01-012022-12-310001305253eigr:PRFCollaborationAgreementMember2018-05-152018-05-150001305253us-gaap:LicensingAgreementsMember2016-04-012016-04-300001305253eigr:BMSTransactionMemberus-gaap:CommonStockMembereigr:PurchaseAgreementMember2016-04-012016-04-300001305253eigr:DevelopmentAndRegulatoryMilestonesMembereigr:BMSTransactionMemberus-gaap:LicensingAgreementsMember2016-04-012016-04-300001305253eigr:BMSTransactionMembereigr:CommercialSalesMembersrt:MaximumMemberus-gaap:LicensingAgreementsMember2016-04-012016-04-300001305253eigr:DevelopmentPhaseTwoMembereigr:BMSTransactionMemberus-gaap:LicensingAgreementsMember2020-10-012020-12-310001305253eigr:DevelopmentPhaseThreeMembereigr:BMSTransactionMemberus-gaap:LicensingAgreementsMember2022-03-012022-03-310001305253us-gaap:ContractBasedIntangibleAssetsMembereigr:MerckTransactionMember2010-09-012010-09-300001305253us-gaap:ContractBasedIntangibleAssetsMembereigr:MerckTransactionMember2010-09-300001305253srt:MaximumMembereigr:MerckTransactionMemberus-gaap:ResearchAndDevelopmentExpenseMemberus-gaap:LicensingAgreementsMember2010-09-012010-09-300001305253eigr:MerckTransactionMemberus-gaap:LicensingAgreementsMember2018-05-152018-05-150001305253eigr:MerckTransactionMemberus-gaap:LicensingAgreementsMember2023-09-300001305253eigr:AvexitidePurchaseAgreementMember2015-09-012015-09-300001305253eigr:AvexitidePurchaseAgreementMembereigr:ServiceBasedPerformanceVestingConditionMember2015-09-300001305253eigr:AvexitidePurchaseAgreementMembereigr:MilestoneBasedPerformanceVestingConditionMember2015-09-300001305253eigr:AvexitidePurchaseAgreementMembersrt:MaximumMember2015-09-012015-09-300001305253eigr:AvexitidePurchaseAgreementMember2015-09-012023-12-310001305253eigr:InnovatusLoanMemberus-gaap:LineOfCreditMemberus-gaap:SecuredDebtMember2022-06-01eigr:tranche0001305253eigr:VariableRateComponentOneMembereigr:InnovatusLoanMembersrt:MinimumMemberus-gaap:LineOfCreditMemberus-gaap:SecuredDebtMember2022-06-012022-06-010001305253eigr:InnovatusLoanMemberus-gaap:LineOfCreditMembereigr:VariableRateComponentTwoMemberus-gaap:SecuredDebtMember2022-06-012022-06-010001305253eigr:InnovatusLoanMembersrt:MinimumMemberus-gaap:LineOfCreditMemberus-gaap:SecuredDebtMember2022-06-010001305253eigr:InnovatusLoanMemberus-gaap:LineOfCreditMembersrt:MaximumMemberus-gaap:SecuredDebtMember2022-06-010001305253eigr:InnovatusLoanMembereigr:SecuredDebtTrancheAMemberus-gaap:LineOfCreditMember2022-06-300001305253eigr:InnovatusLoanMemberus-gaap:LineOfCreditMembereigr:SecuredDebtTrancheBAndCMember2022-06-300001305253eigr:InnovatusLoanMemberus-gaap:LineOfCreditMembereigr:SecuredDebtTrancheBMember2022-06-300001305253eigr:InnovatusLoanMemberus-gaap:LineOfCreditMemberus-gaap:SecuredDebtMember2023-01-012023-12-310001305253us-gaap:LineOfCreditMembereigr:InnovatusStockPurchaseAgreementMemberus-gaap:SecuredDebtMember2022-06-012022-06-010001305253us-gaap:LineOfCreditMembereigr:InnovatusStockPurchaseAgreementMemberus-gaap:SecuredDebtMember2022-06-010001305253us-gaap:LineOfCreditMembereigr:OxfordLoanMemberus-gaap:SecuredDebtMember2022-06-010001305253eigr:SecuredDebtAmendedTrancheAMemberus-gaap:LineOfCreditMembereigr:AmendedOxfordLoanMember2022-06-010001305253eigr:SecuredDebtAmendedTrancheAMemberus-gaap:LineOfCreditMembereigr:AmendedOxfordLoanMember2022-06-012022-06-010001305253eigr:SecuredDebtAmendedTrancheAMemberus-gaap:LineOfCreditMembereigr:AmendedOxfordLoanMember2022-01-012022-12-310001305253eigr:AmendedTrancheAMembereigr:InnovatusLoanMember2023-12-3100013052532020-12-012020-12-310001305253eigr:AtTheMarketOfferingMember2020-12-012020-12-310001305253eigr:AtTheMarketOfferingMember2022-01-012022-03-310001305253srt:MaximumMember2022-03-250001305253eigr:AtTheMarketOfferingMember2022-03-252022-03-250001305253srt:MinimumMembereigr:RestatedTwoThousandThirteenEquityIncentivePlanMemberMember2023-01-012023-12-310001305253srt:MinimumMembereigr:RestatedTwoThousandThirteenEquityIncentivePlanMemberMembereigr:HoldersOfTenPercentOrMoreOfVotingPowerMember2023-01-012023-12-310001305253eigr:RestatedTwoThousandThirteenEquityIncentivePlanMemberMembersrt:MaximumMember2023-01-012023-12-310001305253eigr:RestatedTwoThousandThirteenEquityIncentivePlanMemberMember2023-01-012023-12-310001305253eigr:RestatedTwoThousandThirteenEquityIncentivePlanMemberMember2023-12-310001305253eigr:InducementPlan2021Member2021-06-300001305253eigr:InducementPlan2021Member2023-12-310001305253us-gaap:StockOptionMember2023-01-012023-12-310001305253us-gaap:StockOptionMember2022-01-012022-12-310001305253srt:MinimumMemberus-gaap:EmployeeStockOptionMember2023-01-012023-12-310001305253srt:MaximumMemberus-gaap:EmployeeStockOptionMember2023-01-012023-12-310001305253srt:MinimumMemberus-gaap:EmployeeStockOptionMember2022-01-012022-12-310001305253srt:MaximumMemberus-gaap:EmployeeStockOptionMember2022-01-012022-12-310001305253us-gaap:EmployeeStockOptionMember2023-01-012023-12-310001305253us-gaap:EmployeeStockOptionMember2022-01-012022-12-310001305253eigr:TwoThousandThirteenEmployeeStockPurchasePlanMember2016-12-3100013052532016-01-012016-12-310001305253eigr:TwoThousandThirteenEmployeeStockPurchasePlanMember2023-12-310001305253us-gaap:RestrictedStockUnitsRSUMember2020-01-012020-03-310001305253srt:DirectorMemberus-gaap:RestrictedStockUnitsRSUMember2020-01-012020-03-310001305253eigr:EmployeesMemberus-gaap:RestrictedStockUnitsRSUMemberus-gaap:ShareBasedCompensationAwardTrancheOneMember2020-01-012020-03-310001305253eigr:EmployeesMemberus-gaap:RestrictedStockUnitsRSUMemberus-gaap:ShareBasedCompensationAwardTrancheTwoMember2020-01-012020-03-310001305253eigr:EmployeesMemberus-gaap:RestrictedStockUnitsRSUMemberus-gaap:ShareBasedCompensationAwardTrancheThreeMember2020-01-012020-03-310001305253eigr:PerformanceStockUnitsPSUMember2023-01-012023-12-310001305253eigr:PerformanceStockUnitsPSUMember2022-01-012022-12-310001305253eigr:FourTerminatedEmployeesMembereigr:PerformanceStockUnitsPSUMember2023-05-012023-05-310001305253us-gaap:RestrictedStockUnitsRSUMember2023-01-012023-12-310001305253us-gaap:RestrictedStockUnitsRSUMember2022-01-012022-12-310001305253eigr:RestrictedStockUnitsAndPerformanceStockUnitMember2022-12-310001305253eigr:RestrictedStockUnitsAndPerformanceStockUnitMember2023-01-012023-12-310001305253eigr:RestrictedStockUnitsAndPerformanceStockUnitMember2023-12-310001305253eigr:UnvestedStockOptionsPerformanceBasedRestrictedStockUnitsAndTimeBasedRSUsMembereigr:FormerCEOMember2023-02-060001305253eigr:UnvestedStockOptionsPerformanceBasedRestrictedStockUnitsAndTimeBasedRSUsMembereigr:FormerCEOMember2023-01-012023-12-310001305253us-gaap:ResearchAndDevelopmentExpenseMember2023-01-012023-12-310001305253us-gaap:ResearchAndDevelopmentExpenseMember2022-01-012022-12-310001305253us-gaap:SellingGeneralAndAdministrativeExpensesMember2023-01-012023-12-310001305253us-gaap:SellingGeneralAndAdministrativeExpensesMember2022-01-012022-12-310001305253us-gaap:DomesticCountryMember2023-12-310001305253us-gaap:StateAndLocalJurisdictionMember2023-12-310001305253us-gaap:DomesticCountryMembereigr:OrphanDrugCreditCarryforwardMember2023-12-310001305253eigr:SchoenVEigerBioPharmaceuticalsMember2022-11-08eigr:executive0001305253eigr:TheProgeriaResearchFoundationVEigerBioPharmaceuticalsMember2022-11-15eigr:claim0001305253us-gaap:SubsequentEventMemberus-gaap:DisposalGroupHeldforsaleNotDiscontinuedOperationsMembereigr:ZokinvyProgramMember2024-03-310001305253us-gaap:SubsequentEventMemberus-gaap:DisposalGroupHeldforsaleNotDiscontinuedOperationsMembereigr:ZokinvyProgramMember2024-04-0300013052532023-10-012023-12-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
_________________________________________________________________
Form 10-K
_________________________________________________________________
(Mark One)
| | | | | |
| ☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2023
or
| | | | | |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number 001-36183
_________________________________________________________________
Eiger BioPharmaceuticals, Inc.
(Exact name of registrant as specified in its charter)
_________________________________________________________________
| | | | | |
| Delaware | 33-0971591 |
(State or other jurisdiction of
incorporation or organization) | (I.R.S. Employer
Identification No.) |
| |
2155 Park Boulevard, Palo Alto, CA | 94306 |
| (Address of principal executive offices) | (Zip Code) |
(650) 272 6138
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| Common Stock, par value $0.001 per share | EIGR | The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act: None
_________________________________________________________________
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act:
| | | | | | | | | | | | | | |
| Large accelerated filer | ☐ | | Accelerated filer | ☐ |
| Non-accelerated filer | ☒ | | Smaller reporting company | ☒ |
| Emerging growth company | ☐ | | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ¨
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☒
The aggregate market value of voting and non-voting common stock held by non-affiliates of the registrant as of June 30, 2023 totaled approximately $30.0 million based on the closing price of $21.12 as reported by the Nasdaq Global Market (adjusted to reflect the one-for-thirty reverse stock split effected on January 5, 2024). This calculation excludes 57,119 shares held by executive officers, directors and stockholders that the Registrant has concluded are affiliates of the Registrant. Exclusion of such shares should not be construed to indicate that any such person possesses the power, direct or indirect, to direct or cause the direction of the management or policies of the registrant or that such person is controlled by or under common control with the Registrant.
The number of outstanding shares of the registrant’s common stock, par value $0.001 per share, as of April 4, 2024 was 1,480,797.
DOCUMENTS INCORPORATED BY REFERENCE
Part III incorporates certain information by reference from the registrant’s proxy statement for the 2024 Annual Meeting of Shareholders or an amendment to this Form 10-K to be filed no later than 120 days after the close of the registrant’s fiscal year ended December 31, 2023.
Our independent registered public accounting firm is KPMG LLP, San Francisco, CA, Auditor ID: 185
EIGER BIOPHARMACEUTICALS, INC.
TABLE OF CONTENTS
| | | | | | | | | | | |
| |
| | | |
| | | |
| | | |
| ITEM 1C. | | |
| | | |
| | | |
| | | |
| | | |
| |
| | | |
| | | |
| | | |
| | | |
| | | |
| ITEM 9. | | |
| ITEM 9A. | | |
| ITEM 9B. | | |
| ITEM 9C. | | |
| | | |
PART III | |
| ITEM 10. | | |
| ITEM 11. | | |
| ITEM 12. | | |
| ITEM 13. | | |
| ITEM 14. | | |
| | | |
PART IV | |
| ITEM 15. | | |
| ITEM 16. | | |
PART I
Forward-Looking Statements
This Annual Report on Form 10-K, including the sections entitled “Business,” “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” may contain “forward-looking statements.” We may, in some cases, use words such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “will,” “would” or the negative of those terms, and similar expressions that convey uncertainty of future events or outcomes, to identify these forward-looking statements. Any statements contained herein that are not statements of historical facts may be deemed to be forward-looking statements. Forward-looking statements in this Annual Report include, but are not limited to, statements about:
•the status, progress or outcome of the Company’s proceedings under Chapter 11 of the United States Bankruptcy Code, and/or any other strategic alternatives or contingency planning initiatives;
•our ability to maintain supply of our commercial and clinical trial materials;
•our ability to scale our commercial operations;
•our ability to finance the continued advancement of our portfolio products;
•our plans to research, develop and commercialize our product candidates;
•our ability to attract collaborators with development, regulatory and commercialization expertise;
•the size and growth potential of the markets for our product candidates, and our ability to serve those markets;
•our ability to obtain favorable reimbursement and pricing and the rate and degree of market acceptance of our product candidates;
•our ability to manufacture product necessary to support regulatory approvals and timely meet commercial requirements;
•regulatory developments in the U.S. and foreign countries;
•the performance of our third-party suppliers and manufacturers;
•the success of competing therapies that are or may become available;
•our ability to attract and retain key scientific or management personnel;
•the accuracy of our estimates regarding expenses, future revenues, capital requirements and needs for additional financing; and
•our expectations regarding our ability to obtain and maintain intellectual property protection for our product candidates.
These forward-looking statements reflect our management’s beliefs and views with respect to future events and are based on estimates and assumptions as of the filing date of this Annual Report and are subject to risks and uncertainties. We discuss many of these risks in greater detail under “Risk Factors.” Moreover, we operate in a very competitive and rapidly changing environment. New risks emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. Given these uncertainties, you should not place undue reliance on these forward-looking statements. Except as required by law, we undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise.
ITEM 1. Business
Overview
We are a commercial-stage biopharmaceutical company focused on the development of innovative therapies for hepatitis delta virus (HDV) and other serious diseases. All of our rare disease programs in our portfolio have FDA Breakthrough Therapy designation.
Our HDV platform includes lonafarnib, a first-in-class, oral farnesylation inhibitor that targets a critical host process involved in viral replication.
D-LIVR is the pivotal Phase 3 study of lonafarnib boosted with ritonavir, alone or in combination with peginterferon alfa-2a, for HDV. The study completed enrollment of 407 patients, and we announced Week 48 data in December 2022. Updated Week 72 data were presented at the EASL meeting in June 2023.
A pre-NDA meeting with FDA to discuss the results of the DLIV-R study was held in May 2023. The outcome of the meeting provides a pathway for submission of an NDA and the potential for accelerated approval of lonafarnib-based regimens as a finite 48-week treatment for chronic HDV infection.
The LIMT-2 Phase 3 study of peginterferon lambda (lambda) for HDV was stopped in September 2023 based on the recommendation of the Data Safety Monitoring Board (DSMB) for the study following its quarterly safety review due to an emergent safety signal after 144 patients had been enrolled.
Our portfolio also includes avexitide, a first in class, well-characterized GLP-1 antagonist, as a targeted treatment for two metabolic diseases with high unmet medical needs and no approved therapies: congenital hyperinsulinism (HI) and post-bariatric hypoglycemia (PBH). Avexitide has completed Phase 2 and is Phase 3 ready for both indications. The FDA approved our first commercial product, Zokinvy (lonafarnib), to reduce risk of mortality of Hutchinson-Gilford progeria syndrome (HGPS) and for treatment of processing-deficient progeroid laminopathies (PL) with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, in November 2020. Collectively known as progeria, these are ultra-rare and rapidly fatal genetic conditions of accelerated aging in children. In July 2022, we announced that the European Commission (EC) granted Eiger a centralized marketing authorization (MA). The EC's centralized MA is valid in all 27 EU member states plus Iceland, Liechtenstein, and Norway. In August 2022, the Medicine and Healthcare products Regulatory Agency (MHRA) granted approval in the UK. In January 2024, the Pharmaceutical and Medical Devices Agency (PMDA) granted approval in Japan. We have obtained a reimbursed price in Germany and have applied for a named patient program approvals in Spain and the UK. In January 2024, Eiger and its partner AnGes, Inc. received marketing approval in Japan from the Ministry of Health, Labour and Welfare for Zokinvy for the treatment of HGPS and processing-deficient progeroid laminopathies.
We commercially launched Zokinvy in the U.S. in January 2021 and started to record product revenue in the first quarter of 2021. Our first European sales were recognized in the fourth quarter of 2022. In May 2022 we entered into a Marketing and Distribution Agreement with AnGes, a company in Japan. AnGes is the marketing authorization holder in Japan and responsible for all regulatory interactions with PDMA. The partnership structure requires AnGes to pay Eiger $1.5 million upon achieving three pre-commercialization milestones.
In June 2023, in connection with an extensive portfolio prioritization review, we announced that the Company is focusing its clinical development efforts on advancing avexitide in hyperinsulinemic hypoglycemia (HH) indications. We will continue to commercialize Zokinvy for the treatment of Hutchinson-Gilford progeria syndrome (HGPS) and processing-deficient progeroid laminopathies (PL). In addition, we are evaluating strategic partnering options for our virology assets, lonafarnib and peginterferon lambda. During 2023, we have also undertaken certain cost savings initiatives, including a 55% reduction in workforce to date and efforts to reduce spending related to our hepatitis delta (HDV) development program. In June 2023, we also announced that we had appointed David Apelian, MD, PhD, MBA, who had served as interim CEO since December 2022, as our next CEO.
Voluntary Filing Under Chapter 11
As previously reported in our Current Report on Form 8-K filed with the U.S. Securities and Exchange Commission on April 1, 2024, the Company and its wholly owned subsidiaries commenced voluntary proceedings under Chapter 11 of the United States Bankruptcy Code (Chapter 11) in the United States Bankruptcy Court for the Northern District of Texas (Bankruptcy Court). The Chapter 11 proceedings are jointly administered under the caption In re Eiger BioPharmaceuticals, Inc., et al, Case No. 24-80040 (Chapter 11 Cases). The Company continues to operate its business in accordance with the applicable provisions of the Bankruptcy Code and orders of the Bankruptcy Court.
On March 31, 2024, we entered into a “stalking horse” asset purchase agreement (Asset Purchase Agreement) with Sentynl Therapeutics, Inc. (Purchaser), pursuant to which the Purchaser has agreed to acquire substantially all of the rights of the Company to its Zokinvy® program, including the Company’s in-license from Merck Sharp & Dohme Corp. (successor-in-interest of Schering Corporation) (Transferred Assets). The acquisition of the Transferred Assets by the Purchaser pursuant to the Asset Purchase Agreement is subject to approval of the Bankruptcy Court and one or more auctions, if necessary, to
solicit higher or otherwise better bids. On April 1, 2024, we filed a motion (Bidding Procedures Motion) seeking approval of, among other things, certain bidding procedures (Bidding Procedures), which will establish procedures for the selection of the highest or otherwise best bids for the sale of the Transfer Assets and other assets. Other interested bidders would be permitted to participate in the auction if they submit qualifying bids that are higher or otherwise better than the Asset Purchase Agreement. The Asset Purchase Agreement acts as a baseline for competitive bids for the acquisition of the Transferred Assets. The Bidding Procedures Motion additionally seeks the Bankruptcy Court’s approval of the Asset Purchase Agreement and designation of the Purchaser as the “stalking horse” bidder for the Transferred Assets.
Under the Asset Purchase Agreement, the Purchaser has agreed, subject to the Bankruptcy Court’s approval and absent any higher or otherwise better bid, to acquire the Transferred Assets from the Debtors for $26 million, subject to certain adjustments, including per diem reductions if the sale closes after April 24, 2024, in accordance with the terms and conditions of the Asset Purchase Agreement, plus the assumption of specified liabilities related to the Transferred Assets. The Debtors in these Chapter 11 Cases, are Eiger BioPharmaceuticals, Inc. and our direct subsidiaries. The Asset Purchase Agreement includes customary representations and warranties and various customary covenants under the circumstances that are subject to certain limitations, including, without limitation, a termination fee, expense reimbursement and the right to designate executory contracts and unexpired leases to assume or reject. On April 3, 2024, the Purchaser increased the purchase price to $30.0 million subject to certain adjustments, including per diem reductions if the sale closes after April 24, 2024.
Subject to certain specific exceptions under the Bankruptcy Code, the Chapter 11 Cases automatically stayed most judicial or administrative actions against the Company and its direct subsidiaries and efforts by creditors to collect on or otherwise exercise rights or remedies with respect to obligations of the Company and its direct subsidiaries incurred prior to the petition date.
We have historically incurred operating losses in each year since inception and we expect to incur losses for the foreseeable future. We had a net loss of $75.0 million and $96.8 million for the years ended December 31, 2023 and 2022, respectively. As of December 31, 2023, we had $25.4 million of cash and cash equivalents, and an accumulated deficit of $512.2 million. Substantially all of our operating losses have resulted from expenses incurred in connection with our research and development programs and from selling, general and administrative costs associated with our operations.
We expect to incur significant expenses and increasing operating losses for the foreseeable future. We expect that our operating losses will fluctuate significantly from quarter to quarter and year to year due to timing of clinical development programs and efforts to achieve additional regulatory approvals. In June 2023, we announced that we had conducted a formal program prioritization assessment with a focus on enhancing long-term shareholder value while fulfilling the promise of advancing our high-potential product candidates for patients with serious diseases.
Based on our recurring losses from operations incurred since inception, expectation of continuing operating losses and negative cash flows for the foreseeable future, and need to raise additional capital to finance our future operations, management has concluded that there is substantial doubt regarding Eiger's ability to continue as a going concern beyond twelve months after the date that our consolidated financial statements are issued, which was a principal reason for our decision to seek bankruptcy protection under Chapter 11.
Clinical Product Candidates
1.Lonafarnib (LNF) for HDV
LNF is a well-characterized, orally bioavailable, first-in-class farnesylation inhibitor that has completed a Phase 3 clinical trial for HDV infection. HDV is the most severe form of viral hepatitis for which there is currently no FDA-approved therapy. Chronic HDV infection can lead to a rapid progression to liver cirrhosis, a greater likelihood of developing liver cancer, and has the highest fatality rate of all the chronic hepatitis infections.

We licensed LNF from Merck in 2010. LNF is a small molecule that blocks the production of HDV virus particles by inhibiting a key step, called farnesylation, in the virus life cycle. We have completed five Phase 2 dose-finding studies in 129 HDV-infected patients. LNF has demonstrated dose-dependent activity in reducing HDV viral load both as a monotherapy and in combination with ritonavir (RTV) and/or PEG IFN-alfa-2a. Phase 2 studies have identified two lonafarnib-based regimens, which are in our Phase 3 registration program. Both regimens have achieved clinically meaningful composite endpoints of HDV RNA decline ≥ 2 logs from baseline and normalized alanine aminotransferase (ALT), a key liver enzyme, at Week 24: all-oral regimen of LNF 50 mg boosted with RTV twice daily and combination regimen of LNF boosted with RTV combined with PEG IFN-alfa-2a. Predominantly grade 1 gastrointestinal (GI) adverse events (AE) were observed in Phase 2 amongst per-protocol treated patients.
Our Phase 3 registration program consists of a completed single, pivotal, international trial, called D-LIVR, that is designed to support U.S. and EU regulatory approvals. D-LIVR has generated data for two distinct lonafarnib-based ritonavir-boosted regimens for approval. The study included an all-oral arm of lonafarnib boosted with ritonavir and a combination arm of lonafarnib boosted with ritonavir combined with peginterferon-alfa-2a. Each arm was compared to placebo. The primary efficacy endpoint was met: both regimens demonstrated statically significant superiority compared with placebo. Off-treatment data from 24-weeks follow-up demonstrated incrementally greater treatment responses than those observed at the end-of-treatment (Week-48).
LNF for treatment of HDV infection has been granted Orphan Drug designation by the FDA and EMA, Fast Track and Breakthrough Therapy designations by the FDA and PRIME designation by the EMA.
A pre-NDA meeting with FDA was held in May 2023, the outcome of which provides a pathway for submission of an NDA and the potential for accelerated approval of lonafarnib-based regimens as a finite 48-week treatment for chronic HDV infection. FDA's accelerated approval program enables expedited approval of drugs that treat serious conditions and fill an unmet medical need. Approval becomes possible when a surrogate endpoint suggestive of a longer-term clinical benefit is realized (i.e., reduced viral load, improved liver function). A requirement of accelerated approval is the sponsor's implementation of a long-term follow-up study intended to confirm clinical benefit (i.e., reduced mortality, reduction in liver cancer). As a condition of accelerated approval Eiger will need to agree with FDA on a long-term follow-up plan. The study or studies will need to be actively recruiting prior to accelerated approval being granted to LNF for HDV.
2.Peginterferon Lambda (lambda) for HDV
Lambda was in clinical development for HDV. Lambda is a well-characterized, first-in-class, type III, well-tolerated interferon (IFN), that stimulates immune responses that are critical for the development of host protection during viral infections.
We licensed worldwide rights to lambda from Bristol Myers Squibb in April 2016. Lambda has been administered in clinical trials involving over 4,000 patients infected with the Hepatitis B Virus (HBV), Hepatitis C Virus (HCV), Hepatitis D Virus (HDV), and SARS-CoV-2. Lambda has not been approved for any indication.
In September 2023, the Phase 3 LIMT-2 study in patients with chronic HDV infection was prematurely stopped upon the recommendation of the DSMB for the trial due to an emergent safety signal. The FDA issued a full Clinical Hold letter. We are currently in the process of monitoring patients for safety after cessation of treatment.
3.Combination Therapy for HDV
We have also generated data with the combination of LNF + RTV and lambda for the treatment of HDV. The Phase 2 LIFT-1 study was a single arm investigator sponsored study of lambda combined with LNF boosted with RTV for 24 weeks treatment with 24 weeks follow-up. Primary endpoint was > 2 log decline in HDV RNA at end of treatment. In November 2020, we reported positive end-of-study data from a Phase 2 single arm study called LIFT-1, conducted at the National Institutes of Health’s National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) in 26 HDV-infected patients.
4.Peginterferon Lambda (lambda) for COVID-19
We have also generated data with lambda in multiple Phase 2 and Phase 3 investigator sponsored studies of mild and moderate COVID-19 patients.
The TOGETHER study was an investigator sponsored Phase 3, multi-center, randomized, placebo-controlled adaptive platform study evaluating therapeutics in newly diagnosed, high-risk, non-hospitalized patients with COVID-19 across twelve clinical trial sites in Brazil and 5 sites in Canada.
The primary endpoint compared the number of emergency setting visits, hospitalizations, and/or deaths in treated patients versus placebo through Day 28. TOGETHER completed enrollment of over 1,900 predominantly vaccinated patients at high risk for developing complications from progression of COVID-19 randomized 1:1 lambda vs. placebo. Following our press release on September 6, 2022, in which we updated on the status of our planned EUA based on recent communications with the FDA, we submitted a pre-EUA meeting request to the FDA as well as additional morbidity and mortality outcomes data and analyses from the investigator-sponsored TOGETHER study. This included further statistical modeling and efficacy analyses of the study's primary and secondary endpoints and long-term follow-up data that we believe continues to support the topline outcomes reported in March. In response, the FDA denied the request for a pre-EUA meeting. Citing its concerns with the conduct of the TOGETHER study, the FDA concluded that any authorization request based on these data is unlikely to meet the statutory criteria for issuance of an EUA in the current context of the pandemic.
The FDA suggested that, given lambda's mechanism of action and the ongoing need for improved COVID-19 therapeutics, we should consider requesting an end-of-Phase 2 meeting to discuss a company-sponsored pivotal trial that could support an eventual Biologics License Application (BLA). We continue to explore opportunities for the use of lambda for COVID-19 and other respiratory viral infections.
The potential to for further development of Lambda as a treatment for COVID-19 and other respiratory viral infections will be considered after the final safety data from LIMT-2 have been submitted and reviewed by the FDA.
5.Avexitide for Congenital Hyperinsulinism (HI)
Avexitide is a well-characterized peptide that we are developing as a treatment for congenital hyperinsulinism (HI), an ultra-rare, pediatric metabolic disorder. HI is the most frequent cause of persistent hypoglycemia in neonates and children and is characterized by fasting hypoglycemia and results in permanent brain damage with neurodevelopmental deficits in up to 50% of patients. Near-total pancreatectomy is often indicated and leads to life-long insulin-dependent diabetes (IDDM). Safe and effective therapies are urgently needed to prevent brain damage, IDDM and death.
Avexitide binds to the GLP-1 receptor on pancreatic beta-cells and behaves as a GLP-1 antagonist and inverse agonist. Published studies demonstrate that GLP-1 receptor antagonism reduces fasting and amino-acid induced cAMP accumulation and thereby decreases calcium-stimulated insulin secretion. Avexitide has demonstrated clinical proof of concept and has been dosed in nearly 40 patients with HI at Children’s Hospital of Philadelphia (CHOP).
Avexitide has been granted Breakthrough Therapy designation by the FDA for the treatment of HI, Orphan Drug designation by the EMA for the treatment of HI and Orphan Drug designation by the FDA for the treatment of hyperinsulinemic hypoglycemia, which includes HI. Avexitide has also been granted Rare Pediatric Disease designation by the FDA.
6.Avexitide for Post-Bariatric Hypoglycemia (PBH)
Our portfolio also includes avexitide as a treatment for post-bariatric hypoglycemia (PBH), a debilitating and potentially life-threatening condition for which there is no approved therapy. This disorder occurs in a subset of patients after bariatric and other upper gastrointestinal (GI) surgeries, including Roux-en-Y gastric bypass (RYGB), Sleeve Gastrectomy (SG), total gastrectomy (TG), esophagectomy and Nissen fundoplication. Affected patients experience frequent symptomatic hypoglycemia, with blood glucose concentrations often low enough to cause seizures, altered mental status, loss of consciousness and even death. Bariatric procedures are usually performed for management of medically complicated obesity, TG and esophagectomy for gastric and esophageal cancers, respectively, and Nissen fundoplication for gastroesophageal reflux disease (GERD).
Five clinical studies have been completed demonstrating clinical proof of concept in over 70 patients suffering from PBH. Avexitide is a glucagon-like peptide-1 (GLP-1) receptor antagonist that competes with endogenous GLP-1 and has been shown in Phase 2 clinical trials to reduce the excessive post-prandial insulin release that characterizes this disorder. These Phase 2 data were generated using intravenous or subcutaneous (SC) formulation delivery. Pharmacokinetics from these Phase 2 SC studies indicate that the SC formulation could enable once or twice a day dosing. We developed a proprietary SC liquid formulation and completed a Phase 1 dose-ranging pharmacokinetics trial in healthy humans. In October 2018, we reported positive topline data from PREVENT, a multi-center, placebo-controlled study investigating the safety and durability of effect of 28-day dosing of SC avexitide in post-bariatric surgical patients. The primary efficacy endpoint of improved postprandial glucose nadir during mixed meal tolerance testing (MMTT) was achieved with statistical significance with fewer participants requiring glycemic rescue during each of the active dosing regimens than during placebo dosing. Significant reductions in hypoglycemia were also observed in the home setting, with fewer Level 1-3 hypoglycemia events and less time spent with glucose <54 mg/dL as measured by patient electronic diary (eDiary), self-monitored blood glucose (SMBG), and blinded continuous glucose monitor (CGM). In June 2022, we reported positive results from the Phase 2b study, supporting the broader potential of avexitide in patients with PBH and hyperinsulinemic hypoglycemia related to GI surgeries. In this open-label, investigator-initiated, cross-over study, the primary and secondary endpoints, which measured frequency of hypoglycemia and amount of time in hypoglycemia, were met with statistical significance. Avexitide treatment in addition to medical nutrition therapy (MNT) demonstrated significant reductions in the rates of Levels 1-3 hypoglycemia as measured by eDiary and SMBG compared to MNT alone. Objective assessment by blinded CGM also demonstrated significant reductions in the rates of hypoglycemia and showed significant reductions in percent time in hypoglycemia.
Avexitide for the treatment of hyperinsulinemic hypoglycemia has been granted Orphan Drug designation by the FDA and for the treatment of non-insulinoma pancreatogenous hypoglycemia syndrome (NIPHS) by the EMA. NIPHS describes a spectrum of acquired metabolic disorders characterized by inappropriately high insulin levels (hyperinsulinemia) and low blood glucose levels (hypoglycemia), which includes PBH. Avexitide for the treatment of PBH has also been granted Breakthrough Therapy designation by the FDA. Following Scientific Advice meetings with regulatory agencies, we have agreement on a single pivotal Phase 3 study.
Approved Product
1.Zokinvy (lonafarnib) for Hutchinson-Gilford progeria syndrome (HGPS) and Processing-Deficient Progeroid Laminopathies (PL)
In November 2020, we received FDA approval for Zokinvy to reduce the risk of mortality in HGPS and to treat processing-deficient PL. Collectively known as progeria, these are ultra-rare and rapidly fatal genetic conditions of accelerated aging in children. Zokinvy is our first approved product, and the first approved therapy for these indications. There are approximately 20 identified patients in the U.S. who are eligible for treatment with Zokinvy.
On July 20, 2022, we announced that the European Commission (EC) granted Eiger a centralized marketing authorization (MA) under the exceptional circumstances procedure for Zokinvy for the treatment of HGPS and PL, ultra-rare and rapidly fatal genetic conditions of accelerated aging in children. In July 2022, we announced that the European Commission (EC) granted Eiger a centralized marketing authorization (MA). The EC's centralized MA is valid in all 27 EU member states plus Iceland, Liechtenstein, and Norway. In August 2022, the Medicine and Healthcare products Regulatory Agency (MHRA) granted approval in the UK.
Zokinvy is a disease-modifying agent that has demonstrated a statistically significant survival benefit in children and young adults with HGPS. At last follow-up time (i.e., August 2021) the mean lifespan of patients with HGPS and treated with Zokinvy increased by 4.3 years. The most commonly reported adverse reactions were gastrointestinal (vomiting, diarrhea,
nausea), and most were mild or moderate (Grade 1 or 2) in severity. Many patients with HGPS have received continuous Zokinvy therapy for more than 10 years.
HGPS is an ultra-rare and rapidly fatal genetic condition of accelerated aging in children. HGPS is caused by a point mutation in the LMNA gene, encoding the lamin A protein, yielding the farnesylated aberrant protein called progerin. Lamin A protein is part of the structural scaffolding that holds the nucleus together. Researchers now believe that progerin may make the nucleus unstable, and that cellular instability may lead to the process of premature aging in HGPS. Children with HGPS die of the same heart disease that affects millions of normally aging adults, arteriosclerosis, but at an average age of 14.5 years if untreated. Disease manifestations include severe failure to thrive, scleroderma-like skin, global lipodystrophy, alopecia, joint contractures, skeletal dysplasia, global accelerated atherosclerosis with cardiovascular decline, and debilitating strokes.
Processing-deficient progeroid laminopathies are genetic conditions of accelerated aging caused by a constellation of mutations in the LMNA and/or ZMPSTE24 genes yielding farnesylated proteins that are distinct from progerin. While non-progerin producing, these genetic mutations result in disease manifestations with phenotypes that have overlap with, but are distinct from, HGPS. Collectively, worldwide prevalence of progeroid laminopathies is believed to be approximately 200 patients.
In November 2020, we entered into an amendment to our license agreement with Merck to include not only all uses of LNF related to the treatment of HGPS, but also progeroid laminopathies.
In January 2024, Eiger and its partner AnGes, Inc. received marketing approval in Japan from the Ministry of Health, Labour and Welfare for Zokinvy for the treatment of HGPS and processing-deficient progeria laminopathies.
Business Model and Management Team
We have a highly experienced management team whose members have, in the course of their prior employment, participated in bringing numerous product candidates through regulatory approval and into commercialization. We plan to leverage our management team’s breadth and depth of experience in clinical and regulatory product development as well as market development and commercialization to identify potentially promising product candidates to address unmet medical needs.
Our current product candidate portfolio has been obtained by in-licensing from pharmaceutical companies and academic institutions. With our focus on HDV and other serious diseases, our strategy is to acquire and retain some or all commercialization rights to our products in significant territories to diversify risk, identify a rapid regulatory pathway to approval and minimize the development investment in order to maximize long-term value for our stockholders. Over time, depending upon the data and potential market opportunity, we expect to develop an integrated commercial organization, which we believe can be targeted and cost effective for selected, promising orphan disease designated programs. We plan to balance these interests with opportunities to out-license assets from our portfolio enhance stockholder value through partnerships and other strategic relationships.
Members of our executive management team have worked in other private and public biotechnology companies such as Alza Corporation and Schering-Plough, each of which was acquired by a larger biopharmaceutical industry company. Our executive management team also has previous work experience at biopharmaceutical companies, including Achillion, GlobeImmune, Bristol-Myers Squibb, Gilead, AbbVie, and Novo Nordisk in the field of hepatitis, as well as in the development and commercialization of novel therapeutics for other serious diseases and rare diseases.
Hepatitis Delta Virus Overview
About Hepatitis Delta Virus
HDV is a small circular ribonucleic acid (RNA) virus that expresses only one protein, the hepatitis delta antigen (HDAg). There are two forms of HDAg – small and large. These two forms of HDAg and the single-stranded RNA genome are surrounded by a lipid envelope, which is embedded with Hepatitis B Virus (HBV) surface antigen (HBsAg) proteins. HDV does not encode its own envelope proteins and must acquire them from HBV during the final steps of replication. Hence, natural HDV infections always occur in the presence of a co-existing HBV infection. HBsAg is the only element of HBV relied upon by HDV. HDV replication can occur independently of HBV replication.
HDV is the most severe form of viral hepatitis. HDV can be acquired either by co-infection (a simultaneous co-infection
with HDV and HBV) or by super-infection (HDV infection of someone already harboring a chronic HBV infection). Both co-infection and super-infection with HDV result in more severe complications compared to infection with HBV alone. These complications include a greater likelihood of experiencing liver failure in acute infections and a rapid progression to liver cirrhosis, with an increased chance of developing liver cancer in chronic infections. HDV has the highest fatality rate of all the hepatitis infections at up to 20%. Although HDV/HBV simultaneous co-infection in adults usually resolves completely, in some cases it can become fulminant hepatitis, which carries a very high mortality rate. In the case of super-infections, the predominant form of HDV, HDV super-infection leads to a more severe form of disease than chronic HBV mono-infection. In a study published in 1987 in the Journal of Infectious Diseases, histological liver deterioration was observed in 77% of HBV patients co-infected with HDV over a 15-year follow-up period, versus 30% of patients infected with HBV alone (p<0.01). In a 2013 study of chronic HBV patients published in the Journal of Gastroenterology and Hepatology, cirrhosis was present in 73% of HBV patients co-infected with HDV, compared to only 22% of those infected with HBV alone. Patients co-infected with HDV are more than twice-as-likely to develop liver-related complications, cirrhosis, or require liver transplants than matched patients infected with HBV alone.


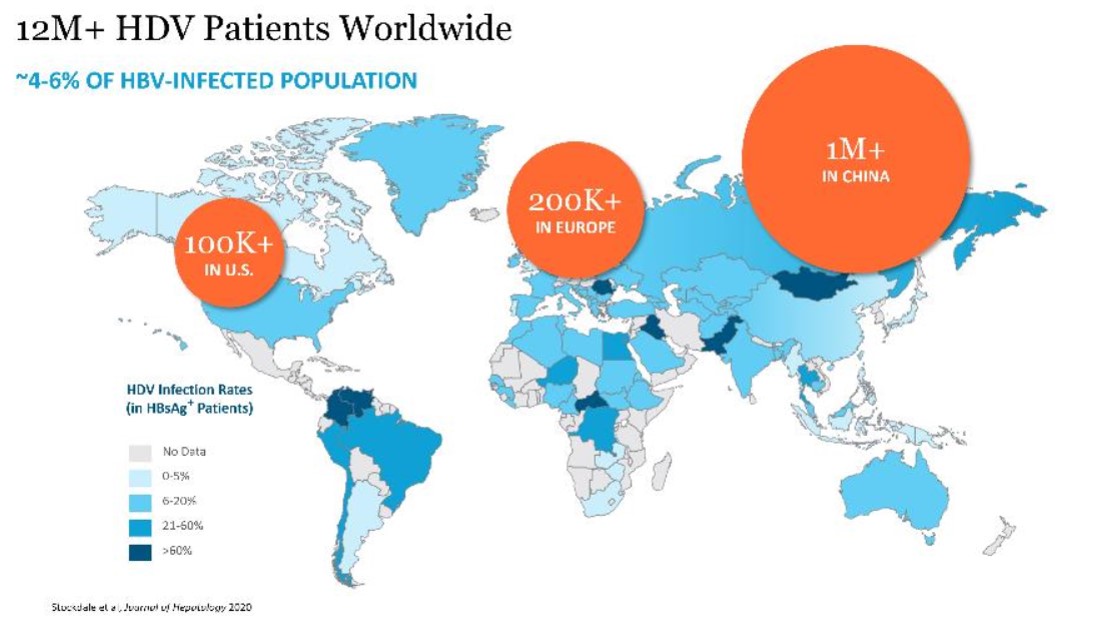
HDV is generally spread through exchange of body fluids either sexually or through contact with infected blood. Globally, it is estimated that between 4.3% and 5.7% of the 240 million worldwide chronic HBV population, or 10 to 14 million people, are infected with HDV. The prevalence of HDV in patients infected with chronic HBV is even higher in certain regions, including certain parts of Mongolia, China, Russia, Central Asia, Pakistan, Turkey, Africa and South America, with an HDV prevalence as high as 60% being reported in HBV-infected patients in Mongolia and Pakistan. The
prevalence of HDV has recently begun to increase in Western Europe and the United States due to migration from countries with high infection rates.
The Role of HDV Screening in Identifying Patients Who May Benefit from LNF and/or Lambda
Active HDV infections are best detected by reverse transcriptase-polymerase chain reaction (RT-PCR) assays for genomic RNA. These assays yield a quantitative assessment of the number of viral particles, or viral load, in serum. A commercial assay for quantitative HDV RNA has been available in Europe (Robogene®) since 2015. Quest Diagnostics and ARUP Laboratories offer commercial assays for quantitative HDV RNA testing in the United States. Both assays are calibrated using the World Health Organization HDV standard provided by the Paul Ehrlich Institute in Germany.
Our initial discussions with payors have indicated that they would be willing to reimburse healthcare providers for HDV RNA quantitative assays that are carried out following a positive HBsAg test for HBV. Greater availability of commercial assays will increase the number of assays performed and increase the number of identified patients who can potentially benefit from an HDV therapies such as LNF and lambda.
Current Therapy for HDV
Currently, there is no FDA-approved therapy for hepatitis delta virus infection. Hepcludex (buleviritide) was conditionally approved in Europe in 2020 for treatment of chronic HDV, and a BLA was submitted to the U.S. FDA in November 2021. In October 2022, the FDA issued a Complete Response Letter (CRL) citing concerns regarding the manufacture and delivery of bulevirtide.
The American Association for the Study of Liver Diseases (AASLD) guidelines suggest treatment of chronic hepatitis delta infections with IFN-alfa. In clinical trials of IFN-alfa or PEG IFN-alfa, between 25% and 33% of HDV infected patients were able to achieve undetectable HDV RNA after a minimum of 48 weeks of therapy, with some requiring two years of therapy. However, long-term therapy with IFN-alfa is known to be associated with numerous adverse events and tolerability is a significant problem for some of these patients. In addition, rebound of HDV RNA occurs in greater than 50% of patients.
HBV nucleoside analogs that suppress HBV DNA are ineffective against HDV since they are ineffective in suppressing the expression HBsAg. Current development programs targeting HBV functional cures are in early stages and are not expected to eliminate extra-hepatic reservoirs of HBsAg. Given that HDV only requires small amounts of HBsAg for virion assembly, functional cures, if achieved, will not eradicate HDV.
HDV Replication and Farnesylation
After HDV enters a target cell hepatocyte, the genome is translocated to the nucleus where genome replication occurs and the two forms of HDAg small delta antigen (SHDAg) and large delta antigen (LHDAg) are produced. The newly formed HDV genome and the small and large delta antigen must acquire a lipid envelope from HBV to complete the assembly process. An important interaction between HDV and HBV proteins has been shown to depend on the presence of the last
four amino acids of the large delta antigen, comprising a CXXX box motif, where C represents cysteine and X denotes any other amino acid. This amino acid sequence is required for LHDAg to be farnesylated by a host enzyme which covalently attaches a 15-carbon prenyl lipid (farnesyl-moiety) to the cysteine of the CXXX box. Farnesylation of the large delta antigen renders it more lipophilic, promotes its association with HBsAg and is essential for initiating the HDV particle formation process. Our approach involves targeting this host process called farnesylation, or protein farnesylation, which has been shown to be essential for the last steps in HDV replication, the assembly and release of new virus progeny.
In the 1980s, farnesyltransferase inhibitors were developed by multiple pharmaceutical companies for oncology indications. Addition of a farnesyl or prenyl lipid group to the Ras protein (Ras) a well-known and important regulator of cellular proliferation, allows for membrane association. Once membrane bound, Ras may then be activated. The importance of activated Ras in tumor development was demonstrated by sequence analyses of tumors from patients where up to 30% have mutations involving Ras. Several farnesylation inhibitors were developed in oncology and taken into the clinic and in some cases through late-stage clinical development. However, these programs did not lead to approvals, due to a lack of compelling efficacy. The class-related, dose-limiting toxicity has been gastrointestinal side effects including nausea, vomiting, diarrhea and weight loss.
Published studies demonstrate that farnesyltransferase inhibitors block HDV viral production both in cellular experiments and in HDV transgenic mice. Targeting farnesylation or farnesyltransferase, a host target, significantly reduces the likelihood of HDV developing resistance to escape effects of antiviral therapy. Viruses mutate quickly and there is a higher rate of mutations in viral replication compared to mammalian cell division. However, no matter how much HDV may mutate, these changes are unlikely to alter the host process of farnesylation which HDV requires to complete packaging. Thus, targeting a host farnesylation process provides what we believe to be a higher barrier to resistance. Identification of clinic-ready farnesylation inhibitors has allowed us to move rapidly into proof-of-concept studies in humans.
Our First HDV Opportunity: Lonafarnib (LNF) for HDV
Lonafarnib (LNF) is a well-characterized, orally active inhibitor of farnesyltransferase. LNF inhibits the farnesylation step of HDV replication inside liver cells and blocks the ability of the virus to multiply. Since farnesylation is a host process, not under control of HDV, and LNF inhibits farnesylation, we believe that there is also a potentially higher barrier to resistance with LNF therapy. LNF for the treatment of HDV infection has been granted Orphan Drug designation in Europe and the United States, and LNF in combination with RTV has been granted Fast Track and Breakthrough Therapy designations from the FDA for the treatment of chronic HDV infections.
In the United States, we have issued patents, U.S. Patent No. 10,076,512; 10,828,283; and 11,311,519, all entitled Treatment of Hepatitis Delta Virus Infection. The issued claims cover a broad range of RTV-boosted LNF doses and durations with and without interferons. The European Patent Office, the Chinese Patent Office and the Japan Patent Office have also granted patents with claims covering a broad range of LNF boosted with RTV dosing regimens for the treatment of chronic HDV infection.
LNF Phase 2 Clinical Data
We licensed LNF from Merck in 2010, and have relied upon Merck’s prior Phase 1, 2 and 3 clinical experience with LNF in over 2,000 patients to understand safety and pharmacokinetics.
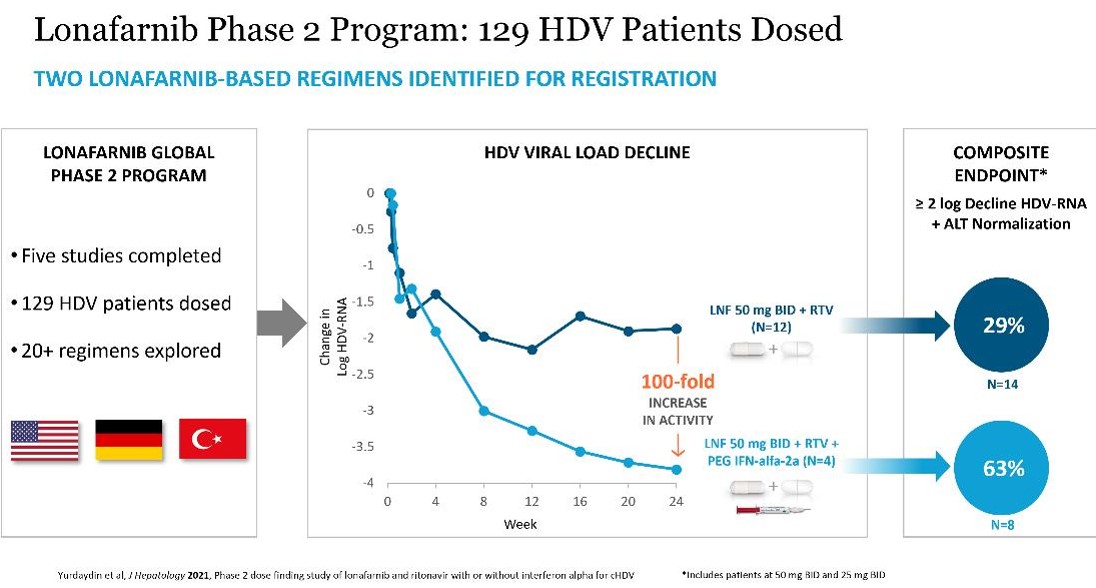
We have completed five Phase 2 trials (POC, LOWR-1, LOWR-2, LOWR-3, LOWR-4) with LNF in 129 HDV-infected patients. The Phase 2 LOWR HDV (Lonafarnib With Ritonavir in HDV) Program identified dose(s) and regimen(s) for registration.
•POC Study (Placebo-controlled LNF monotherapy) (N=14)
•LOWR-1 Study (Combination: LNF with RTV or PEG IFN-alfa-2a) (N=21)
•LOWR-2 Study (Dose Finding: LNF + RTV ± PEG IFN-alfa-2a) (N=58)
•LOWR-3 Study (QD Dosing: LNF + RTV) (N=21)
•LOWR-4 Study (Dose-Escalation: LNF + RTV) (N=15)
The Phase 2 NIH proof-of-concept study demonstrated statistically significant decreases in HDV RNA viral load with two LNF active groups versus placebo for 28-days. A statistically significant correlation between increasing LNF serum levels
and decreasing HDV RNA viral loads was also observed, demonstrating that higher serum levels resulted in greater decline in HDV RNA.
The Phase 2 LOWR studies demonstrated benefits of twice daily RTV-boosting of LNF for up to 24 weeks of dosing. RTV is a pharmacokinetic (PK) enhancer known to inhibit the metabolism of LNF, allowing lower doses of LNF to be administered, while resulting in higher systemic concentrations of LNF. The Phase 2 LOWR HDV studies identified two LNF-based regimens that can achieve clinically meaningful composite endpoints of HDV RNA decline ≥ 2 logs from baseline and normalized ALT at Week 24: all-oral regimen of LNF 50 mg BID boosted with RTV twice daily and combination regimen of LNF 50 or 25 mg BID boosted with RTV combined with PEG IFN-alfa-2a (see figures below). These dosing regimens were associated with predominantly grade 1 GI AEs amongst per-protocol treated patient.
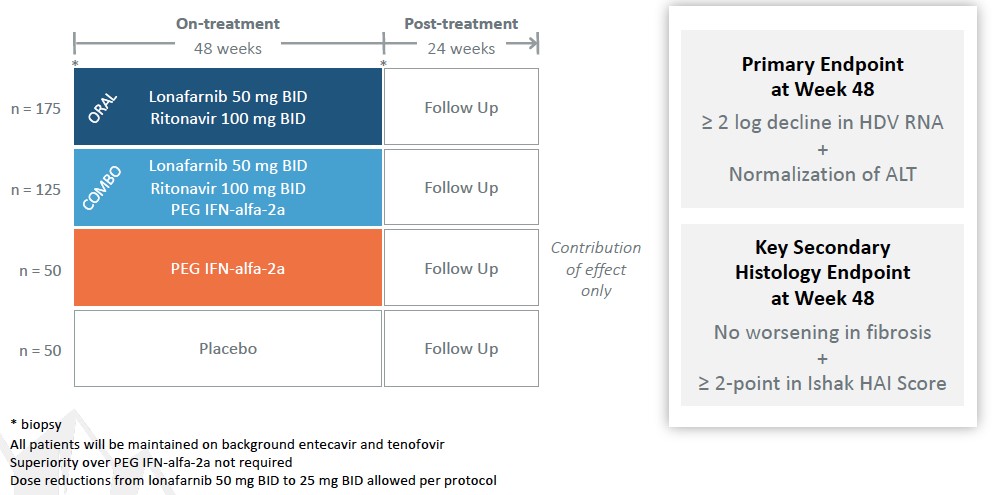
Phase 3 D-LIVR Study
D-LIVR (Delta-Liver Improvement and Virologic Response in HDV) is an international, multi-center, Phase 3 study of LNF-treated patients (total N=407 patients including controls) to evaluate an all-oral arm of LNF boosted with RTV and a combination arm of LNF boosted with RTV and peginterferon-alfa-2a (PEG IFN-alfa-2a), with each arm compared to a placebo arm (background HBV nucleos(t)ide only), in HDV-infected patients. A PEG IFN-alfa-2a alone arm is included to demonstrate contribution of effect only. The LNF containing arms are not required to demonstrate superiority over PEG IFN-alfa-2a alone. The primary endpoint is a composite of a ≥ 2 log decline in HDV RNA and ALT normalization at end of 48 weeks of treatment with LNF-based regimens versus placebo.
D-LIVR is an international study, with over 100 sites across over twenty countries. Week 48 topline data was reported on December 8, 2022, with Week 72 data presented at the EASL meeting in June 2023.
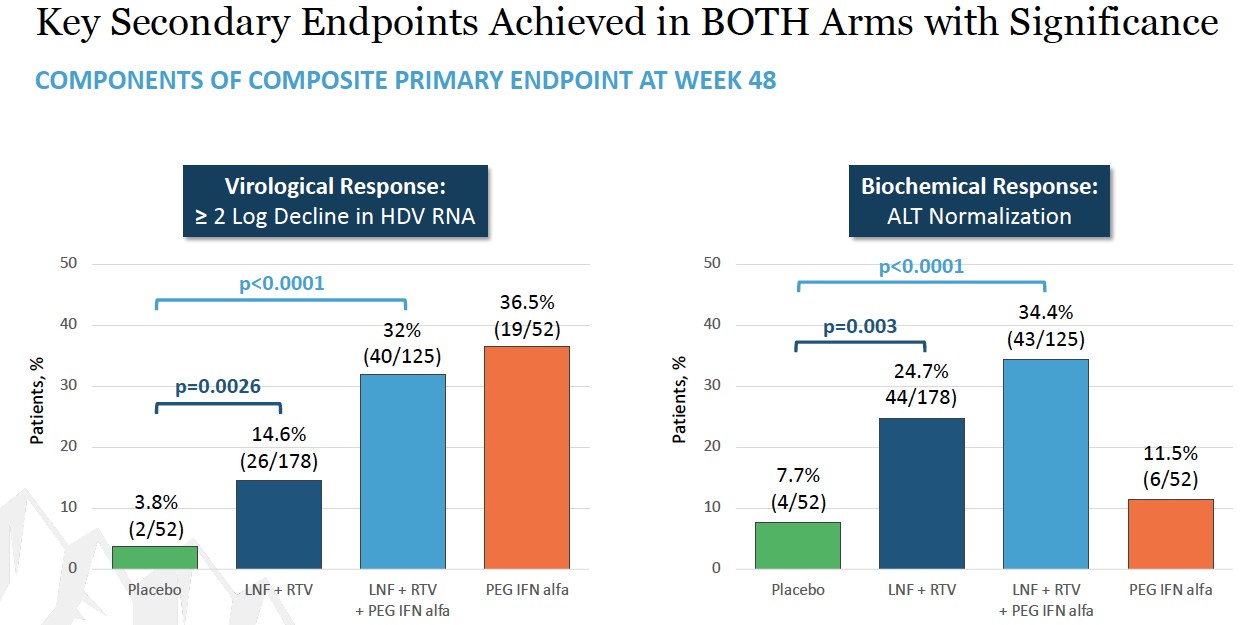
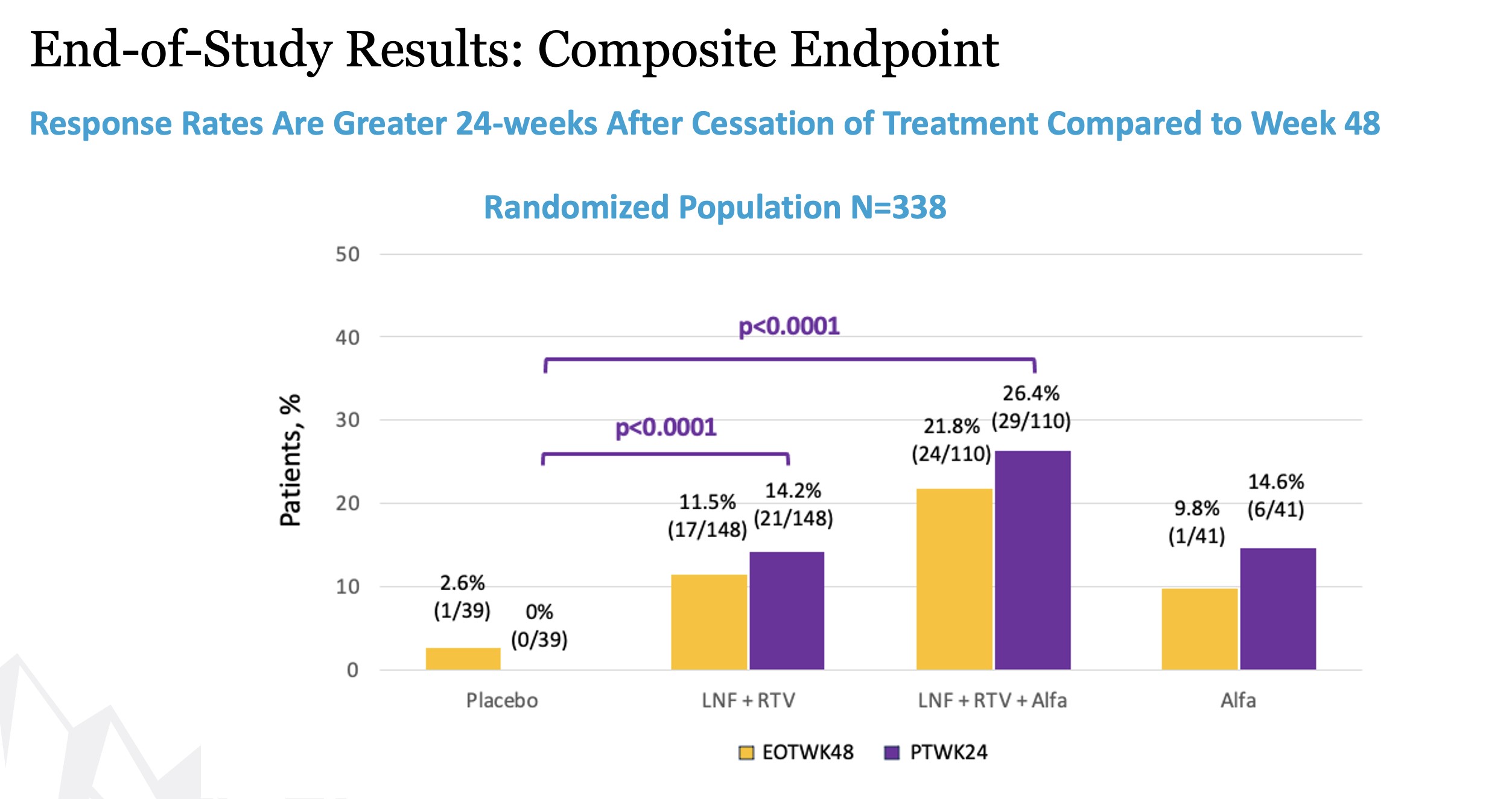
Week 48 results showed that both treatment arms achieved statistical significance over placebo in the composite primary endpoint as well as the component virologic and biochemical responses. Study participants receiving the all-oral therapy and combination therapy showed a composite response of 10.1% (p=0.0044) and 19.2% (p <0.0001), respectively, compared to those receiving placebo (1.9%). A peginterferon alfa comparator arm was included in the study to show contribution of effect. The composite response rate in the all-oral arm was comparable to the peginterferon alfa arm (10.1% vs 9.6%). The composite response rate in the combination arm was twice that of the peginterferon alfa arm (19.2% vs 9.6%).

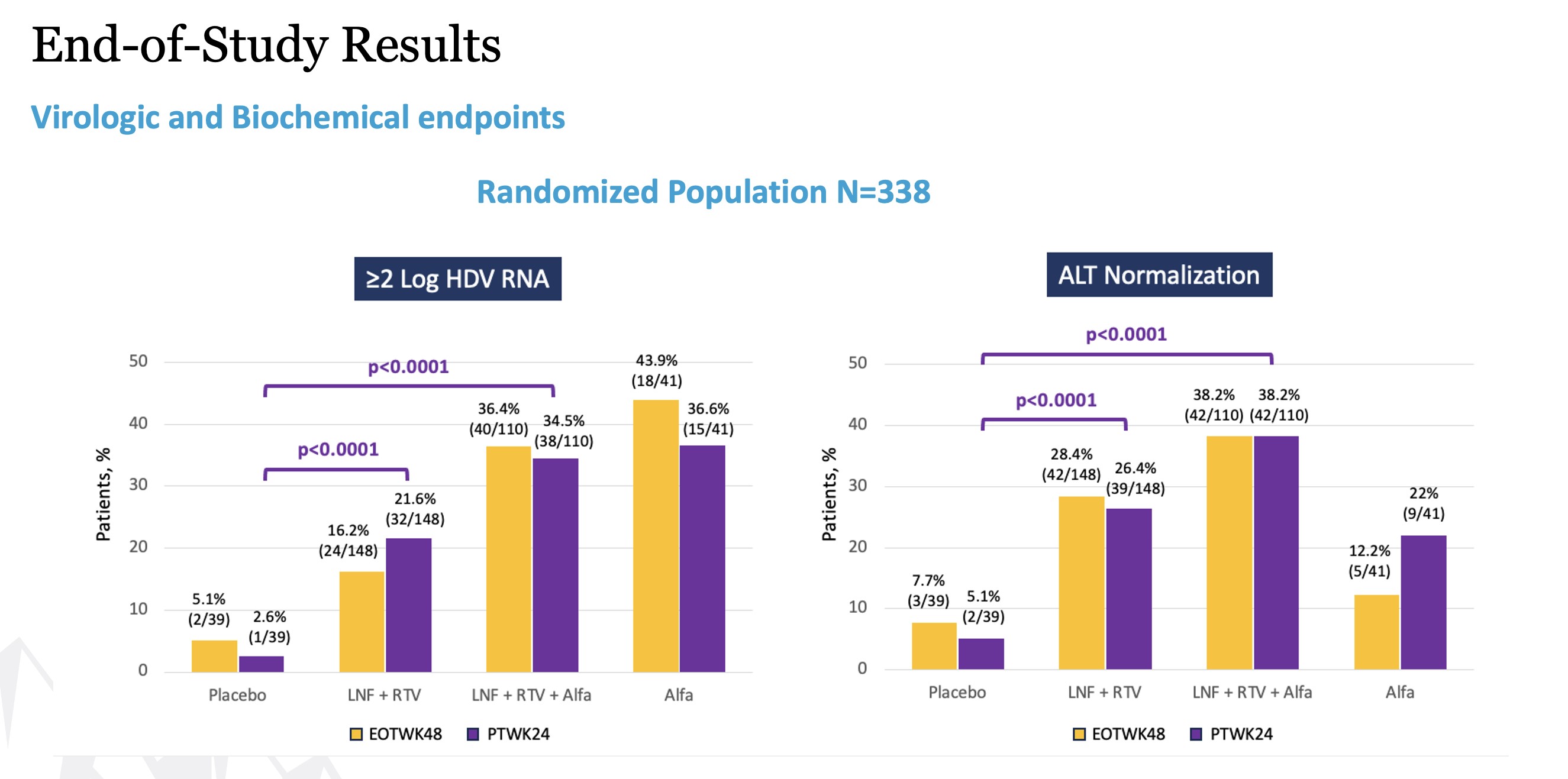
Breaking the composite primary endpoint into its two component parts also showed statistically significant improvements for both parameters. Looking first at virologic response, defined as a greater than or equal to 2 log reduction of HDV RNA, 14.6% of the study participants receiving the all-oral therapy showed improvement with a p-value of 0.0026, and 32.0% of the patients receiving combination therapy showed improvement with a p-value less than 0.0001, compared to 3.8% of those patients receiving placebo. For ALT normalization, 24.7% of the study participants receiving the all-oral therapy
showed improvement with a p-value of 0.003, and 34.4% of those patients receiving the combination therapy showed improvement with a p-value less than 0.0001, compared to 7.7% of those patients receiving placebo.
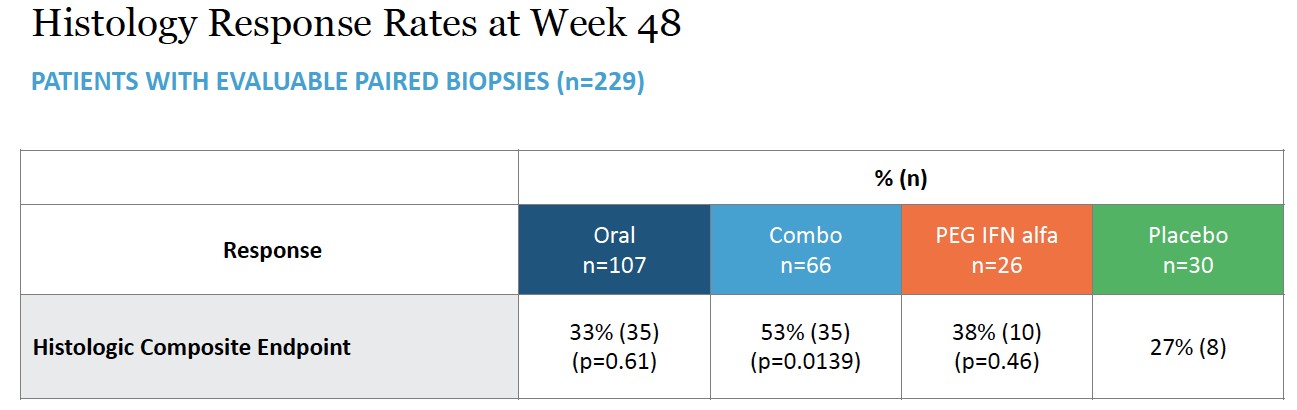
The key secondary histological endpoint was defined as ≥2-point improvement in histological activity index (HAI) and no worsening of Ishak fibrosis scoring as determined by blinded assessment of paired liver biopsies (n=229) collected at baseline and Week 48. This was demonstrated in 35 of 66 patients (53%, p=0.0139) with statistical significance in the combination arm versus 8 of 30 patients (27%) receiving placebo. Response was demonstrated in 35 of 107 patients (33%, p=0.61) in the all-oral arm versus placebo. Response in the peginterferon alfa comparator arm was 10 of 26 patients (38%).
Remaining secondary endpoints including virologic, biochemical, and composite responses at (24-weeks post-treatment) were reported at EASL in June 2023. At 24 weeks after cessation of treatment, response rates for both lonafarnib-based regimens were incrementally greater than those at Week 48.
Our Second HDV Program: Peginterferon Lambda (lambda) for HDV
Due to the safety signal observed in the LIMT-2 Phase 3 Study we will no longer pursue development of lambda for the treatment of chronic HDV. We are still exploring opportunities to use IFN-lambda for the treatment of COVID-19 and other important respiratory viral infections such as influenza and respiratory syncytial virus (RSV).
Congenital Hyperinsulinism (HI) Overview
Congenital Hyperinsulinism (HI) is an ultra-rare pediatric metabolic disease. HI presents with persistent fasting hypoglycemia, usually in the neonatal period, requiring intensive care hospitalization, administration of high rates of intravenous glucose through central lines, continuous intravenous administration of glucagon and in many instances surgical treatment by subtotal pancreatectomy during the neonatal period or during infancy. Persistent hypoglycemia can lead to permanent brain damage with neurodevelopmental deficits in up to 50% of patients, and subtotal pancreatectomy
ultimately leads to life-long insulin-dependent diabetes (IDDM) in most instances. Safe and effective therapies are urgently needed to prevent brain damage, IDDM, and death.
From an etiologic perspective, loss-of-function mutation in the ATP-sensitive K+ (KATP) channel is responsible for the most common and severe form of HI (KATP HI). This mutation leads to constitutive plasma membrane depolarization of the pancreatic beta cell resulting in elevated cytosolic calcium (Ca2+) and dysregulated insulin secretion, as illustrated in the below figure. Avexitide binds to the glucagon-like peptide-1 (GLP-1) receptor. In preclinical studies, GLP-1 receptor antagonism has been shown to lower baseline cAMP levels, resulting in decreased insulin secretion despite elevated Ca2+ levels.

This most common and severe form of HI is refractory to medical treatment with diazoxide and affects approximately 60% of all patients. This form of HI results in focal and diffuse forms of HI. Focal disease, occurring in approximately 50% of patients with KATP HI, resolves in approximately 97% of cases after partial pancreatectomy. Diffuse disease persists in 41% of cases after subtotal pancreatectomy. However, by the age of 14 years old, data show that 91% of patients undergoing subtotal pancreatectomy develop insulin-dependent diabetes, at which point hyperinsulinism is no longer present. Thus, HI is a congenital, rare pediatric disease with life-threatening manifestations of severe hyperinsulinemic hypoglycemia occurring primarily during the neonatal period, infancy, childhood and adolescence.
Clinical Data to Date
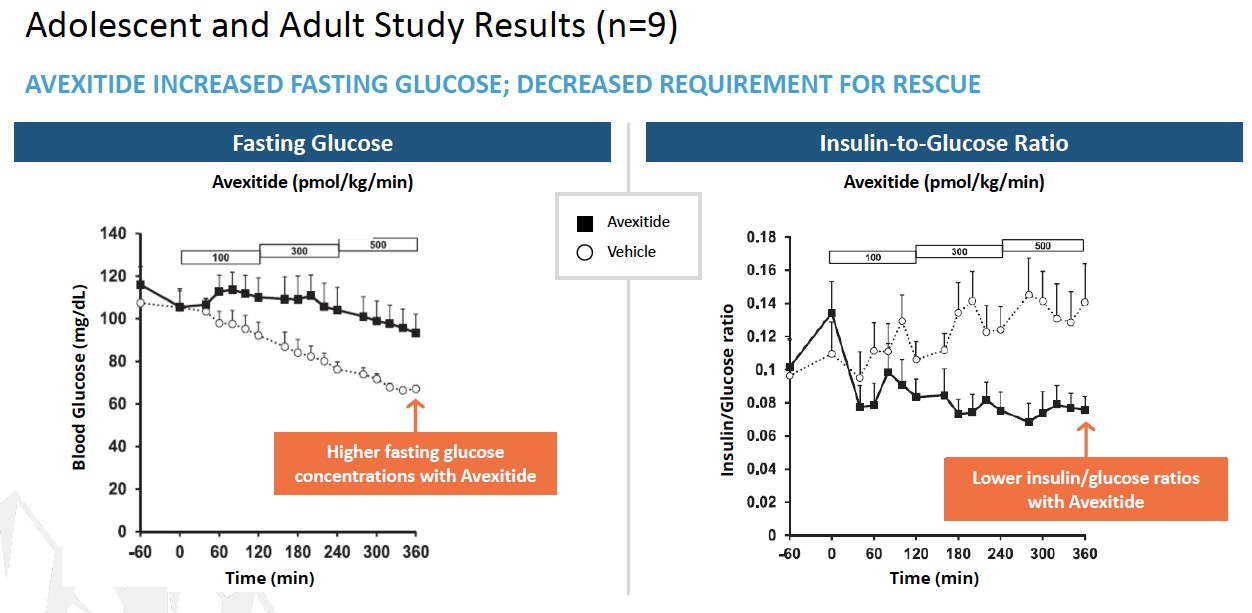
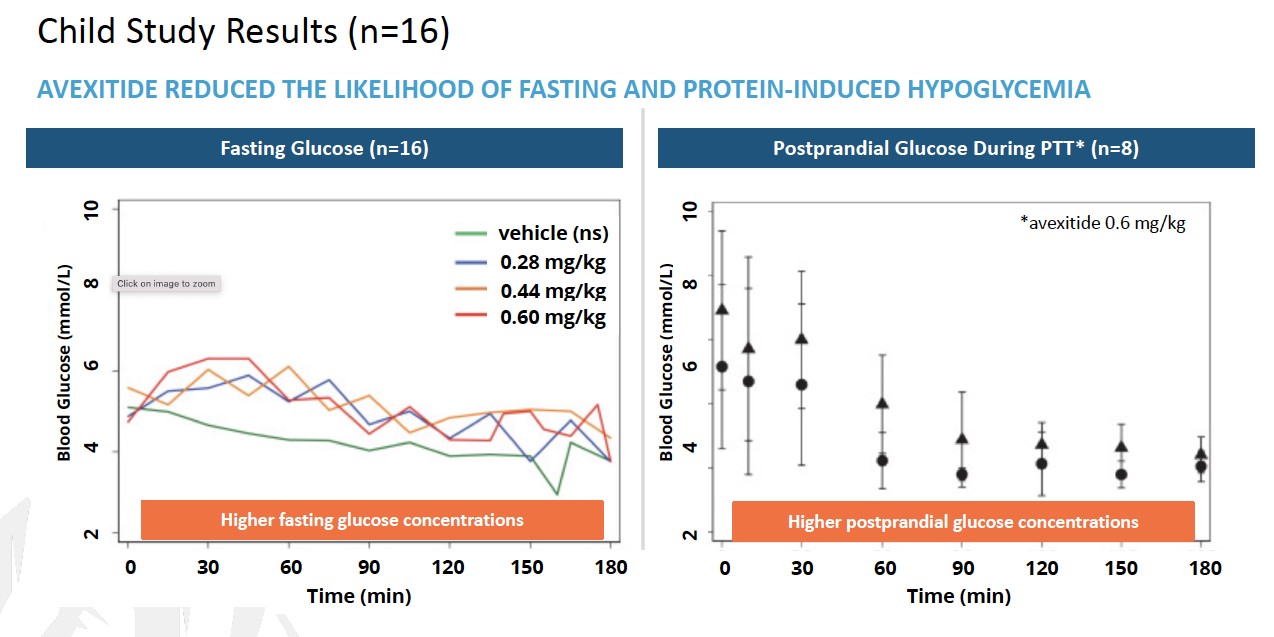
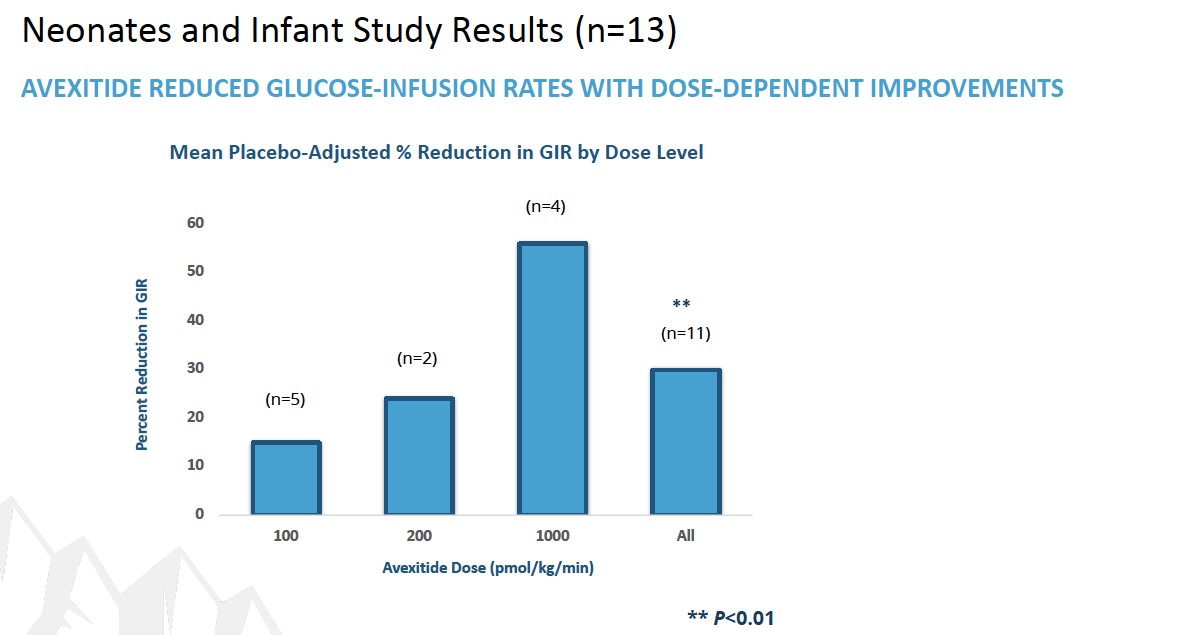
To date, a total of 39 patients with KATP HI enrolled in 3 clinical investigations at Children’s Hospital of Philadelphia (CHOP) have received avexitide administered by continuous IV infusion: 10 adolescents and adults, 16 children, and 13 neonates and infants. Data generated across all 3 clinical investigations suggest that GLP-1 receptor antagonism may effectively reduce fasting and protein-induced hypoglycemia in pediatric patients with HI, and may reduce glucose infusion requirements in neonates and infants with HI. Avexitide treatment was well tolerated, with no serious drug-related adverse
events (AEs) reported. Data from each of the three studies are presented in the figures below. Avexitide for treatment of HI has been granted Breakthrough Therapy Designation and Rare Pediatric Disease designation by the FDA.
In April 2022, Eiger announced it is initiating the Phase 3 registrational program for avexitide in congenital hyperinsulinism (HI) by the end of the year. In connection with our Phase 3 startup activities, we observed low levels of product-related impurities in the finished drug product. Although not unusual for this class of compounds, we are working with our CMOs to control and qualify these materials and plan to initiate dosing when an adequate supply of materials with a sufficient shelf life has been released. Avexitide has been granted Orphan Drug designation in the U.S. by the FDA for
the treatment of hyperinsulinemic hypoglycemia (which includes HI) and has also been granted Rare Pediatric Disease designation making it potentially eligible for a priority review voucher upon regulatory approval. To our knowledge, avexitide is the only investigational therapy for HI that has been granted Breakthrough Therapy designation by the FDA.
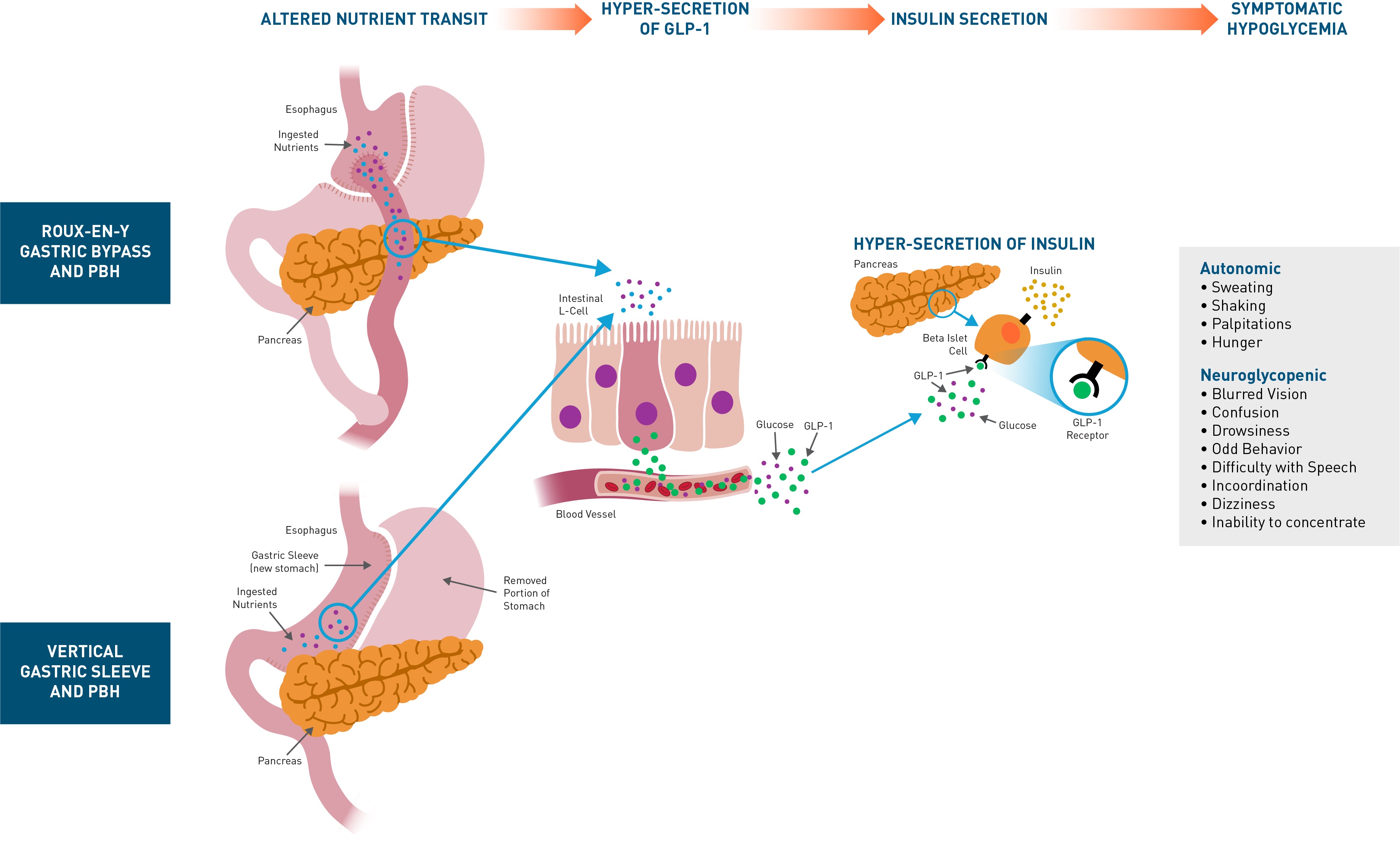
Post-bariatric Hypoglycemia (PBH) Overview
As the use of bariatric surgical procedures has increased worldwide due to rising obesity and Type 2 diabetes, hypoglycemia related to bariatric surgery (PBH) has been increasingly reported. PBH can occur after procedures that involve reducing the size of the stomach with a sleeve gastrectomy (SG) or resecting and re-routing the small intestine to a small stomach pouch (Roux-en-Y gastric bypass). This disorder leads to frequent symptomatic hypoglycemia, often resulting in glucose concentrations low enough to cause seizures, altered mental status, loss of consciousness, cognitive dysfunction, disability and death. Quality of life can be severely diminished, and many patients cannot care for themselves or others, work, drive, or be left alone. There is no approved treatment for this condition.
PBH is characterized by inappropriately high insulin concentrations when glucose levels are low and usually occurs 1–3 hours after oral nutrient ingestion, with hyperinsulinemia occurring in response to oral but not intravenous glucose, due to an exaggerated incretin effect. Plasma concentrations of glucagon-like peptide-1 (GLP-1), secreted by intestinal L-cells in response to oral nutrients, are markedly elevated after meal intake in affected patients. GLP-1 is a gastrointestinal hormone secreted in response to meals that binds to GLP-1 receptors on the beta cells of the pancreas, enhancing the release of insulin and lowering blood glucose levels. In patients with PBH, exaggerated GLP-1 secretion results in dysregulated secretion of insulin and resultant symptomatic hypoglycemia, as shown in the below figure.
GLP-1 excess with postprandial hypoglycemia has also been described as a complication of other GI surgeries, including gastrectomy, esophagectomy, and Nissen fundoplication.
Clinical Data to Date
Five clinical studies involving over 70 patients with PBH have demonstrated that pharmacologic blockade of the GLP-1 receptor can reduce hypoglycemia and associated symptoms. We believe that avexitide may represent the first targeted medical treatment for patients with PBH and hypoglycemia related to other GI surgeries. In studies completed to date, there have been no serious adverse events attributed to avexitide.
Phase 2 PREVENT Clinical Trial
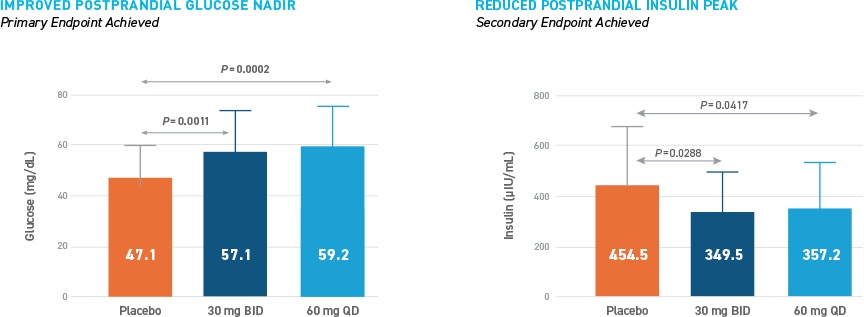
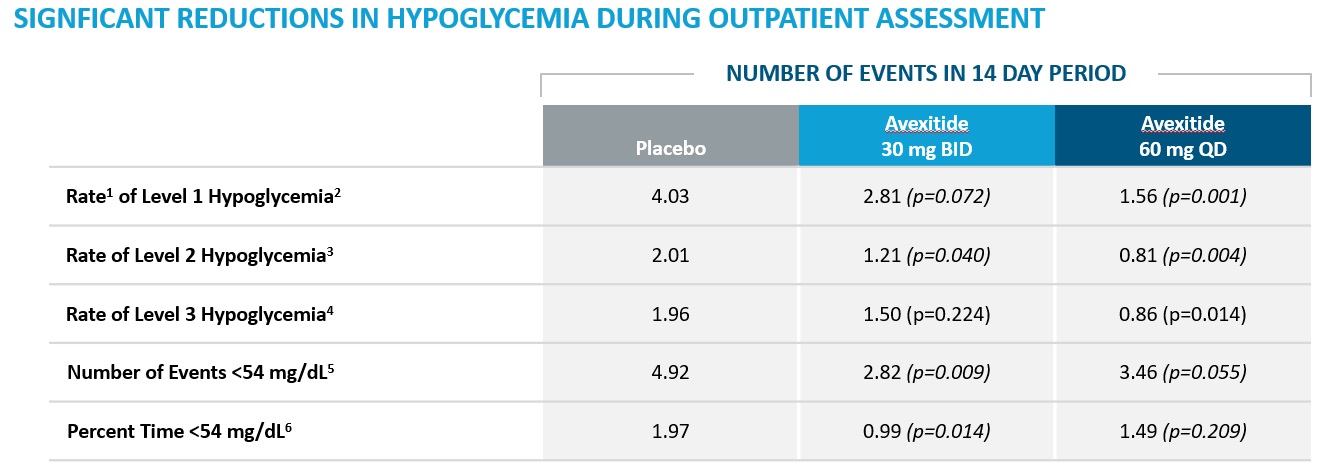
The PREVENT study was a Phase 2, multicenter, randomized, single-blind, placebo-controlled cross-over study to assess the efficacy and safety of 28-day dosing of avexitide in patients with PBH. A total of 18 patients were enrolled and treated with two dosing regimens of avexitide once daily (QD) and twice daily (BID) for 28 days. All patients participated in three 14-day treatment periods, involving placebo subcutaneous (SC) injections, avexitide 60 mg QD SC injections, and avexitide 30 mg BID SC injections. Patients self-administered injections in the outpatient setting throughout the study duration and additionally underwent in-clinic mixed meal tolerance test (MMTT) provocations with concomitant blood draws and symptom assessments following each 14-day treatment period. Participants' metabolic and clinical outcomes were assessed in the outpatient setting by electronic diary (eDiary), self -monitoring of blood glucose (SMBG), and continuous glucose monitoring (CGM) and postprandial glucose, insulin, and symptom responses were assessed during MMTT provocation in the in-clinic setting. The study schematic is shown below.

The primary efficacy endpoint of improved postprandial glucose nadir during MMTT was achieved with statistical significance with avexitide 30 mg BID and 60 mg QD, with fewer participants requiring glycemic rescue during each of the active dosing regimens than during placebo dosing. The secondary endpoint of reduced postprandial insulin peak during MMTT was also statistically significant with avexitide 30 mg BID and 60 mg QD. These results are shown in the figure below.
Metabolic and clinical improvements were also observed in the outpatient setting as assessed by eDiary, SMBG and blinded CGM, with fewer episodes of Levels 1-3 hypoglycemia observed during both avexitide dosing regimens as compared to during placebo dosing, as shown in the table below.
1Rate is defined as number of episodes in a 14 day period
2Level 1 Hypoglycemia is defined as hypoglycemia symptoms confirmed by SBGM concentrations of <70 mg/dL
3Level 2 Hypoglycemia (Clinically Important Hypoglycemia) is defined as SMBG < 54 mg/dL (3.0 mmol/L)
4Level 3 Hypoglycemia (Severe Hypoglycemia) is defined as a severe event characterized by altered mental and/or physical functioning that requires assistance from another person for recovery. This applies regardless of whether a patient actually receives external assistance
5Number of events <54 mg/dL is defined as the number of episodes with CGM values <54 mg/dL sustained for at least 15 min during daytime hours (8AM to midnight) during each treatment period normalized to 14 days
6Percent time is expressed as the percentage of CGM values <54 mg/dL during daytime hours (8am-12am) during each treatment period normalized to 14 days
Avexitide was well tolerated. There were no treatment-related serious adverse events and no participant withdrawals.
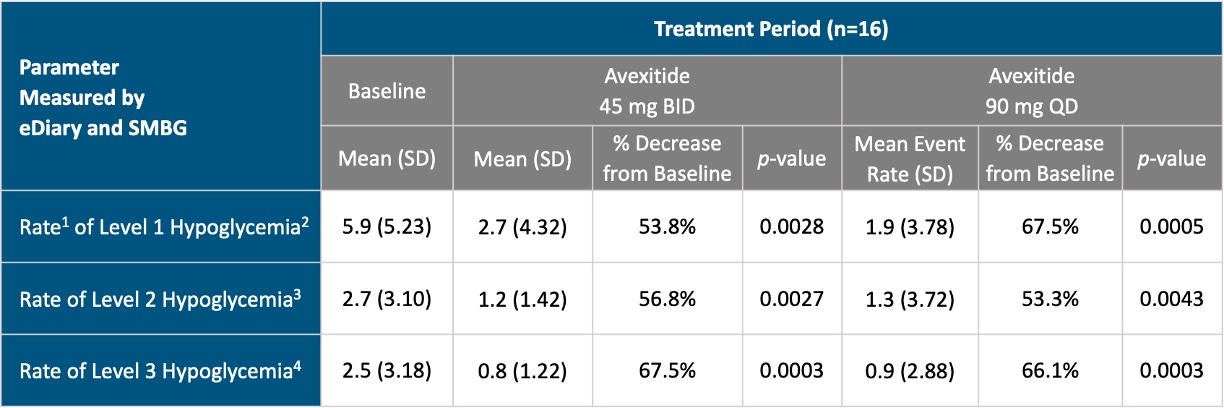
Phase 2b Clinical Trial
This Phase 2b, single-center, open-label, investigator-initiated, cross-over study assessed the safety and efficacy of 90 mg total daily dose of avexitide administered as a once daily 90 mg dose (QD) and as a 45 mg twice daily dose (BID) compared to standard of care (MNT) in patients with severe, diet-refractory hyperinsulinemic hypoglycemia, including patients with PBH after RYGB or VSG, and patients with hypoglycemia related to other GI surgeries, including total gastrectomy, or Nissen fundoplication. In this study, patients underwent a 14-day Run-In period consisting of standard of care (MNT). Enrolled patients were then randomized to 14 days of avexitide 90 mg QD treatment (Period 1) followed by 14 days of avexitide 45 mg BID treatment (Period 2), or vice versa, with a 2-day washout in between. Throughout the 14-day Run-In period and the two treatment periods, metabolic and symptomatic parameters were assessed by SMBG, eDiary, and blinded CGM, while adhering to MNT. The study schematic is shown below.

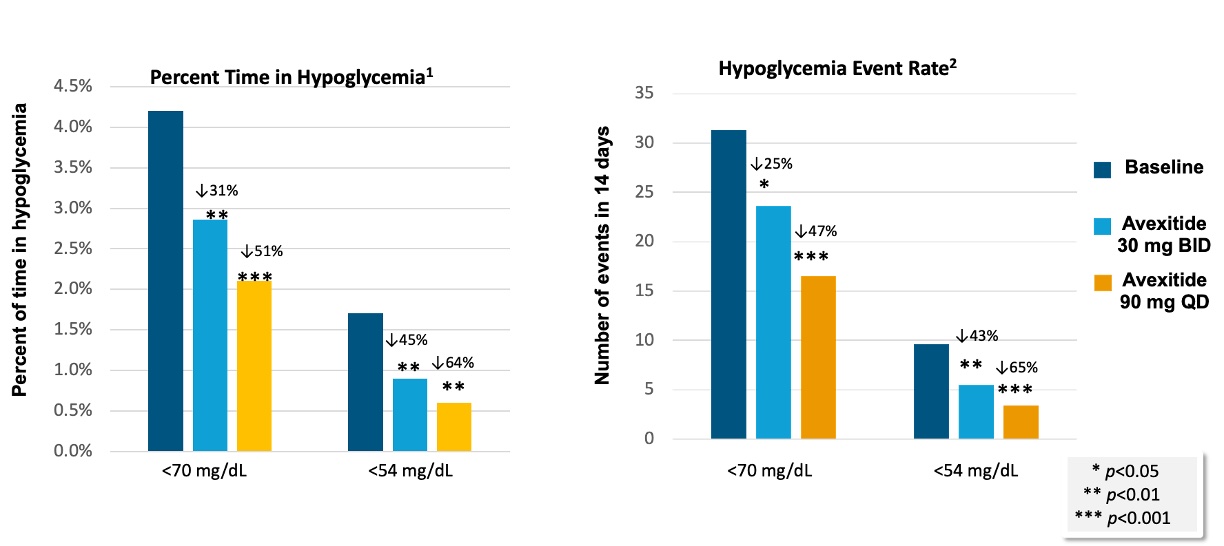
Compared with MNT alone, avexitide 45 mg BID and 90 mg QD significantly reduced the rates of Levels 1, 2, and 3 hypoglycemia as measured by SMBG and eDiary. Objective assessment by blinded CGM data corroborated SMBG and eDiary results, with both dosing regimens demonstrating significant reductions in hypoglycemia events and time spent in hypoglycemia. A greater magnitude of effect was observed with once daily (90 mg QD) vs. the twice daily (45 mg BID) dosing regimen, as measured by blinded CGM. These results are shown in the table and figure below.
1.Rate is defined as number of episodes in a 14 day period
2.Level 1 Hypoglycemia is defined as hypoglycemia symptoms confirmed by SBGM concentrations of <70 mg/dL
3.Level 2 Hypoglycemia (Clinically Important Hypoglycemia) is defined as SMBG < 54 mg/dL (3.0 mmol/L)
4.Level 3 Hypoglycemia (Severe Hypoglycemia) is defined as a severe event characterized by altered mental and/or physical functioning that requires assistance from another person for recovery. This applies regardless of whether a patient actually receives external assistance
1.Percent time is expressed as the percentage of CGM values that are <70 mg/dL or <54 mg/divided by the total CGM wear time
2.Event Rate is defined as number of events below range (70 or 54 mg/dL) sustained for at least 15 min during each treatment period as measured by CGM, normalized to 14 days.
Avexitide was well-tolerated with no safety signals identified and no participant withdrawals. Adverse events (AEs) were mild to moderate in severity and transient. The most common AEs included diarrhea, headache, bloating, and injection site reaction / bruising.
Manufacturing
We currently contract with third parties for the Chemistry, Manufacturing and Controls (CMC) development and commercial manufacturing of our FDA-approved product, Zokinvy, and all of our clinical product candidates, and intend to do so in the future. We do not own or operate manufacturing facilities for the production of clinical trial materials of our product candidates and have no plans to build our own clinical or commercial scale manufacturing capabilities. We believe that the use of contract development and manufacturing organizations (CDMOs) eliminates the need for us to directly invest in laboratories, manufacturing facilities, equipment and additional staff. Although we rely on contract development partners and manufacturers, our personnel and consultants have extensive manufacturing experience overseeing our CDMOs.
To date, our third-party labs and manufacturers have met the development and manufacturing requirements for the product candidates. We expect existing third-party labs and manufacturers to be capable of providing sufficient quantities of our
product candidates to meet anticipated launch requirements and supply initial commercial demands. We have identified or plan to identify additional commercial sources of capacity as we move our product candidates to and through Phase 3 clinical trials as dictated by projected demand. We believe there are alternate sources of manufacturing that could be identified and enabled to satisfy our clinical and commercial requirements, however, we cannot be certain that identifying and establishing alternative relationships with such sources can be successful, cost effective, or completed on a timely basis without significant delay in the development or commercialization of our product candidates. All of our third-party manufacturers are subject to periodic audits to confirm compliance with applicable regulations and must pass inspection before we can manufacture our products for commercial sales.
We have qualified and finalized or are in process of finalizing commercial supply agreements with contract manufacturers of Zokinvy and Jitixib (lonafarnib). The brand name Jitixib was given conditional approval by the US FDA for lonafarnib 25mg capsules. Having a distinct brand name for lonafarnib for the treatment of HDV will ensure there is no confusion with the strengths of lonafarnib (50mg/75mg) used for HGPS and marketed as Zokinvy. In our NDA for lonafarnib 25mg capsules we will seek final approval to use Jitixib as the brand’s trade name. We have identified commercial manufacturers for lambda and avexitide and plan to proceed with qualifications as we enter Phase 3 and prepare for regulatory submissions.
Lonafarnib
The drug product for completed lonafarnib (LNF) Phase 2 clinical studies for the treatment of HDV and HGPS was manufactured by Merck. We have successfully completed the technology transfer for manufacture of the LNF drug substance and the LNF drug product to our third-party manufacturers. All future HDV clinical trials will be conducted with product manufactured by these CMOs. These manufacturers produce our commercial supply for HGPS and processing-deficient progeroid laminopathies.
Lambda
We have completed the technology transfer from BMS to our CDMOs for our lambda program. As part of the license agreement, sufficient inventory of product was obtained to complete our Phase 2 and initiate our Phase 3 clinical trials. We have manufactured additional batches of lambda products and demonstrated equivalence to the materials produced by BMS.
Both lonafarnib and lambda are GMP products from all CDMOs.
Avexitide
The drug substance and drug product used for the production of avexitide clinical trial material for the treatment of HI and PBH is manufactured by our third-party CDMOs.
Intellectual Property
We strive to protect and in-license those proprietary technologies, inventions, and improvements we believe are important to our business. We seek and maintain, where available, patent protection for our product candidates including: composition dosage, formulation, use, manufacturing process, among others. We have also licensed patents and patent applications that cover certain of our product candidates and/or their manufacture, use, or formulation. We also rely on trade secrets, know-how, and continuing innovation to develop and maintain our competitive position. We cannot be certain that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents granted to us in the future will be commercially useful in protecting our technology.
We also rely, or plan to rely, on regulatory exclusivity, including Orphan Drug designation and New Chemical Entity (NCE) and Biologic License Application (BLA) exclusivities, as well as trade secrets and carefully monitor our proprietary information to protect all aspects of our business.
We plan to continue to expand our intellectual property portfolio by filing patent applications for our product candidates. We file and prosecute patent applications in the United States and Europe and, when appropriate, additional countries, including Japan, Korea, Canada and China.
Our success will depend significantly upon our ability to: (i) obtain and maintain patents and other exclusivity protections for commercially important technology, inventions and know-how related to our business; (ii) prosecute our patent applications to issue as patents and defend and enforce our patents; (iii) maintain our licenses to use intellectual property owned by others; (iv) preserve the confidentiality of our trade secrets, and (v) operate without infringing the valid and enforceable patents and other proprietary rights of others. In addition to maintaining our existing proprietary assets, we seek to strengthen our proprietary positions when economically reasonable to do so. Our ability to augment our proprietary position relies on its: (i) know-how; (ii) ability to access technological innovations, and (iii) ability to in-license technology when appropriate.
The patent positions of pharmaceutical/biotechnology companies like us are generally uncertain and involve complex legal, scientific, and factual issues. In addition, the scope claimed in a patent application can be significantly reduced during the patent prosecution process before any patent issues. After issuance of a patent, if the issued patent is challenged, then the courts or a patent office can redefine the scope of the patent, including by invalidating some or all of the patent claims, or rendering the patent unenforceable in its entirety. Consequently, we do not know with certainty whether patents will issue in each country in which we or our licensors file patent applications, or if those patent applications, if ever issued, will issue with claims that cover our product candidates, or, even if they do issue, whether the patent or its relevant claims will remain enforceable upon challenge. Accordingly, we cannot predict with certainty whether the patent applications we are currently pursuing will issue as patents in a particular jurisdiction or whether the claims of any issued patents will provide sufficient proprietary protection from potential competitors to make any of our products commercially successful. Any of our patents, including already issued in-licensed patents or any patents that may issue to us or our licensors in the future, could be challenged, narrowed, circumvented, or invalidated by third parties. Newly filed patent applications in the United States Patent and Trademark Office (the USPTO) and certain other patent offices are maintained in secrecy for a minimum of 18 months, and publications of discoveries in the scientific or patent literature often lag far behind the actual discoveries themselves. For these reasons, we cannot be certain that inventions claimed in pending patent applications were not invented by another party prior to our invention or disclosed or claimed in a patent application filed before the effective filing date of our applications, in either of which case the claims may not be patentable to us. For certain applications with an effective filing date prior to March 13, 2013, we may have to participate in interference proceedings declared by the USPTO to determine priority of invention. Also, while we are not currently participating in any interferences or post-grant challenge proceedings, such as patent oppositions, post-grant reexamination proceedings, inter parties review proceedings and patent litigation, that seek to invalidate claims of pending patent applications or issued patents, we may have to participate in such proceedings in the future. Such proceedings could result in substantial cost, even if the eventual outcome is favorable to us.
The term of individual patents depends upon the legal term of the patents in the countries where they are issued. In most countries, the standard patent term for inventions relating to human drugs and their formulation and use is 20 years from the date of filing the first non-provisional patent or international application under the Patent Cooperation Treaty of 1970 (PCT).
The PCT is an international patent law treaty that provides a single PCT application can be converted into a patent application in any of the more than 154 PCT contracting states, providing a cost-effective means for seeking patent protection in numerous regions or countries. Conversion of a PCT application into an application in any of the contracting states typically occurs about 30 months after a priority application is filed, or about 18 months after the PCT application filing date. An applicant must undertake prosecution within the allotted time in the patent offices of any, or a combination, of the contracting states or in a regional patent office it determines to undertake patent issuance in protection in such country or territory.
We own or in-license a number of patents in the U.S. and foreign countries that cover our products, product candidates, and methods of their use. With respect to our owned or in-licensed issued patents in the U.S. and Europe, we may be entitled to obtain an extension of patent term to extend the patent expiration date. For example, in the U.S., this extended coverage period is known as patent term extension (PTE) and can only be obtained provided we apply for and receive a marketing authorization for a product. The period of extension may be up to five years beyond the expiration of the patent, but cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval. Only one patent among those eligible for an extension may be extended. In Europe, Supplementary Protection Certificates (SPC) may also be available to patents, which would be available by applying to the member states. However, there is no guarantee that the applicable authorities, including the FDA, will agree with our assessment of whether such extensions should be granted, and even if granted, the length of such extensions. The exact duration of the extension depends on the time we spend in clinical studies as well as getting marketing approval from the FDA.
Patent Protection of Our Product Candidates
The exclusivity positions for our clinical-stage product candidates as of December 31, 2023 are summarized below.
LNF. We have licensed from Merck a portfolio of patents and know how covering the compound, formulations of the compound, and synthesis, but these expire before the anticipated launch date of the LNF product candidate. In the United States, we have obtained patent protection for the use of LNF in combination with RTV for the treatment of HDV infection. Eiger’s U.S. Patent Nos. 10,076,512; 10,828,283; and 11,311,519 entitled, Treatment of Hepatitis Delta Virus Infection, include claims covering a broad range of RTV-boosted LNF doses and durations with and without interferons. The patents have terms that extend at least until 2035. Additional claims are being pursued in a continuation application. The European Patent Office, the Chinese Patent office and the Japanese Patent Office have also granted patents with claims covering a broad range of LNF boosted with RTV dosing regimens for the treatment of HDV infection. These patents will have a term that extends to 2035. In Europe, China and Japan, additional claims are being pursued in divisional applications. We have now obtained patent protection with claims covering treatment of HDV with LNF boosted with RTV in the U.S., Europe, China and Japan, which are key major pharmaceutical markets. A corresponding patent application claiming the use of lonafarnib boosted with ritonavir is pending in Korea. Additionally, US Patent 10,835,496 and a European Patent issued claiming particular dosage forms for ritonavir-boosted lonafarnib for the treatment of HDV infections. A Notice of Allowance was also received for an application in South Korea. These patents extend protection until at least 2036.
We have in-licensed from The Progeria Research Foundation patents covering the methods of treating Hutchinson-Gilford progeria syndrome (HGPS), and progeroid laminopathies. The patents provide protection until at least 2024 and an application for patent term extension (PTE) that could extend the protection for one of the patents until 2029 has been filed. We have also filed a patent application in the U.S. directed to methods of treating HGPS and progeroid laminopathies that if issued would provide further protection until at least 2039. In addition, LNF has been granted Orphan Drug designation by the FDA and the EMA in this indication, which respectively may provide up to seven and ten years of regulatory exclusivity.
Lambda. We have in-licensed from BMS a portfolio of patents relating to the manufacture, use, and compositions of interferon lambda modified by polyethylene glycol derivatization (lambda). The key United States composition of matter patent in this portfolio expires in 2025, but we expect to be eligible for the full five years of patent term extension for that patent. In addition, we expect regulatory approval for lambda to be filed under a BLA, which if granted would provide 12 years reference product exclusivity (4 years in filing exclusivity; 12 years for data), as well as Orphan Drug exclusivity for treatment of HDV infection.
In the United States and Europe, we have obtained patent protection for the use of lambda for treatment of HDV infection. U.S. Patent No. 10,953,072 issued with claims covering the use of lambda to treat HDV. The term of this patent extends at least until 2037. The European Patent Office has also granted a patent with claims covering use of lambda to treat HDV. Additional claims are being pursued in continuation/divisional applications in the U.S. and Europe. Applications are also pending in China, Japan and Korea. Any patents that issue from these applications will expire in 2037.
We also filed a PCT application relating to the use of lambda in HDV that has matured into patent applications in the United States, Europe, Australia, Brazil, Canada, China, Eurasia, Israel, India, Japan, South Korea, Mexico, and South Africa. Any patents that issue from these applications should offer protection until at least 2039.
In addition, we have filed a PCT application related to the use of lambda and lonafarnib and ritonavir to treat HDV, that has matured into patent applications in the United States, Europe, Australia, Brazil, Canada, China, Israel, Japan, South Korea, Mexico, Russia, Ukraine, and South Africa. Any patents that issue from these applications should offer protection until at least 2040. Lastly, we also filed a PCT application related to the use of lambda to treat COVID-19 that has matured into patent applications in the United States, Europe, Australia, Brazil, Canada, Chile, China, Columbia, Eurasia, Israel, Japan, South Korea, Mexico, New Zealand, Saudi Arabia, Singapore, Ukraine, Vietnam, and South Africa.
Avexitide. We have in-licensed from Stanford two PCT applications that claim the use of avexitide and other agents in the treatment of hypoglycemia associated with bariatric surgery, including in PBH. The USPTO issued US 10,639,354, US 10,660,937, US 10,993,992, and US 10,993,991, which provide protection at least until 2036. Up to five years of patent term extension will be available in the United States. The European Patent Office has also granted two patents with claims covering use of avexitide to treat hypoglycemia, which will expire in 2036. Two Australian patents and a Chilean patent
have also issued with similar claims, which will also expire in 2036 Additional claims are being pursued in continuation/divisional applications in the U.S. and Europe. Applications are also pending in Canada, and Chile. Any patents that issue from these applications will expire in 2036. Additionally, avexitide has been granted Orphan Drug designation in the treatment of hyperinsulinemic hypoglycemia by the FDA and the EMA, which provides seven years and ten years of regulatory exclusivity in the United States and Europe, respectively.
In the United States, we have obtained patent protection for formulations of avexitide and the use of these formulations in the treatment of hypoglycemia associated with bariatric surgery. The USPTO issued US 11,020,484, which will provide protection at least until 2037. Additional claims are being pursued in continuation application. Applications are also pending in Europe, Australia, Brazil, Canada, Chile, China, Hong Kong, Israel, India, and Japan. Any patents that issue from these applications will expire in 2037.
We have also filed a PCT application related to method of treating HI and PBH, that has matured into patent applications in the United States, Australia, Brazil, Canada, Chile, China, Europe, Israel, and Japan. If issued, the patents will provide protection until 2039. We have also filed a PCT application related to method of treating congenital hyperinsulinism, which will offer protection until 2042 if issued into patents at the national stage.
We have in-licensed patents and patent applications from the Trustees of the University of Pennsylvania (UPenn) and Children’s Hospital of Philadelphia (CHOP), relating to hyperinsulinemia hypoglycemia. The in-licensed patents and applications relate to multiple hyperinsulinemic disorders, including PBH and HI. The patents, which are issued in the U.S. and Europe, provide protection until 2028. There are continuation applications pending from which we are pursuing additional claim coverage.
Other Proprietary Rights and Processes
We also rely on trade secret protection for some of our confidential and proprietary information. It is our policy to require our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors to execute confidentiality agreements upon the commencement of employment or consulting relationships with us. These agreements provide that all confidential information concerning our business, scientific, development or financial affairs that are either developed or made known to the individual during the course of the individual’s relationship with us are to be kept confidential and not disclosed to third parties except in specific circumstances. Although we take steps to protect our proprietary information and trade secrets, including through contractual means with our employees and consultants, third parties may independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets and disclose our technology. If these events happen, we may not be able to meaningfully protect our trade secrets.
Our agreements with employees also provide that all inventions conceived by the employee in the course of employment with us or based on the employee’s use of our confidential information are our exclusive property or that we have an exclusive royalty free license to use such technology.
Competition
The biopharmaceutical industry is highly competitive. We face competition from major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide. Given the significant unmet medical need for novel therapies to treat Progeria, chronic hepatitis delta virus infection, congenital hyperinsulinism, post-bariatric hypoglycemia, and COVID-19, these conditions are where various treatments from many companies are used and where many public and private universities and research organizations are actively engaged in the discovery, research and development of product candidates. As a result, there are and will likely continue to be extensive resources invested in the discovery and development of new products to treat these unmet medical needs. We anticipate facing intense and increasing competition as new products enter the market and advanced technologies become available.
In addition, there are numerous multinational pharmaceutical companies and large biotechnology companies currently marketing or pursuing the development of products or product candidates targeting the same indications as our product candidates. Many of our competitors, either alone or with strategic partners, have or will have substantially greater financial, technical and human resources than us. Accordingly, our competitors may be more successful than us in developing or marketing products and technologies that are more effective, safer or less costly. Additionally, our competitors may obtain regulatory approval for their products more rapidly and may achieve more widespread market acceptance. Accelerated mergers and acquisitions activity in the biotechnology and pharmaceutical industries may result in
even more resources being concentrated among a smaller number of our competitors. These companies also compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical study sites and patient registration for clinical studies and acquiring technologies complementary to, or necessary for, our programs. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
Our potential competitors and the related stage of development of their product candidates in target indications is as follows:
•HDV: Gilead (conditional approval in Europe, BLA submitted in the U.S.), Vir (Phase 2), Janssen Research & Development, LLC (Phase 2), Replicor, Inc. (Phase 2), PharmaEssentia (Phase 2), Albireo (preclinical), Assembly (preclinical)
•HGPS and Progeroid Laminopathies: PRG Science & Technology (Phase 1)
•HI: Zealand Pharmaceuticals (Phase 3 completed), Rezolute, Inc (Phase 3 Ex-US), Hamni (Phase 2)
•PBH: Recordati (Phase 2), Vogenyx (Phase 2), MBX (Phase 1/2)
There are other therapies that are used or may be used for our targeted indications, and these other products in clinical development or marketed for other indications may be used in competition with our product candidates if we are able to identify potential market opportunities of interest. For example, HDV has not been generally identified as a target for development compared to hepatitis B or hepatitis C, and products on the market or in development for those indications may potentially be tested in HDV as the understanding of the potential medical need for therapies in this indication become more widely understood.
We believe that the key competitive factors that will affect the development and commercial success of our product candidates are efficacy, safety and tolerability profile, convenience in dosing, product labeling, cost-effectiveness, price, the level of generic competition and the availability of reimbursement from the government and other third-parties. Our commercial opportunity could be reduced or eliminated for any of our products if our competitors have products that are approved earlier than our product candidates or are superior compared to our product candidates or if our product candidates do not result in an improvement in condition compared to those other products.
License and Asset Purchase Agreements
License Agreement with Merck
In September 2010, we entered into an exclusive license agreement with Schering Corporation, subsequently acquired by Merck, which provides us with the exclusive right to develop and commercialize lonafarnib. As consideration for such exclusive right, we issued Private Eiger convertible preferred stock with a fair value of $0.5 million when the agreement was executed in September 2010. This preferred stock was converted to 911 shares (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) of common stock upon the Merger. In addition, we are obligated to pay Merck up to an aggregate of $27.0 million in development milestones and will be required to pay tiered royalties based on aggregate annual net sales of all licensed products ranging from mid-single to low double-digit royalties on net sales. Our obligation to pay royalties to Merck expires on a country-by-country and product-by-product basis on the later of the expiration of the last to expire patent assigned to us under the agreement on the tenth anniversary of the first commercial sale of the product. In May 2015, the first regulatory milestone was achieved, and we paid the related milestone payment of $1.0 million to Merck. Upon completion of D-LIVR, we will owe Merck a $1.0 million milestone. We announced primary topline data of D-LIVR in December 2022, and in September 2023, the Phase 3 study was completed and the $1.0 million related milestone expense was recorded in accrued liabilities on the consolidated balance sheet. The final results are presented in the above section on lonafarnib.
The Merck License will continue for so long as we owe royalty payments to Merck under the agreement. Each party has the right to terminate the Merck License Agreement for the other party’s uncured material breach or bankruptcy, including the Chapter 11 Cases. Merck also has the right to terminate the agreement if we discontinue development and commercialization of LNF for a specified period of time. In addition, we have the right to terminate the agreement, with notice, for any reason.
On May 15, 2018, we entered into an amendment to the exclusive license agreement with Merck, which provides for expansion of the existing exclusively licensed field of use under the license agreement with Merck to include all uses of lonafarnib related to the treatment of Hutchinson-Gilford progeria syndrome (HGPS) in humans at no cost to us. We have
the sole responsibility and the continuing obligation for the manufacture and supply of lonafarnib to The Progeria Research Foundation. Merck will not receive milestone payments in relation to lonafarnib for the treatment of HGPS and progeroid laminopathies or any royalty payments for sales of lonafarnib to treat the currently estimated HGPS and progeroid laminopathies patient population worldwide. On November 3, 2020, we entered into an amendment to the exclusive license agreement with Merck which expanded the definition of HGPS to also include progeroid laminopathies.
Asset Purchase Agreement with AbbVie Inc.
On November 20, 2020, we entered into an asset purchase agreement (the AbbVie Agreement) with AbbVie Inc. (AbbVie) to sell our Rare Pediatric Disease Priority Review Voucher, which was awarded on November 20, 2020 upon approval by the FDA of our new drug application for Zokinvy® for HGPS and processing-deficient progeroid laminopathies (the PRV) to AbbVie. The AbbVie Agreement contains customary representations, warranties, covenants, and indemnification provisions subject to certain limitations.
In consideration for the PRV, AbbVie agreed to pay us $95.0 million. The transaction closed in January 2021. Such consideration was shared equally with The Progeria Research Foundation (the PRF) in accordance with the terms of our Collaboration and Supply Agreement, dated May 15, 2018, with the PRF, pursuant to which we and the PRF will equally share any proceeds from the sale of a priority review voucher that we may receive as the sponsor of a rare pediatric disease product application. We retained approximately $46.5 million of the net proceeds from the sale of the PRV.
Asset Purchase Agreement with Eiger Group International, Inc.
In December 2010, we entered into an Asset Purchase Agreement with Eiger Group International, Inc. (EGI) dated December 8, 2010 (the EGI APA). Dr. Jeffrey Glenn, a former member of our board of directors, is the sole owner of EGI.
Under the EGI APA, we purchased all the assets including intellectual property rights related to the use of farnesyltransferase inhibitors as anti-viral agents and methods to treat viral infection with those inhibitors. We also purchased all assets including intellectual property rights related to the use of inhibitors of prenylation, prenyl cysteine methyltransferase, and a specified protease as anti-viral agents and methods to treat viral infection with those inhibitors. We are obligated to use commercially reasonable efforts to develop and commercialize the licensed products in major markets.
Under the EGI APA, we paid EGI an upfront payment of $0.4 million. Additionally, we are obligated to pay EGI a low single-digit royalty based on aggregate annual net sales of products developed using the intellectual property. Within the first ten years after commercialization, we may make a one-time payment of $0.5 million for each contract for the three types of product related to such intellectual property that would reduce the payment term for the three products to the tenth anniversary of the first commercial sale. The obligation to pay royalties expires on a country-by-country and product-by-product basis on the later of either when the product is no longer sold in any country or the earliest of the tenth anniversary of the first commercial sale of the product.
The term of the EGI APA extends until expiration of all payment obligations, and we may terminate the agreement upon notice to EGI. EGI may terminate the EGI APA if we fail to use commercially reasonable efforts to develop and commercialize licensed products. In addition, each party may terminate the EGI APA for the other party’s uncured material breach or bankruptcy, including the Chapter 11 Cases. In the event of any termination, other than termination by us for EGI’s breach, we will assign the purchased assets back to EGI.
In November 2012, we entered into an agreement with EGI whereby we sold all of the assets related to the compound clemizole, including any related intellectual property. EGI is obligated to pay to us a high single-digit royalty on future aggregate annual net sales, subject to certain reductions and exceptions. EGI’s obligation to pay royalties expires on a country-by-country and product-by-product basis on the later of either expiration of the last to expire patent sold to EGI under the agreement or the earliest of the tenth anniversary of the first commercial sale of the product. As of December 31, 2022, the product has not achieved regulatory approval.
Asset Purchase Agreement with Tracey McLaughlin and Colleen Craig
We entered into an Asset Purchase Agreement with two individuals, Dr. Tracey McLaughlin and Dr. Colleen Craig (the Sellers) dated September 25, 2015 (the Exendin APA). We also entered into a consulting agreement with the Sellers as part of the agreement.
Under the Exendin APA, we purchased all the assets and the intellectual property rights related to the compound avexitide from the Sellers, including an assignment of a license agreement with Stanford which covered exclusive rights with respect to the compound avexitide. Under the assigned Stanford exclusive license agreement, we are obligated to pay Stanford a low, single-digit royalty on net sales after the first commercial sale of any product developed based on avexitide.
Under the Exendin APA, we are obligated to pay development milestone payments in aggregate up to $1.0 million to each of the Sellers and a low, single-digit royalty to each of the Sellers based on aggregate annual net sales of all products developed based on avexitide subject to certain reductions and exceptions. Our obligation to pay royalties expires on the expiration of the last to expire valid claim as defined in the agreement. We also agreed to retain each of the Sellers as consultants pursuant to consulting agreements, each with a term of one year, subject to annual renewal. The consulting agreements related to the Exendin APA have expired. During the year ended December 31, 2017, upon the successful completion of the Phase 2 trials, the development milestone was achieved, and we paid the related milestone payment of $0.1 million to each of the Sellers.
License Agreement with Bristol-Myers Squibb Company
In April 2016, we entered into a License Agreement and a Common Stock Purchase Agreement with Bristol-Myers Squibb Company, together BMS, the BMS Purchase Agreement and the BMS License Agreement. Under the BMS License Agreement, BMS granted us an exclusive, worldwide, license to research, develop, manufacture, and sell products containing the proprietary BMS molecule known as PEG-interferon Lambda-1a (peginterferon lambda or the Licensed Product) for all therapeutic and diagnostic uses in humans and animals.
We are responsible for the development and commercialization of the Licensed Product at our sole cost and expense. In April 2016, under the BMS License Agreement we paid an upfront payment of $2.0 million in cash and issued 5,252 shares (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) of our common stock to BMS with an aggregate fair value of $3.2 million. The BMS Purchase Agreement grants BMS certain registration rights with respect to the shares of common stock delivered, and BMS has agreed to certain trading and other restrictions with respect to the shares purchased.
Under the BMS License Agreement, we are obligated to make development and regulatory milestone payments totaling $61.0 million and commercial sales milestones of up to $128.0 million after the achievement of specified milestones. We are also obligated to pay BMS annual net sales royalties in the range of mid-single to mid-teens, depending on net sales levels. If we grant a sublicense, we are obligated to pay BMS a portion of the sublicensing income received. In March 2022, we recorded a $5.0 million milestone expense related to the initiation of the Phase 3 LIMT-2 study of lambda for HDV.
License Agreement with the Trustees of the UPenn and CHOP
In May 2019, we entered into a license agreement (the UPenn/CHOP Agreement) with the Trustees of the UPenn and the CHOP, under which we obtained an exclusive, royalty-bearing, worldwide license to develop, manufacture and sell certain GLP-1 receptor antagonist(s) products to treat all human and animal conditions. We also obtained an exclusive, royalty-bearing, sublicensable, worldwide license to certain data developed by CHOP. We are responsible for the development and commercialization of the licensed products at our sole cost and expense.
As part of the consideration for the rights granted to us under the UPenn/CHOP Agreement, we paid UPenn a one-time, non-refundable issue fee of $1.0 million in 2019. In addition, we are obligated to pay UPenn a specified annual license maintenance fee, up to $2.5 million in development milestones upon marketing authorization in one or more countries, and sales milestones of up to $18.0 million. We will also be required to pay UPenn a flat royalty in the low-single digits on our net sales of all licensed products, subject to specified reductions and offsets, and specified minimum annual royalty payments. Our obligation to pay royalties expires on a product-by-product and country-by-country basis, on the later of: (i) the expiration of the last valid claim in the licensed patents in any country, or (ii) the tenth anniversary of the first commercial sale of the product in such country. No milestones have been achieved as of December 31, 2023.
We may terminate the UPenn/CHOP Agreement in its entirety for any reason by providing prior written notice to UPenn and CHOP. UPenn or CHOP may terminate the UPenn/CHOP Agreement, upon a written notice, for our failure to achieve the specified diligence milestones within the specified periods, subject to our extension rights.
Government Regulations and Product Approvals
Government authorities in the United States, at the federal, state and local level, and in other countries extensively regulate, among other things, the research, development, testing, manufacture, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, import and export of pharmaceutical products such as those we are developing. The processes for obtaining regulatory approvals in the United States and in foreign countries, along with subsequent compliance with applicable statutes and regulations, require the expenditure of substantial time and financial resources.
FDA Approval Process
All of our current product candidates are subject to regulation in the United States by the FDA under the Federal Food, Drug, and Cosmetic Act (FDC Act) and its implementing regulations. Our lambda product candidate is additionally subject to regulation as a biologic under the Public Health Service Act. The FDA subjects drugs and biologics to extensive pre and post market regulation. Failure to comply with the FDC Act and other federal and state statutes and regulations may subject a company to a variety of administrative or judicial sanctions, such as FDA refusal to approve pending new drug applications (NDAs), BLAs, withdrawal of approvals, clinical holds, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties or criminal penalties.
FDA approval is required before any new biologic, drug or dosage form, including a new use of a previously approved product, can be marketed in the United States. The process required by the FDA before a new product may be marketed in the United States is long, expensive, and inherently uncertain. Product development in the United States typically involves completion of preclinical laboratory and animal tests, submission to the FDA of an IND application, which must become effective before clinical testing may commence, approval by an independent institutional review board (IRB) at each clinical site before each trial may be initiated, performance of adequate and well controlled clinical trials to establish the safety, efficacy, purity and/or potency of the product for each indication for which FDA approval is sought, submission to the FDA of an NDA or BLA, satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced, and FDA review and approval of the NDA or BLA. Developing the data to satisfy FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity, and novelty of the product, disease or indication.
Preclinical tests include laboratory evaluation of the product’s chemistry, formulation, and toxicity, as well as animal studies to characterize and assess the potential safety, efficacy, purity and/or potency of the product. The conduct of the preclinical tests must comply with federal regulations and requirements, including good laboratory practice (GLP) regulations. These preclinical results are submitted to the FDA as part of an IND along with other information, including information about the product’s chemistry, manufacturing and controls, and a proposed clinical trial protocol. Long term preclinical studies including reproductive toxicity and carcinogenicity may be initiated or continue after the IND is submitted.
An IND must become effective before United States clinical trials may begin. A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA has neither commented on nor questioned the IND within this 30-day period, the IND automatically becomes effective and the clinical trial proposed in the IND may begin. If the FDA raises any concerns or questions and places the clinical trial on a clinical hold, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, a submission of an IND may not result in FDA authorization to commence a clinical trial. A separate submission to an existing IND must also be made for each successive clinical trial conducted during product development.
Clinical trials involve the administration of the investigational new drug or biologic to human subjects under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations, including good clinical practice (GCP) requirements for conducting, monitoring, recording and reporting the results of clinical trials, in order to ensure that the data and results are scientifically credible and accurate and that the trial subjects are adequately informed of the potential risks of participating in clinical trials; and (ii) with protocols that detail, among other things, the objectives of the trial, the parameters to be used in monitoring safety, efficacy, purity and/or potency criteria to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
The FDA may order the temporary, or permanent, discontinuation of a clinical trial at any time, or impose other sanctions, if it believes that the clinical trial either is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. The study protocol and informed consent information for patients in clinical
trials must also be submitted to and approved by an IRB at each study site before the study commences at that site and the IRB must monitor the clinical trial until it is completed. An IRB may also require the clinical trial at that site to be halted, either temporarily or permanently, for failure to comply with the IRB’s requirements or if the product candidate has been associated with unexpected serious harm to patients, or the IRB may impose other conditions. The study sponsor or the FDA may also suspend or discontinue a clinical trial at any time on various grounds, including a determination that the subjects are being exposed to an unacceptable health risk.
Clinical trials to support an NDA or BLA for marketing approval are typically conducted in three sequential phases, although there is leeway to overlap or combine these phases.
•Phase 1. The product candidate is initially introduced into healthy human subjects or patients with the target disease or condition, and is tested to assess safety, dosage tolerance, pharmacokinetics and pharmacological activity, and, when possible, to ascertain evidence of efficacy. The product candidate may also be tested in patients with severe or life-threatening diseases to gain an early indication of its effectiveness.
•Phase 2. The trials are conducted using a limited patient population for the purposes of preliminarily determining the effectiveness of the product in that particular indication, ascertaining dosage tolerance, discerning the optimal dosage, and identifying possible adverse effects and safety risks.
•Phase 3. If a compound demonstrates evidence of efficacy and has an acceptable safety profile in the Phase 2 clinical trials, then Phase 3 clinical trials are undertaken to obtain additional information from an expanded and diverse patient population, at multiple, geographically dispersed clinical trial sites, in randomized controlled studies often with a double-blind design to maximize the reproducibility of the study results. Typically, a minimum of two positive Phase 3 clinical trials are submitted to support the product’s marketing application. These Phase 3 clinical trials are intended to provide sufficient data demonstrating evidence of the safety, efficacy, purity and potency of the product such that the FDA can evaluate the overall benefit-risk of the product and provide adequate information for the labeling and package insert for the product. Trials conducted outside of the United States under similar, GCP-compliant conditions in accordance with local applicable laws may also be acceptable to the FDA in support of product approval.
Sponsors of clinical trials for investigational products must publicly disclose certain clinical trial information, including detailed trial design. These requirements are subject to specific timelines and apply to most Phase 3 clinical trials of FDA-regulated products.
In some cases, the FDA may condition approval of an NDA or BLA for a product candidate on the sponsor’s agreement to conduct additional clinical trials after approval. In other cases, a sponsor may voluntarily conduct additional clinical trials post approval to gain more information about the product. Such post approval trials are typically referred to as Phase 4 clinical trials.
Progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and more frequently if serious adverse events occur. Additionally, some clinical trials are overseen by an independent group of qualified experts organized by the clinical trial sponsor, known as a data safety monitoring board or committee. This group provides authorization for whether or not a trial may move forward at designated check points based on access to certain data from the study. Phase 1, Phase 2, Phase 3 and Phase 4 clinical trials may not be completed successfully within any specified period, or at all.
Concurrent with clinical trials, companies usually finalize a process for manufacturing the product in commercial quantities in accordance with current good manufacturing practice (cGMP) requirements. The manufacturing process must be capable of consistently producing quality batches of the product candidate and, among other things, the manufacturer must develop methods for testing the identity, strength, quality and purity of the final product. Additionally, appropriate packaging must be selected and tested, and stability studies must be conducted to demonstrate that the product candidate does not undergo unacceptable deterioration over its shelf life.
After completion of the required clinical testing, an NDA or BLA is prepared and submitted to the FDA requesting approval to market the drug or biologic for one or more specified indications. FDA review and approval of the NDA or BLA is required before marketing of the product may begin in the United States. The NDA or BLA must include the results of all preclinical, clinical, and other testing, including negative or ambiguous results as well as positive findings, together with other detailed information including compilation of data relating to the product’s pharmacology, chemistry,
manufacture, and controls. The application must also contain extensive manufacturing information. The FDA reviews an NDA or BLA to determine, among other things, whether a product is safe and effective for its intended use. The cost of preparing and submitting an NDA or BLA is substantial. Under federal law, the submission of most NDAs and BLAs is subject to both a substantial application user fee and annual program user fees. These fees are typically increased annually.
The FDA has 60 days from its receipt of an NDA or BLA to determine whether the application will be accepted for filing based on the agency’s threshold determination that the application is sufficiently complete to permit substantive review. The FDA may request additional information rather than accept an application for filing. In this event, the application must be resubmitted with the additional information. Once the submission is accepted for filing, the FDA begins an in-depth review.
Under the Prescription Drug User Fee Act (PDUFA) guidelines that are currently in effect, the FDA has agreed to certain performance goals in the review of applications. Standard applications are generally reviewed within ten months of filing, or twelve months from submission. Although the FDA often meets its user fee performance goals, it can extend these timelines if necessary, and FDA review may not occur on a timely basis. The FDA usually refers applications for novel products, or products that present difficult questions of safety, efficacy, purity and/or potency, to an advisory committee—a panel of independent experts, typically including clinicians and other scientific experts—for review, evaluation, and a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendation of the advisory committee, but it generally follows its recommendations. Before approving an NDA or BLA, the FDA will typically inspect one, or more, clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the product is manufactured. The FDA will not approve an application unless it verifies that compliance with cGMP requirements is satisfactory and that the manufacturing processes and facilities are adequate to assure consistent production of the product within required specifications. The FDA will not approve a product unless the application contains data showing substantial evidence that it is safe and effective in the indication studied.
After the FDA evaluates the application and conducts its inspections, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies contained in the submission and may require substantial additional testing or information in order for the FDA to reconsider the application, including potentially significant, expensive and time-consuming requirements related to clinical trials, nonclinical studies or manufacturing. Even if such data are submitted, the FDA may ultimately decide that the NDA or BLA does not satisfy the criteria for approval. Data from clinical trials are not always conclusive, and the FDA may interpret data differently than we do. If and when those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the application, the FDA will typically issue an approval letter. The FDA has committed to reviewing such resubmissions in two or six months depending on the type of additional information requested. FDA approval is never guaranteed. The FDA may refuse to approve an application if applicable regulatory criteria are not satisfied.
An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications. The approval for a product may be significantly more limited than requested in the application, including limitations on the specific diseases and dosages or the indications for use, which could restrict the commercial value of the product. The FDA may also require that certain contraindications, warnings, or precautions be included in the product’s package insert, or labeling.
In addition, as a condition of approval, the FDA may require a risk evaluation and mitigation strategy (REMS) to help ensure that the benefits of the product outweigh the potential risks. REMS can include medication guidelines, communication plans for healthcare professionals, and elements to assure safe use (ETASU). ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, including dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS or use of a companion diagnostic with a product can materially affect the potential market and profitability of the product. Moreover, product approval may require, as a condition of approval, substantial post-approval testing and surveillance to monitor the product’s safety, efficacy, purity and/or potency. The FDA may also condition approval on, among other things, changes to proposed labeling or development of adequate controls and specifications.
Once granted, product approvals may be withdrawn if compliance with regulatory standards are not maintained or problems are identified following initial marketing. In addition, after approval, some types of changes to the approved product, such as adding new indications, manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval. There also are continuing, annual program user fees for any marketed products, as well as new application fees for supplemental applications with clinical data.
505(b)(2) NDA Pathway
As an alternative path to FDA approval, an applicant may submit an NDA under Section 505(b)(2) of the Food, Drug and Cosmetic Act. A Section 505(b)(2) NDA is an application that contains full reports of investigations of safety and effectiveness, but where at least some of the information required for approval comes from studies not conducted by, or for, the applicant and for which the applicant has not obtained a right of reference or use from the person by or for whom the investigations were conducted. This type of application permits reliance for such approvals on literature or on an FDA finding of safety, effectiveness or both for an approved drug product.
Orphan Drugs
Under the Orphan Drug Act, the FDA may grant an Orphan Drug designation to products intended to treat a rare disease or condition—generally one that affects fewer than 200,000 individuals in the United States. Orphan Drug designation must be requested before submitting the NDA or BLA. After the FDA grants Orphan Drug designation, the FDA publicly discloses the product’s identity and its intended orphan use. Orphan Drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. The first active moiety to be approved to treat a disease with FDA’s Orphan Drug designation is entitled to a seven-year period of marketing exclusivity in the United States for that product, for that indication. During the seven-year exclusivity period, the FDA may not approve any other applications to market the same product for the same orphan indication, regardless of patent status, except in limited circumstances, such as a showing of clinical superiority to the product with orphan exclusivity or if the FDA finds that the holder of the orphan exclusivity has not shown that it can assure the availability of sufficient quantities of the Orphan Drug to meet the needs of patients with the disease or condition for which the product was designated. Orphan Drug exclusivity does not prevent the FDA from approving a different chemical/biological entity for the same disease or condition. An Orphan Drug designation also does not preclude the same product from being developed for a different disease or condition. Among the other benefits of Orphan Drug designation are tax credits for certain research expenses and a waiver of the application user fee.
Rare Pediatric Disease (RPD) designation by the FDA enables priority review voucher (PRV) eligibility upon U.S. market approval of a designated product for rare pediatric diseases. The RPD-PRV program is intended to encourage development of therapies to prevent and treat rare pediatric diseases. The voucher, which is awarded upon NDA or BLA approval to the sponsor of a designated RPD can be sold or transferred to another entity and used by the holder to receive priority review for a future NDA or BLA submission, which reduces the FDA review time of such future submission from ten to six months.
Expedited Development and Review Programs
The FDA offers several expedited development and review programs for qualifying product candidates. The Fast Track program is intended to expedite or facilitate the process for reviewing new products that meet certain criteria. Specifically, new products are eligible for Fast Track designation if they are intended to treat a serious or life-threatening disease or condition and demonstrate the potential to address unmet medical needs for the disease or condition. Fast Track designation applies to the combination of the product and the specific indication for which it is being studied. The sponsor of a fast track product has opportunities for frequent interactions with the review team during product development and, once an NDA or BLA is submitted, the product may be eligible for priority review. A fast track product may also be eligible for rolling review, where the FDA may consider for review sections of the NDA or BLA on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the application, the FDA agrees to accept sections of the application and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the application.
A product intended to treat a serious or life-threatening disease or condition may also be eligible for Breakthrough Therapy designation to expedite its development and review. A product can receive Breakthrough Therapy designation if preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The designation includes all of the Fast Track program features, as well as more intensive FDA interaction and guidance beginning as early as Phase 1 and an organizational commitment to expedite the development and review of the product, including involvement of senior managers. PRIME designation by the EMA confers comparable benefits on qualifying drug product candidates.
Any marketing application for a biologic submitted to the FDA for approval, including a product with a Fast Track designation and/or Breakthrough Therapy designation, may be eligible for other types of FDA programs intended to
expedite the FDA review and approval process, such as priority review and accelerated approval. A product is eligible for priority review if it has the potential to provide a significant improvement in the treatment, diagnosis or prevention of a serious disease or condition compared to marketed products. For products containing new molecular entities, priority review designation means the FDA’s goal is to take action on the marketing application within six months of the 60-day filing date (compared with ten months under standard review). A product is eligible for accelerated approval if the product provides a meaningful therapeutic advantage to patients over available treatments for a serious or life threatening condition, based upon a determination that the product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit. The FDA may also grant accelerated approval for such product for such a condition when the product has an effect on an intermediate clinical endpoint that can be measured earlier than an effect on irreversible morbidity or mortality (IMM) and that is reasonably likely to predict an effect on IMM or other clinical benefit, taking into account the severity, rarity or prevalence of the condition and the availability or lack of alternative treatments. Products granted accelerated approval must meet the same statutory standards for safety, efficacy, purity and/or potency as those granted traditional approval and accelerated approval is typically contingent on a requirement to conduct additional post-approval confirmatory studies to verify and describe the product’s clinical benefit. The FDA may withdraw approval using expedited procedures if, among other things, the sponsor fails to conduct any required post-approval study with due diligence or a such study fails to verify and describe a clinical benefit.
Fast Track designation, Breakthrough Therapy designation, priority review, accelerated approval do not change the standards for approval but may expedite the development or approval process.
Advertising and Promotion
Products manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing post-approval regulatory requirements. For instance, the FDA closely regulates the post-approval marketing, labeling, advertising and promotion of products, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities and promotional activities involving the Internet. Failure to comply with these requirements can result in adverse publicity as well as significant penalties, including the issuance of warning letters directing a company to correct any deviations from the FDA’s standards. The FDA may also impose a requirement that future advertising and promotional materials be pre-cleared by the FDA, and the Company may face federal and/or state civil and criminal investigations and prosecutions.
Products may be marketed only for the approved indications and in accordance with the provisions of the approved labeling. Changes to some of the conditions established in an approved application, including changes in indications, labeling, or manufacturing processes or facilities, require submission and FDA approval of a new application or supplement before the change can be implemented. A supplement for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures and actions in reviewing supplements as it does in reviewing NDAs or BLAs. Obtaining new indication is an important part of managing the life cycle of the product.
Adverse Event Reporting and cGMP Compliance
Recordkeeping, adverse event reporting and the submission of periodic reports are required following the FDA’s approval of an NDA or BLA. The FDA also may require post-marketing testing or Phase 4 clinical trials, REMS, or surveillance to monitor the effects of an approved product. In addition, the FDA may place conditions on an approval that could restrict the distribution or use of the product. Furthermore, manufacture, packaging, labeling, storage and distribution procedures must continue to conform to cGMPs after approval. Manufacturers and certain of their subcontractors are required to register their establishments with the FDA and certain state agencies and are subject to periodic unannounced inspections by the FDA and certain state agencies to assess compliance with ongoing regulatory requirements, including cGMPs. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP requirements and impose reporting and documentation requirements upon the sponsor and any third-party manufacturers that the sponsor may decide to use. Accordingly, manufacturers must continue to expend time, money, and effort in the areas of production and quality control to maintain compliance with cGMPs. Failure to comply with the statutory and regulatory requirements can subject a manufacturer to possible legal or regulatory action, such as warning letters, suspension of manufacturing, seizure of product, injunctive action or possible civil penalties. We cannot be certain that we or our present or future third-party manufacturers or suppliers will be able to comply with the cGMP regulations and other ongoing FDA regulatory requirements. If we or our present or future third-party manufacturers or suppliers are not able to comply with these requirements, the FDA may halt our clinical trials, require us to recall a product from distribution or withdraw approval for that product. Regulatory authorities may also withdraw product approvals, request product recalls, or impose marketing
restrictions through labeling changes or product removals upon discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes.
Other Healthcare Laws and Compliance Requirements
In the United States, our activities are potentially subject to regulation by federal, state, and local authorities in addition to the FDA. These other agencies include, without limitation, the Centers for Medicare and Medicaid Services (CMS), the Office of Inspector General for the U.S. Department of Health and Human Services, other divisions of the U.S. Department of Health and Human Services, the U.S. Department of Justice and individual U.S. Attorney offices within the Department of Justice, as well as state and local governments. Such agencies enforce a variety of laws, including without limitation, anti-kickback and false claims laws, data privacy and security laws, coverage, reimbursement and drug price reporting laws, and physician payment transparency laws.
The federal Anti-Kickback Statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving any remuneration (including any kickback, bribe or rebate), directly or indirectly, overtly or covertly, to induce or in return for purchasing, leasing, ordering or arranging for or recommending the purchase, lease or order of any good, facility, item or service reimbursable, in whole or in part, under Medicare, Medicaid or other federal healthcare programs. The term “remuneration” has been broadly interpreted to include anything of value. The Anti-Kickback Statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on one hand and prescribers, purchasers and formulary managers, among others, on the other, including, for example, consulting/speaking arrangements, discount and rebate offers, grants, charitable contributions, and patient support offerings, among others. Although there are a number of statutory exceptions and regulatory safe harbors protecting some common activities from prosecution under the Anti-Kickback Statute, the exceptions and safe harbors are drawn narrowly. Practices that involve remuneration that may be alleged to be intended to induce prescribing, purchases or recommendations may be subject to scrutiny if they do not qualify for an exception or safe harbor. Failure to meet all of the requirements of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review of all of its facts and circumstances. Several courts have interpreted the statute’s intent requirement to mean that if any one purpose of an arrangement involving remuneration is to induce referrals of federal healthcare covered business, the Anti-Kickback Statute has been violated. Violations of the federal Anti-Kickback Statute are punishable by imprisonment for up to ten years and statutory fines of up to $100,000. Additional criminal fines can be imposed under federal U.S. criminal procedure laws. Civil penalties include statutory amounts of up to $100,000 (adjusted for inflation) per violation, assessments of up to three times the total payments between the parties to the arrangement, and exclusion from participation in the federal healthcare programs or suspension from future participation in Medicare and Medicaid. Further, violation of the federal Anti-Kickback Statute can also form the basis for False Claims Act liability (discussed below).
The federal civil False Claims Act prohibits, among other things, any person or entity from, among other things, knowingly presenting, or causing to be presented, a false or fraudulent claim for payment to or approval by the federal government or knowingly making, using or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government. A claim includes any request or demand for money or property presented to the U.S. government. Pharmaceutical and other healthcare companies have been prosecuted under these laws for, among other things, allegedly providing free product or other items or services of value to customers with the expectation that the customers would recommend the use of certain products or bill federal programs for such products. Companies have been prosecuted for causing false claims to be submitted because of, for example, the alleged marketing of products for unapproved, and thus non-reimbursable, uses, for the alleged submission of incorrect government price reporting data to certain Federal health care programs, for alleged violations of the federal Anti-Kickback Statute, and for other sales and marketing practices. Violations of the False Claims Act can result in civil penalties of up more than $25,000 per false claim or statement (an amount adjusted annually for inflation) plus three times the amount of damages sustained by the government. In addition, the civil monetary penalties statute imposes penalties against any person or entity that, among other things, is determined to have knowingly presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent.
The federal Health Insurance Portability and Accountability Act of 1996 (HIPAA) created new federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services.
Also, many states have similar fraud and abuse statutes or regulations that may be broader in scope and may apply regardless of payor, in addition to items and services reimbursed under Medicaid and other state programs.
We may be subject to data privacy and information security laws and regulations, including both foreign and domestic, in the locations in which we conduct our business. HIPAA and its respective implementing regulations imposes specific requirements relating to the privacy, security and transmission of individually identifiable health information and requires the adoption of administrative, physical and technical safeguards to protect such information. In addition, certain state laws govern the privacy and security of health information in certain circumstances, some of which are more stringent than HIPAA and many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
In addition, there has been a recent trend of increased federal and state regulation of payments made to physicians and other healthcare providers. The Physician Payments Sunshine Act imposes, among other things, annual reporting requirements for, among others, covered manufacturers of drugs, devices, biologics, and medical supplies for which payment is available under Medicare, Medicaid, or the Children’s Health Insurance Program, of certain payments and “transfers of value” provided to U.S.-licensed physicians (defined to include doctors, dentists, optometrists, podiatrists and licensed chiropractors), physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, certified nurse midwives and teaching hospitals, as well as tracking and reporting of ownership and investment interests held by U.S.-licensed physicians and their immediate family members. Covered manufacturers must submit reports by the 90th day of each calendar year.
Many states have similar laws and regulations that may differ from each other and federal law in significant ways, thus complicating compliance efforts. For example, states have anti-kickback and false claims laws that may be broader in scope than analogous federal laws and may apply regardless of payer. In addition, state data privacy laws that protect the security of health information may differ from each other and may not be preempted by federal law. In addition, certain states require implementation of commercial compliance programs and compliance with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, impose restrictions on marketing practices, and/or tracking and reporting of gifts, compensation and other remuneration or items of value provided to physicians and other healthcare professionals and entities. Further, a number of states have enacted legislation designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage or facilitate importation from other countries and bulk purchasing. Some states have also established prescription drug affordability boards that are tasked with identifying certain high-cost prescription products that may pose affordability challenges for consumers and payers, conducting cost reviews on such products, and, in some circumstances, imposing upper payment limits on such products. Moreover, several states and local jurisdictions require the registration of sales representatives. These laws may adversely affect our sales, marketing and other activities with respect to any product candidate for which we receive approval to market in the United States by imposing administrative and compliance burdens on us.
If our operations are found to be in violation of any of the healthcare regulatory laws described above or any other laws that apply to it, we may be subject to penalties, including potentially significant criminal, civil and/or administrative penalties, damages, fines, disgorgement, imprisonment, exclusion from participation in government healthcare programs, contractual damages, reputational harm, administrative burdens, diminished profits and future earnings, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
International Regulation
In addition to regulations in the United States, a variety of foreign regulations govern clinical trials, commercial sales, and distribution of products. Whether or not we obtain FDA approval for a product, we or our collaborators must obtain approval of the drug by the comparable regulatory authorities of foreign countries before commencing clinical trials or marketing of the product in those countries. The approval process varies from country to country and the time to approve may be longer or shorter than that required for FDA approval. Further, to the extent that any of our products are sold in a foreign country, we may be subject to additional foreign laws and regulations, which may include, for instance, applicable post-marketing requirements, including safety surveillance, anti-fraud and abuse laws and implementation of corporate compliance programs and reporting of payments or transfers of value to healthcare professionals.
Clinical Trials
In the EU, clinical trials of medicinal products must be conducted in accordance with EU and Member State regulations and the International Conference on Harmonization (ICH) guidelines on good clinical practice (GCP), as well as the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki. In the EU, the Regulation (EU) No 536/2014 (CTR) introduces a centralized application procedure, whereby a clinical trial sponsors seeking approval for a clinical trial in one or more Member States must submit an application to all Member States where the clinical study will be conducted, so-called Member State(s) concerned, through the Clinical Trials Information System (CTIS). A transitional period applies during the transition from the CTD to the CTR. During this period, the following applies: (i) until 30 January 2023, clinical trial sponsors could use CTIS to apply for a clinical trial under the CTR or could choose to apply to conduct a trial under the CTD; (ii) from 31 January 2023, clinical trial sponsors must comply with the CTR when they apply to start a new clinical trial in the EU or European Economic Area (EEA); and (iii) from 31 January 2025, all clinical trials approved under the CTD that continue running must comply with the CTR, and sponsors must have recorded all information in the CTIS. If the sponsor of the clinical trial is not established within the EU, it must appoint an entity within the EU to act as its legal representative. The sponsor must take out a clinical trial insurance policy, and in most Member States, the sponsor is liable to provide ‘no fault’ compensation to any study subject injured in the clinical trial. Any substantial changes to the trial protocol or other information submitted with the CTA must be notified to or approved by the relevant Member State and EC.
Marketing Authorization
To obtain regulatory approval of an investigational drug or biological product under EU regulatory systems, we must submit a marketing authorization application (MAA) either under the so-called centralized or national (mutual-recognition or decentralized) authorization procedures. A marketing authorization may be granted only to an applicant established in the EU. In the case of pediatric patients, Regulation (EC) No 1901/2006, as amended, provides that prior to obtaining a marketing authorization in the EU, applicants have to demonstrate compliance with all measures included in an EMA‑approved Pediatric Investigation Plan, or PIP, covering all subsets of the pediatric population, unless the EMA has granted (1) a product‑specific waiver, (2) a class waiver, or (3) a deferral for one or more of the measures included in the PIP. The centralized procedure provides for the grant of a single marketing authorization by the European Commission following a favorable opinion by the EMA that is valid in all EU Member states, as well as Iceland, Liechtenstein and Norway. The centralized procedure is compulsory for medicines produced by specified biotechnological processes, products designated as orphan medicinal products, and products with a new active substance indicated for the treatment of specified diseases, such as HIV/AIDS, cancer, diabetes, neurodegenerative disorders or autoimmune diseases, other immune dysfunctions and viral diseases. The centralized procedure is optional for other products that represent a significant therapeutic, scientific or technical innovation, or whose authorization would be in the interest of public health or which contain a new active substance for indications other than those specified to be compulsory. Before granting the MA, the EMA or the competent authorities of the Member State concerned make an assessment of the risk-benefit balance of the product on the basis of scientific criteria concerning its quality, safety and efficacy. A MA that has been granted in the EU may be suspended or withdrawn if ongoing regulatory requirements are not met or if safety problems are identified. Among other things, MA holders are required to have risk management plans that use risk minimization strategies beyond product labeling to ensure that the benefits of certain prescription drugs outweigh their risks.
Regulatory Data Protection
In the EU, all new active substances approved on the basis of a complete independent data package benefit from an 8+2+1-year data/market exclusivity regime. This regime consists of (i) a regulatory data protection (RDP) period and market exclusivity period of eight years, (ii) a market exclusivity period of an additional two years after the eight-year period, and (iii) an extended market exclusivity period of one year after the ten-year period if, during the first eight years of those ten years, the marketing authorization holder obtains an approval for one or more new therapeutic indications which, during the scientific evaluation prior to their approval, are determined to bring a significant clinical benefit in comparison with existing therapies. Third parties may reference the nonclinical and clinical data of the original innovator beginning eight years after notification of the grant of the approval in the EU, but the third party may market a generic version after only 10 or, where applicable, 11 years have lapsed from the notification of the grant of the approval.
Orphan Drug Designation
The EMA grants Orphan Drug designation to promote the development of products for the treatment, prevention or diagnosis of life-threatening or chronically debilitating conditions affecting not more than five in 10,000 people in the EU. In addition, Orphan Drug designation can be granted if the drug is intended for a life threatening or chronically debilitating
condition in the EU and without incentives it is unlikely that sales of the drug in the EU would be sufficient to justify the investment required to develop the drug. Orphan Drug designation is only available if there is no other satisfactory method approved in the EU of diagnosing, preventing or treating the condition, or if such a method exists, the proposed Orphan Drug will be of significant benefit to patients. Orphan Drug designation provides opportunities for free or reduced-fee protocol assistance, fee reductions for marketing authorization applications and other post-authorization activities and ten years of market exclusivity following drug approval, which can be extended to 12 years if trials are conducted in accordance with an agreed-upon pediatric investigational plan. The exclusivity period may be reduced to six years if the designation criteria are no longer met, including where it is shown that the product is sufficiently profitable not to justify maintenance of market exclusivity.
Accelerated Assessment Procedures
When an MAA is submitted in the EU in respect of medicinal product for human use which is of major interest from the point of view of public health and therapeutic innovation, the applicant may request an accelerated assessment procedure pursuant to Article 14 (9) of Regulation (EC) 726/2004, as amended. Based on the request, the justifications presented, and the recommendations of the rapporteurs, the Committee for Medicinal Products for Human Use (CHMP) will formulate a decision, without prejudice to the possible positive or negative CHMP opinion on the granting of a MA. If the CHMP accepts the request, the timeframe for the evaluation will be reduced from the standard 210 days for the centralized procedure to 150 days, but it is possible that the CHMP can revert to the standard time limit for the centralized procedure if it considers that it is no longer appropriate to conduct an accelerated assessment. The EMA may also grant eligibility to the Priority Medicines (PRIME) Scheme to support the development of promising medicines that target an unmet medical need, i.e., medicines that may offer a major therapeutic advantage over existing treatments, or benefit patients with no current treatment options for their disease. Through PRIME, the EMA grants proactive support to medicine developers to optimize the generation of robust data on a medicine’s benefits and risks, and enable accelerated assessment of medicines applications.
Conditional Marketing Authorization and Authorization under Exceptional Circumstances
Medicinal products may be eligible for a conditional marketing authorization. The provisions for the granting of such an authorization are further elaborated in Regulation (EC) No. 507/2006. The available data must indicate that the benefits outweigh the risks and the applicant must be in a position to provide the comprehensive clinical data in the future. A conditional marketing authorization is valid for one year, can be renewed annually and may be requested by an applicant or proposed by the EMA’s CHMP for medicinal products which aim at the treatment, prevention or medical diagnosis of seriously debilitating or life-threatening diseases, or that are intended to be used in emergency situations in response to public health threats recognized either by the World Health Organization or by the EU in the framework of Decision No. 1082/2013/EU, or that are designated as orphan medicinal products. The holder will be required to complete ongoing studies or to conduct new studies within a specified period of time with a view to confirming that the benefit-risk balance is positive. In addition, specific obligations may be imposed in relation to the collection of pharmacovigilance data.
Medicinal products may also be granted a marketing authorization in absence of comprehensive data under exceptional circumstances. Unlike a conditional marketing authorization, where marketing approval is granted in the likelihood that the sponsor will provide such data within an agreed timeframe, the authorization under exceptional circumstances may be granted when comprehensive data cannot be obtained even after authorization, because: (i) the indications for which the product in question is intended are encountered so rarely that the applicant cannot reasonably be expected to provide comprehensive evidence, or (ii) in the present state of scientific knowledge, comprehensive information cannot be provided, or (iii) it would be contrary to generally accepted principles of medical ethics to collect such information.
The authorization under exceptional circumstances is granted subject to a requirement for the applicant to meet certain conditions, in particular concerning the safety of the medicinal product, the notification of any incident relating to its use, and actions to be taken. The renewal of the marketing authorization under exceptional circumstances follows the same rules as a “normal” marketing authorization. After five years, the marketing authorization is renewed under exceptional circumstances for an unlimited period, unless the competent authority decides, on justified grounds relating to pharmacovigilance, to proceed with one additional five-year renewal.
EU Pharmaceutical Legislation Review
On 26 April 2023, the European Commission published the proposals to revise the existing EU legislation on pharmaceuticals (EU Pharma Law Review). The revision consists of two proposals, a new directive and a new regulation
(EU Pharma Law Proposal), that will amend and/or repeal and replace the relevant legislation concerning medicinal products for human use, including legislation concerning orphan medicinal products and medicinal products for paediatric use. The EU Pharma Law Review could have a significant impact on the RDP available for to innovative medicinal products in the EU. If adopted in current form, the EU Pharma Law Proposal would reduce the current baseline for data exclusivity from eight to six years, extensible under certain conditions. In addition, the EU Pharma Law Review could have a significant impact on the designation of and incentives offered to orphan medicinal products in the EU. If adopted in current form, the EU Pharma Law Proposal would introduce the possibility for the Commission, by way of delegated acts, to derogate from the current prevalence criterion, and introduce specific criteria for certain conditions, due to the characteristics of such conditions or other scientific reasons. The EU Pharma Law Proposal also proposes changes to the current orphan market exclusivity (OME) approach. If adopted in the current form, the EU Pharma Law Proposal would in most cases reduce the duration of the OME, and replace the current system with separate OME periods for each new indication, to a system with a single OME period for each active substance. Finally, the EU Pharma Law Review could have a significant impact on paediatric medicinal products. If adopted in the current form, the EU Pharma Law Proposal would introduce a new evolutionary PIP system for certain types of developments, allowing the submission of an initial high level PIP, to be subsequently completed at precise stages in the development. In addition, the EU Pharma Law Proposal could introduce the obligation to place medicinal products authorized for a paediatric indication on the market of all Member States where the medicinal product is already placed on the market. The EU Pharma Law Proposal is currently subject to debate amongst the EU legislator and significant changes to the Proposal are expected. A final act is not expected to be adopted before 2026.
Foreign data protection laws, including, without limitation, the EU’s General Data Protection Regulation (EU GDPR), the EU GDPR as transposed into the national laws of the United Kingdom by virtue of section 3 of the European Union (withdrawal) Act 2018 as the UK GDPR (the EU GDPR and the UK GDPR, together the GDPR), the UK Data Protection, 2018 and European Union (EU) Member State and English data protection legislation, also apply to health-related and other personal information that we process, including, without limitation, personal information relating to clinical trial participants in the EU and/or the UK. The GDPR imposes significant obligations on controllers and processors of personal information, including, among other things, to: (i) implement administrative, physical, technical, and organizational safeguards to protect personal information; (ii) establish an appropriate and valid legal basis for processing personal information; (iii) comply with accountability and transparency requirements regarding the processing of personal information, which require controllers to demonstrate and record compliance with the GDPR and to provide more detailed information to data subjects regarding processing; (iv) comply with data protection rights of data subjects including a right of access to and rectification of personal information a right to obtain restriction of processing or to object to processing of personal information, a right to ask for a copy of personal information to be provided to a third party in a useable format and erasing personal information in certain circumstances; (v) report certain personal data breaches to the relevant supervisory authority without undue delay (and no later than 72, where feasible); (vi), obtain explicit consent for collection of sensitive personal information such as health data; and (vii) consider data protection as any new products or services are developed and to limit the amount of personal information processed. In addition, the GDPR prohibits the international transfer of personal information outside of the EU and/or the UK including to the U.S., unless made to a country deemed to have adequate data privacy laws by the European Commission and/or the UK or a data transfer mechanism in accordance with the GDPR (as applicable) has been put in place.
The GDPR allows EU Member States and the UK (as applicable) to make additional laws and regulations further limiting the processing of personal information, including genetic, biometric and health data.
The GDPR also introduces fines of up to €20 million (under the EU GDPR) or £17.5 million (under the UK GDPR) or up to 4% of the annual global revenue of the non-compliant company, whichever is greater, for serious violations of certain of the GDPR’s requirements. The GDPR identifies a list of points to consider when determining the level of fines to impose (including the nature, gravity and duration of the infringement). Data subjects also have a right to compensation for financial or non-financial losses (e.g., distress). Complying with the GDPR may cause us to incur substantial operational and compliance costs or require us to change our business practices. Despite our efforts to bring practices into compliance with the GDPR, we may not be successful either due to internal or external factors such as resource allocation limitations or a lack of vendor cooperation. Non-compliance could result in proceedings against us by governmental entities, regulators, customers, data subjects, suppliers, vendors or other parties. Further, there is a risk that the measures will not be implemented correctly or that individuals within the business will not be fully compliant with the new procedures. If there are breaches of these measures, we could face significant administrative and monetary sanctions as well as reputational damage which may have a material adverse effect on our operations, financial condition and prospects.
Pharmaceutical Coverage, Pricing and Reimbursement
In the United States and many other countries, patients who are prescribed treatments for their conditions and providers performing the prescribed services generally rely on third-party payors to reimburse all or part of the associated healthcare costs. Patients are unlikely to use our products unless coverage is provided, and reimbursement is adequate to cover a significant portion of the cost of our products. Sales of any products for which we receive regulatory approval for commercial sale will therefore depend in part on the availability of reimbursement from third-party payors, including government health administrative authorities, managed care providers, private health insurers, and other organizations.
The process for determining whether a third-party payor will provide coverage for a drug product typically is separate from the process for setting the price of a drug product or for establishing the reimbursement rate that the payor will pay for the drug product once coverage is approved. Third-party payors may limit coverage to specific drug products on an approved list, also known as a formulary, which might not include all of the FDA-approved drugs for a particular indication. A decision by a third-party payor not to cover Zokinvy or any of our product candidates once approved could reduce physician utilization of such products and have a material adverse effect on our sales and financial condition. Moreover, a third-party payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be made available. Adequate third-party reimbursement may not be made available to enable achieving our sales forecast. Additionally, coverage and reimbursement for drug products can differ significantly from payor to payor. One third-party payor’s decision to cover a particular medical product or service does not ensure that other payors will also provide coverage for the medical product or service or will provide coverage at an adequate reimbursement rate. As a result, the coverage determination process will require us to provide scientific and clinical support for the use of our products to each payor separately and will be a time-consuming process.
We expect that 50% to 70% of US patients who are eligible for treatment with Zokinvy will be covered by insurance through a federal healthcare program such as Medicaid, and the remaining patients will be covered by commercial insurance. We have had active engagement with payers that cover the lives of identified patients with Progeria and processing-deficient progeroid laminopathies.
The containment of healthcare costs has become a priority of federal, state and foreign governments, and the prices of drugs have been a focus of this effort. Third-party payors are increasingly challenging the prices charged for therapeutics, medical products and services, examining the medical necessity and reviewing the cost-effectiveness of drug products and medical services, in addition to questioning safety and efficacy. If these third-party payors do not consider our products to be cost-effective compared to other available therapies, they may not cover our products post-FDA approval or, if they do, the level of payment may be insufficient to allow us to achieve our sales forecast. Additionally, coverage policies and third‑party reimbursement rates may change at any time. Therefore, even if favorable coverage and reimbursement status is attained for one or more of our products, less favorable coverage policies and reimbursement rates may be implemented in the future.
In addition, the U.S. government, state legislatures and foreign governments have continued implementing cost-containment programs, including price controls, price transparency measures, inflation rebates, drug price negotiation programs for certain products, and restrictions on coverage and reimbursement and requirements for substitution of generic products. We expect that additional state and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that third-party payors will pay for healthcare products and services, which could result in reduced demand for our products once approved or additional pricing pressures.
Healthcare Reform
In the United States, there have been and continue to be a number of legislative initiatives to reform the delivery and payment for therapeutics and medical services. For example, in March 2010, the Patient Protection and Affordable Care Act, as amended by the Affordable Care Act (collectively, the “ACA”) was passed, which substantially changed the way health care is financed by both governmental and private insurers, and significantly impacts the U.S. pharmaceutical industry. This law was designed to expand access to health insurance coverage for uninsured and underinsured individuals while containing overall healthcare costs. The framework of the ACA and other healthcare reforms continues to evolve as a result of executive, legislative, regulatory, and administrative developments. For example, Congress has enacted several laws that modify certain provisions of the ACA, such as removing penalties, starting January 1, 2019, for not complying with the ACA’s individual mandate to carry health insurance and delaying the implementation of certain ACA-mandated fees. In addition, the 2020 federal spending package permanently eliminated, effective January 1, 2020, the ACA-mandated “Cadillac” tax on high-cost employer-sponsored health coverage and medical device tax and, effective January 1, 2021, the health insurer tax. The American Rescue Plan Act of 2021 temporarily increased premium tax credit assistance established
under the ACA to help eligible individuals cover premiums for health insurance purchased through the health insurance marketplace and removed the 400% federal poverty level limit that otherwise applies for purposes of eligibility to receive premium tax credits. The Inflation Reduction Act of 2022 extended this increased tax credit assistance and removal of the 400% federal poverty limit through 2025. On January 28, 2021, President Biden issued an executive order instructing certain governmental agencies to review and reconsider their existing policies and rules that limit access to healthcare, including among others, reexamining Medicaid demonstration projects and waiver programs that include work requirements, and policies that create unnecessary barriers to obtaining access to health insurance coverage through Medicaid or the ACA. The ACA has also faced various judicial challenges, and on June 17, 2021, the U.S. Supreme Court dismissed a challenge to the ACA brought by several states without specifically ruling on the constitutionality of the ACA. It is unclear how any future healthcare reform measures of the Biden administration or other efforts, if any, to repeal, amend or replace the ACA will impact the ACA, and our business.
Also, there has been heightened governmental scrutiny recently over pharmaceutical pricing practices in light of the rising cost of prescription drugs and biologics. Such scrutiny has resulted in Congressional and federal agency inquiries regarding pricing and related practices, as well as proposed and enacted federal and state legislation and regulation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, impose pricing controls on prescription products, and reform government program reimbursement methodologies for products. For example, the American Rescue Plan Act of 2021 included among its provisions a sunset of the ACA’s cap on pharmaceutical manufacturers’ rebate liability under the Medicaid Drug Rebate Program. Previously, under the ACA, manufacturers’ rebate liability was capped at 100% of the average manufacturer price for a covered outpatient drug. Effective January 1, 2024, manufacturers’ Medicaid Drug Rebate Program rebate liability is no longer capped, potentially resulting in a manufacturer paying more in Medicaid Drug Rebate Program rebates than it receives on the sale of certain covered outpatient drugs.
Additionally, on July 9, 2021, President Biden issued an Executive Order to promote competition in the U.S. economy that included several initiatives addressing prescription drugs. Among other provisions, the Executive Order stated that the Biden administration will “support aggressive legislative reforms that would lower prescription drug prices, including by allowing Medicare to negotiate drug prices, by imposing inflation caps, and through other related reforms.” In response to the Executive Order, on September 9, 2021, HHS issued a Comprehensive Plan for Addressing High Drug Prices that identified potential legislative policies and administrative tools that Congress and the agency can pursue in order to make drug prices more affordable and equitable, improve and promote competition throughout the prescription drug industry, and foster scientific innovation. In August 2022, President Biden signed into law the Inflation Reduction Act of 2022, which implements substantial changes to the Medicare program, including drug pricing reforms and changes to the Medicare Part D benefit design. Among other reforms, the Inflation Reduction Act of 2022 imposes inflation rebates on drug manufacturers for products reimbursed under Medicare Parts B and D if the prices of those products increase faster than inflation; implements changes to the Medicare Part D benefit that, beginning in 2025, will cap benefit annual out-of-pocket spending at $2,000, while imposing new discount obligations for pharmaceutical manufacturers; and, beginning in 2026, establishes a “maximum fair price” for a fixed number of pharmaceutical and biological products covered under Medicare Parts B and D following a price negotiation process with CMS. Since its enactment, CMS has taken steps to implement various drug pricing provisions of the IRA. This includes, without limitation, issuing new guidance on June 30, 2023 detailing the requirements and parameters of the first round of price negotiations, to take place during 2023 and 2024, for products subject to the “maximum fair price” provision that would become effective in 2026; on August 29, 2023, releasing the initial list of 10 drugs subject to price negotiations; and on December 14, 2023 releasing a list of 48 Medicare Part B products that had adjusted coinsurance rates based on the inflationary rebate provisions of the IRA for the time period of January 1, 2024 to March 31, 2024. Several pharmaceutical manufacturers and other industry stakeholders have challenged the law, including through lawsuits brought against HHS, the Secretary of HHS, CMS, and the CMS Administrator challenging the constitutionality and administrative implementation of the IRA’s drug price negotiation provisions. We cannot predict whether the IRA, or any of its component parts, will be overturned, repealed, replaced, or amended nor can we predict the likelihood, nature, or extent of other health reform initiatives that may arise from future legislation, administrative, or other action. However, we expect these initiatives to increase pressure on drug pricing.
The Biden administration continues to direct HHS to consider new healthcare payment and delivery models that would lower drug costs and promote access to innovative therapies for beneficiaries enrolled in the Medicare and Medicaid programs. In addition, at the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. Policy changes, including potential modification or repeal of all or parts of the ACA or the implementation of new health care legislation, could result in
significant changes to the health care system, which may prevent us from being able to generate revenue, attain profitability or commercialize our drugs. We expect that additional state and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in reduced demand or lower pricing for our product candidates, or additional pricing pressures.
In the United States, and other potentially significant markets for our product candidates, government authorities and third-party payors are increasingly attempting to limit or regulate the price of therapeutics and medical services, particularly for new and innovative products and therapies, which has resulted in lower average selling prices. For example, in the United States, there have been several Congressional inquiries and proposed bills designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drugs. Furthermore, the increased emphasis on managed healthcare in the United States and on country and regional pricing and reimbursement controls in the EU will put additional pressure on product pricing, reimbursement and usage, which may adversely affect our future product sales and results of operations. These pressures can arise from rules and practices of managed care groups, judicial decisions and governmental laws and regulations related to Medicare, Medicaid and healthcare reform, pharmaceutical reimbursement policies and pricing in general.
Different pricing and reimbursement schemes exist in other countries. In the EU, governments influence the price of pharmaceutical products through their pricing and reimbursement rules and control of national health care systems that fund a large part of the cost of those products to consumers. Some jurisdictions operate positive and negative list systems under which products can only be effectively marketed once a reimbursement price has been agreed. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials in order to compare the cost effectiveness of a particular product candidate to currently available therapies. Other member states may allow companies to fix their own prices for medicines but monitor and control company profits. The downward pressure on health care costs has become intense. As a result, increasingly high barriers are being erected to the entry of new products. In addition, in some countries, cross- border imports from low-priced markets exert a commercial pressure on pricing within a country.
In December 2021, Regulation (EU) 2021/2282 on HTA, or HTA Regulation, was adopted. The HTA Regulation will apply as from 12 January 2025. It particularly replaces the current system based on the voluntary network of national authorities, and the new framework covers joint clinical assessments, joint scientific consultations, the identification of emerging health technologies, and voluntary cooperation for the national authorities. The HTA Regulation aims to provide a transparent and inclusive framework for health technology assessments in the EU, and it will help EU countries determine the effectiveness and value of new technologies and decide on pricing and reimbursement by health insurers or health systems.
Research and Development Expenses
Our research and development expenses were $62.3 million and $75.3 million for the years ended December 31, 2023 and 2022, respectively.
Employees and Human Capital
As of December 31, 2023, we had a total of 25 full-time employees in the United States and Europe, 15 of whom were primarily engaged in manufacturing and research and development activities, and 10 of whom were engaged in general management and administration. 6 of our employees had either an M.D. or a Ph.D. None of our employees are represented by a labor union or subject to a collective bargaining agreement. We have never experienced any work stoppage and consider our relations with our employees to be good.
Our human capital objectives include, as applicable, identifying, recruiting, retaining, incentivizing and integrating our existing and additional employees. The principal purposes of our equity incentive plans are to attract, retain and motivate talented and diverse employees, consultants and directors through the granting of stock-based compensation awards and cash-based performance bonus awards.
Corporate Information
We were originally incorporated in California in December 2000 as Celladon Corporation (Celladon). In April 2012, Celladon reincorporated in Delaware and had its initial public offering in February of 2014. On March 22, 2016, Eiger
BioPharmaceuticals, Inc. (Private Eiger) completed its merger (Merger) with Celladon in accordance with the terms of the Merger Agreement. Pursuant to the Merger Agreement, Celladon Merger Sub, Inc., a wholly-owned subsidiary of Celladon (Merger Sub) merged with and into Private Eiger, with Private Eiger becoming a wholly-owned subsidiary of Celladon and the surviving corporation of the Merger. Immediately following the Merger, Celladon changed its name to Eiger BioPharmaceuticals, Inc. In connection with the Merger, our common stock began trading on The Nasdaq Global Market with the ticker symbol EIGR on March 23, 2016. Our principal executive offices are located at 2155 Park Blvd in Palo Alto, California 94306, and our telephone number is 650-272-6138. Our corporate website address is www.eigerbio.com. The contents of our website are not incorporated into this Annual Report on Form 10-K and our reference to the URL for our website is intended to be an inactive textual reference only.
This Annual Report on Form 10-K contains references to our trademarks and to trademarks belonging to other entities. Solely for convenience, trademarks and trade names referred to in this Annual Report on Form 10-K, including logos, artwork and other visual displays, may appear without the ® or TM symbols, but such references are not intended to indicate, in any way, that we will not assert, to the fullest extent under applicable law, our rights or the rights of the applicable licensor to these trademarks and trade names. We do not intend our use or display of other companies’ trade names or trademarks to imply a relationship with, or endorsement or sponsorship of us by, any other company.
ITEM 1A. Risk Factors
You should carefully consider the following risk factors, as well as the other information in this report and in our other public filings. The occurrence of any of the following risks could harm our business, financial condition, results of operations and/or growth prospects or cause our actual results to differ materially from those contained in forward-looking statements we have made in this report and those we may make from time to time. You should consider all of the risk factors described when evaluating our business.
Risk Factor Summary
Below is a summary of material factors that make an investment in our common stock speculative or risky. Importantly, this summary does not address all of the risks and uncertainties that we face. Additional discussion of the risks and uncertainties summarized in this risk factor summary, as well as other risks and uncertainties that we face, can be found under “Risk Factors” in Part I, Item 1A of this Annual Report. The below summary is qualified in its entirety by that more complete discussion of such risks and uncertainties. You should carefully consider the risks and uncertainties described under “Risk Factors” in Part I, Item 1A of this Annual Report as part of your evaluation of an investment in our common stock.
•We are a commercial-stage biopharmaceutical company with a limited operating history. We have incurred net losses each year since our inception. We have one U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA) and Japanese Ministry of Health, Labour and Welfare-approved product for commercial sale, Zokinvy, and prior to 2021, have never generated any product revenue and may never be profitable.
•We are subject to risks and uncertainties associated with the Chapter 11 Cases.
•We cannot give any assurance that we will generate data for our current lead product candidate avexitide or any other candidate sufficient to receive regulatory approval, and without regulatory approval we will not be able to market such products.
•Prior to the approval of our NDA for Zokinvy to reduce the risk of mortality in HGPS, and for treatment of processing-deficient PL with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, we had not submitted an application for approval or obtained FDA approval for any product. We may not be able to obtain FDA approval of any future NDA or Biologics License Application (BLA) for our product candidates, which would prohibit commercialization.
•Our business strategy is based upon obtaining and maintaining Orphan Drug designation for our product candidates. If we are unable to obtain or maintain Orphan Drug designation or regulatory approval, our business would be substantially harmed.
•Our future success depends in part on our ability to attract, retain, and motivate qualified personnel.
•Product development involves a lengthy and expensive process with an uncertain outcome, and results of earlier studies may not be predictive of future study results.
•We rely on clinical studies of our product candidates in order to obtain regulatory approval. We may find it difficult to enroll patients in our clinical studies given the limited number of patients who have the diseases for which our product candidates are being studied.
•If clinical studies of our product candidates fail to demonstrate safety, efficacy, purity and/or potency to the satisfaction of the FDA or similar regulatory authorities outside the United States or do not otherwise produce positive results, we may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our product candidates.
•We rely on third parties to conduct our clinical studies, manufacture our product candidates and perform other services. Our ability to obtain regulatory approval or commercialize our product candidates and our business could be impaired if these collaborations are unsuccessful.
•If the market opportunities for our product candidates are smaller than we believe they are, we may not meet our revenue expectations. Because the patient populations in the market for our product candidates may be small, we must be able to successfully identify patients and acquire a significant market share to achieve profitability and growth.
•We face intense competition and rapid technological change and the possibility that our competitors may develop therapies that are similar, more advanced or more effective than ours.
•We currently have limited marketing and sales capabilities for the commercialization of our product candidates.
•Zokinvy sales reflect utilization of the product in the ultra-rare conditions of Hutchinson-Gilford progeria syndrome (HGPS) and processing-deficient progeroid laminopathies (PL) among a small number of patients, and thus sales volume may fluctuate over time due to the inherent mortality in these serious diseases.
•The commercial success of any of our current or future product candidates will depend upon the degree of market acceptance by physicians, patients, third-party payors and others in the medical community. Sales of our products depend substantially on the extent to which the costs of our product candidates will be paid for or reimbursed by healthcare management organizations or government authorities or third-party payors.
•We are currently conducting and will continue to conduct clinical trials for our product candidates outside the United States, which could expose us to risks that could have a material adverse effect on our business, including conflicts in the Middle East and risks in connection with the actions taken by the Russian Federation in Ukraine and surrounding areas.
•We intend to rely on a combination of exclusivity from Orphan Drug designation and our patent rights for our product candidates. If we are unable to maintain exclusivity from the combination of these approaches, then our ability to compete effectively in our markets may be harmed.
•The annual reassessment by the EMA of the risk-benefit balance for Zokinvy including information on the safe and effective use may not be positive, which could lead to a variation, suspension, revocation of our marketing authorization or requirement to fulfil additional specific obligations.
•If we are unable to maintain effective proprietary rights for our product candidates, or if the scope of the intellectual property protection is not sufficiently broad, our competitors could develop and commercialize product candidates similar or identical to ours.
•We may be unsuccessful in obtaining or maintaining necessary rights to our product candidates through acquisitions and in-licenses. If we fail to comply with obligations in the agreements under which we license intellectual property and other rights from third parties or otherwise experience disruptions to our business relationships with our licensors, we could lose license rights that are important to our business.
•We may not be successful in any efforts to identify, license, discover, develop or commercialize additional product candidates.
•Healthcare legislative reform measures may have a material adverse effect on our business and results of operations. If we fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs.
•The proposals to revise the European legislation on pharmaceuticals lead to uncertainties over the regulatory framework that will be applicable to medicinal products in the EU, including orphan medicinal products.
•We face potential product liability, and, if successful claims are brought against us, we may incur substantial liability and costs.
•We are dependent upon information technology systems and any failure or security breach of such systems could result in a material disruption in the development of our product candidates or other business operations as well as result in statutory or contractual obligations or otherwise expose us to liability.
•As a result of the Chapter 11 Cases, our common stock will be delisted from Nasdaq at the opening of business on April 11, 2024, which could have a material adverse effect on the liquidity of our common stock and our stock price.
Risks Related to our Financial Condition, Integration and Capital Requirements
We have incurred losses since our inception, have a limited operating history on which to assess our business, and anticipate that we will continue to incur significant losses for the foreseeable future. We will require substantial additional capital to achieve our goals.
We are a commercial-stage biopharmaceutical company with a limited operating history. We have incurred net losses in each year since our inception. For the years ended December 31, 2023 and 2022 we reported a net loss of $75.0 million and $96.8 million, respectively. As of December 31, 2023, we had an accumulated deficit of $512.2 million. Our prior losses, combined with expected future losses, have had and may continue to have an adverse effect on our stockholders’ (deficit) equity and working capital. Based on our recurring losses from operations incurred since inception, expectation of continuing operating losses and negative cash flows for the foreseeable future, and need to raise additional capital to finance our future operations, management has concluded that there is substantial doubt regarding Eiger's ability to continue as a going concern beyond twelve months after the date that our consolidated financial statements are issued, which was a principal reason for our decision to seek bankruptcy protection under Chapter 11. In addition, we may not be able to access a portion of our existing cash, cash equivalents and short-term securities due to market and other conditions. For example, on March 10, 2023, the Federal Deposit Insurance Corporation (FDIC) was appointed receiver of Silicon Valley Bank (SVB), where we maintain an operating account with a balance that exceeded the FDIC insurance limit. On March 12, 2023, the U.S. Treasury Department, the Federal Reserve and the FDIC jointly announced enabling actions that fully protect all depositors of SVB and that such depositors would have access to all of their funds starting March 13, 2023. On March 26, 2023, the FDIC announced that First Citizens BancShares would acquire the commercial banking business of SVB. Although the cash in our accounts at Silicon Valley Bank did not impact our operations, we cannot be certain that future market corrections or other conditions will not impact our ability to access our cash, cash equivalents and short-term securities. We continue to maintain an operating account at SVB and have begun measures to reduce our deposit concentration risk. If other banks and financial institutions enter receivership or become insolvent in the future in response to financial conditions affecting the banking system and financial markets, our ability to access our existing cash, cash equivalents and short-term securities may be threatened.
In addition, we will continue to require substantial additional capital to continue our clinical development, manufacturing and regulatory approval efforts and potential commercialization activities. Accordingly, we will need to raise substantial additional capital to continue to fund our operations. The amounts and timing of our future funding requirements will depend on many factors, including our ability to achieve regulatory approval and the pace and results of our clinical development efforts. While we are actively seeking additional capital to fund losses from operations and capital funding needs through public or private equity offerings and/or debt financings, through potential additional collaborations or strategic partnerships with other companies, or through a combination of the foregoing, additional funds may not be available when we need them on terms that are favorable to us, or at all. Failure to raise capital as and when needed, on favorable terms or at all, would have a negative impact on our financial condition and our ability to develop our product candidates.
We have devoted substantially all of our financial resources to identify, acquire, and develop our product candidates, including manufacturing of clinical supplies, conducting clinical studies and providing selling, general and administrative support for our operations. To date, we have financed our operations primarily through the sale of equity securities and debt facilities. The amount of our future net losses will depend, in part, on the rate of our future expenditures and our ability to obtain funding through equity or debt financings, strategic collaborations, or grants. Biopharmaceutical product development is a highly speculative undertaking and involves a substantial degree of risk. We expect losses to increase as we advance our clinical development programs in various clinical studies. We may need significant additional resources in order to aggressively move avexitide and our other product candidates forward successfully based on the discussions with the FDA. It may be several years, if ever, before we complete pivotal clinical studies and have additional product candidates approved for commercialization. We expect to invest significant funds into our clinical candidates to advance these compounds to potential regulatory approval.
If we obtain regulatory approval to market one or more additional product candidates, our future revenue will depend upon the size of any markets in which our product candidates may receive approval, and our ability to achieve sufficient market acceptance, pricing, reimbursement from third-party payors, and adequate market share for our product candidates in those markets. Even if we obtain adequate market share for our product candidates, because the potential markets in which our product candidates may ultimately receive regulatory approval could be very small, we may never become profitable despite obtaining such market share and acceptance of our products. We have also agreed with The Progeria Research Foundation to make Zokinvy available to progeria (HGPS and processing-deficient PL) patients under an expanded access program that may not result in payment to us. Future clinical trials of new therapies for progeria conducted by third parties may also result in patients converting from commercially reimbursed Zokinvy to product provided through clinical trials and result in lower revenues received by us.
We expect to continue to incur significant expenses and increasing operating losses for the foreseeable future and our expenses will increase substantially if and as we:
•pursue the clinical development of our product candidates;
•undertake the scale up and commercial manufacturing or have manufactured our product candidates;
•advance our programs into larger, more expensive clinical studies;
•initiate additional nonclinical, clinical, or other studies for our product candidates;
•commercialize and provide expanded access to Zokinvy for the treatment of HGPS and processing-deficient PL;
•identify and develop potential commercial opportunities, such as avexitide for PBH and HI;
•seek regulatory and marketing approvals and reimbursement for our product candidate;
•establish a sales, marketing, and distribution infrastructure to commercialize any products for which we may obtain marketing approval and market ourselves;
•make milestone, royalty or other payments under third-party license agreements;
•develop and educate PBH and HI markets;
•seek to maintain, protect, and expand our intellectual property portfolio;
•seek to attract and retain skilled personnel; and
•create additional infrastructure to support our operations as a public company and our product development and planned future commercialization efforts; and experience any delays or encounter issues with the development and potential for regulatory approval of our clinical candidates such as safety issues, clinical trial accrual delays, longer follow-up for planned studies, additional major studies, or supportive studies necessary to support marketing approval.
Further, the net losses we incur may fluctuate significantly from quarter to quarter and year to year, such that a period-to-period comparison of our results of operations may not be a representative indication of our future performance.
We are subject to risks and uncertainties associated with the Chapter 11 Cases.
The Chapter 11 Cases could have a material adverse effect on the Company’s business, financial condition, results of operations and cash flows. So long as the Chapter 11 Cases continue, our senior management may be required to spend a significant amount of time and effort dealing with bankruptcy proceedings instead of focusing on our business operations. The bankruptcy proceedings also may make it more difficult to retain management and the key personnel necessary to the success and growth of our business. In addition, during the period of time we are involved in the Chapter 11 Cases, our customers and suppliers may lose confidence in our ability to restructure our business and may seek to establish alternative commercial relationships.
Other significant risks associated with the Chapter 11 Cases that could have a material adverse effect on our business, financial condition, results of operations and cash flows include or relate to the following, among others:
•our ability to obtain approval from the Bankruptcy Court with respect to motions or other requests made to the Bankruptcy Court in the Chapter 11 Cases;
•our ability to confirm and consummate the plan of reorganization;
•the effects of the filing of the Chapter 11 Cases on our business and the interests of various constituents, including our shareholders;
•the high costs of the Chapter 11 Cases and related fees;
•our ability to maintain relationships with payors, suppliers, employees, and other third parties as a result of the Chapter 11 Cases;
•Bankruptcy Court rulings in the Chapter 11 Cases as well as the outcome of other pending litigation and the outcome of the Chapter 11 Cases in general;
•the length of time that we will operate with Chapter 11 protection;
•the sufficiency of the Company's cash on hand and ability to obtain sufficient financing to allow us to conduct our business during the pendency of the Chapter 11 Cases, confirm a plan of reorganization, or sell some or all of our assets during the bankruptcy proceedings in orderly fashion;
•third-party motions in the Chapter 11 Cases, which may interfere with our ability to confirm and consummate the plan of reorganization or another restructuring transaction, including motions by third parties that may force us into Chapter 7 proceedings under the Bankruptcy Code, rather than Chapter 11 proceedings; and
•our ability to continue as a going concern.
Because of the risks and uncertainties associated with the Chapter 11 Cases, we may not be able to accurately predict or quantify the ultimate impact the Chapter 11 Cases may have on our business, financial condition, results of operations and cash flows, nor can we accurately predict the ultimate impact the Chapter 11 Cases may have on our corporate or capital structure.
Prior to 2021, we never generated any product revenue and may never be profitable.
We have one product approved for commercialization in the U.S., EU, and Japan for two ultra-rare diseases. Zokinvy works to (i) reduce the risk of mortality in HGPS, and (ii) treat processing-deficient PL with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations. Zokinvy was approved by the FDA in November 2020 and launched commercially in the U.S. in January 2021. In July 2022, our MAA for Zokinvy was approved by the EC, under the exceptional circumstances procedure. The MA is subject to the EMA’s continued review on an annual basis of new efficacy and safety information which may become available. Our ability to generate substantial revenue and achieve profitability depends on our ability to (i) obtain the regulatory and marketing approvals necessary to commercialize Zokinvy in foreign jurisdictions, alone or with strategic collaboration partners, and (ii) to successfully complete the development of, and obtain the regulatory and marketing approvals necessary to commercialize, more of our product candidates in the U.S. or foreign jurisdictions. We do not anticipate generating significant product revenue for the foreseeable future. Our ability to generate future product revenue depends heavily on our success in many areas, including, but not limited to:
•completing research and development of our product candidates;
•obtaining additional and maintaining current regulatory and marketing approvals for our product candidates;
•manufacturing product candidates and establishing and maintaining supply and manufacturing relationships with third parties that meet regulatory requirements and our supply needs in sufficient quantities to meet market demand for our product candidates, if approved;
•marketing, launching and commercializing product candidates for which we obtain regulatory and marketing approval, either directly or with a collaborator or distributor;
•gaining market acceptance of our product candidates as treatment options;
•addressing any competing products;
•protecting and enforcing our intellectual property rights, including patents, trade secrets, and know-how;
•negotiating favorable terms in and maintaining any collaboration, licensing, or other arrangements into which we may enter;
•obtaining reimbursement or pricing for our product candidates that supports profitability; and
•attracting, hiring, and retaining qualified personnel.
Even if we obtain additional product approvals for commercial sale, we anticipate incurring significant costs associated with commercializing any approved product candidate. Our current portfolio of product candidates has been in-licensed from third parties and we will have to develop or acquire manufacturing capabilities in order to continue development and potential commercialization of our product candidates. Additionally, if we are not able to generate sufficient revenue from the sale of any approved products, we may never become profitable.
Raising additional capital may cause dilution to our stockholders, restrict our operations or require us to relinquish rights.
To the extent that we raise additional capital through the sale of equity, debt or other securities convertible into equity, stockholder ownership interest will be diluted, and the terms of these new securities may include liquidation or other preferences that adversely affect your rights as a common stockholder such as the Loan and Security Agreement we entered into with Innovatus Life Sciences Lending Fund I, LP (Innovatus) in June 2022 (the Innovatus Loan). The Innovatus Loan was a $75.0 million debt financing arrangement with Innovatus wherein we borrowed the first tranche of $40.0 million upon closing of the debt financing in June 2022. The Innovatus Loan is secured by perfected first priority liens on our assets. The Innovatus Loan includes customary events of default, including failure to pay amounts due, breaches of covenants and warranties, material adverse change events, certain cross defaults and judgments, and insolvency. The Innovatus Loan provides that upon the filing of the Bankruptcy Petitions, the principal amount together with accrued and unpaid fees and interest thereon shall become immediately due and payable. However, any efforts to enforce such payment obligations under the Innovatus Loan are automatically stayed as a result of the Bankruptcy Petitions, and the creditors’ rights of enforcement in respect of the Innovatus Loan are subject to the applicable provisions of the Bankruptcy Code. In connection with this default or any other default, the lenders under the Innovatus Loan can impose a 5.0% interest rate penalty, restrict access to additional borrowings under the loan and security agreement and accelerate the maturity of the debt, any of which could materially affect our liquidity and ability to fund our operations.
If we raise additional funds through collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish valuable rights to our product candidates or future revenue streams or grant licenses on terms that are not favorable to us. In addition, while we are seeking strategic partners for our virology assets, lonafarnib and peginterferon lambda, we may not be able to identify strategic partners, or enter into agreements on terms favorable to us, or at all. In addition, such transactions may require consents from third parties, which we may be unable to obtain. Further, our ability to raise additional capital may be adversely impacted by worsening global economic conditions and continuing disruptions to and volatility in the credit and financial markets in the U.S. and worldwide. We cannot assure you that we will be able to obtain additional funding if and when necessary to fund our entire portfolio of product candidates to meet our projected plans. If we are unable to obtain funding on a timely basis, we may be required to delay or discontinue one or more of our development programs or the commercialization of any product candidates or be unable to expand our operations or otherwise capitalize on potential business opportunities, which could materially affect our business, financial condition, and results of operations.
Covenants in the Innovatus Loan restrict our business and operations in many ways, and if we do not effectively comply with our covenants, our financial conditions and results of operations could be adversely affected.
The Innovatus Loan provides for up to $75.0 million in term loans due on August 31, 2027, of which $41.5 million in principal is outstanding as of December 31, 2023. All of our current and future assets, secure our borrowings under the Innovatus Loan. The Innovatus Loan requires that we comply with certain covenants applicable to us, including among other things, covenants restricting dispositions, changes in business, management, ownership or business locations, mergers or acquisitions, indebtedness, encumbrances, distributions, investments, transactions with affiliates and subordinated debt, any of which could restrict our business and operations, particularly our ability to respond to changes in our business or to take specified actions to take advantage of certain business opportunities that may be presented to us. Our failure to comply with any of the covenants could result in a default under the Innovatus Loan, which could permit the lenders to declare all or part of any outstanding borrowings to be immediately due and payable. The Innovatus Loan provides that upon the filing of the Chapter 11 Cases, the principal amount together with accrued and unpaid fees and interest thereon became immediately due and payable. However, any efforts to enforce such payment obligations under the Innovatus Loan are automatically stayed as a result of the Chapter 11 Cases, and the creditors’ rights of enforcement in respect of the Innovatus Loan are subject to the applicable provisions of the Bankruptcy Code. If we are unable to repay any accelerated or other amounts due, the lenders under the Innovatus Loan could proceed against the collateral granted to them to secure that debt, and our inability to use or dispose of those assets would seriously harm our business. In addition, should we be unable to comply with these covenants or if we default on any portion of our outstanding borrowings, the lenders can also impose a 5.0% interest rate penalty, restrict access to additional borrowings under the loan and security agreement, and accelerate the maturity of the debt. Any default under the Innovatus Loan, including the default discussed above, would materially affect our liquidity and ability to fund our operations and complete our planned clinical trials and regulatory filings would be substantially impaired.
Risks Related to the Development of our Product Candidates
We are dependent on the success of our product candidates, which are in various stages of clinical development. Certain of our product candidates have produced results in academic settings to date or for other indications than those that we contemplate, and we cannot give any assurance that we will generate data for any of our product candidates sufficient to receive regulatory approval in our planned indications, which will be required before they can be commercialized.
To date, we have invested substantially all of our efforts and financial resources to identify, acquire, and develop our portfolio of product candidates. Our future success is dependent on our ability to further develop, obtain regulatory approvals for, and commercialize one or more of these product candidates. Our NDA for Zokinvy to reduce the risk of mortality in HGPS, and for treatment of processing-deficient progeroid laminopathies (PL) with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, was approved in November 2020. Prior to the U.S. Zokinvy commercial launch in 2021, we had not generated revenue from sales of any products and may never be able to develop or commercialize additional product candidates. In addition, we have a commitment to provide access to Zokinvy for patients with HGPS and PL with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, for no or minimal cost to those patients.
With respect to potential commercial products, we currently have one product candidate, avexitide, that has completed Phase 2 for PBH and HI. Phase 3 study start up activities for avexitide in PBH and for avexitide in HI have been paused until additional funding is secured. It may be years before our studies are completed, and new studies are initiated, if at all.
We provide our geographically diverse clinical sites with good clinical practice protocols. We review and monitor the execution of our protocols at our various sites in an effort to understand those protocols are being followed. There can be no assurance that the data we develop for our product candidates in our planned indications will be sufficient or complete enough to obtain regulatory approval. Likewise, there can be no assurance that the data obtained from foreign clinical trial sites in studies not conducted under an investigational new drug, or IND, application will be accepted in support of an application for regulatory approval or authorization for use in the U.S. Similarly, data obtained from foreign clinical trial sites may not be accepted by other foreign regulatory authorities in support of an application for regulatory approval or authorization for use in these jurisdictions. We are not permitted to market or promote any of our product candidates before we receive regulatory approval from the FDA or comparable foreign regulatory authorities, and we may never receive such regulatory approval for any of our current product candidates. We cannot be certain that any of our product candidates will be successful in clinical studies or receive regulatory approval. Further, our product candidates may not receive regulatory approval even if they are successful in clinical studies. If we do not receive regulatory approvals for our product candidates, we may not be able to continue our operations.
We may not be able to obtain FDA approval of any future NDA or BLA for our product candidates.
The clinical development, manufacturing, labeling, packaging, storage, recordkeeping, advertising, promotion, export, import, marketing and distribution and other possible activities relating to lonafarnib, avexitide and any other product candidate that we may develop in the future are subject to extensive regulation in the United States. Prior to the approval of our NDA for Zokinvy to reduce the risk of mortality in HGPS, and for treatment of PL with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, we had not submitted an application for approval or obtained FDA approval for any product.
Approval of an NDA or a BLA is not guaranteed, and the approval process is an expensive and uncertain process that may take several years. The FDA and foreign regulatory entities also have substantial discretion in the approval process. The number and types of preclinical studies and clinical trials that will be required for approval varies depending on the product candidate, the disease or the condition that the product candidate is designed to target and the regulations applicable to any particular product candidate. Data are subject to varying interpretation and the FDA may not agree that our clinical data support that any of our product candidates are safe and effective for the proposed therapeutic use. Despite the time and expense associated with preclinical studies and clinical trials, failure can occur at any stage, and we could encounter problems that require us to repeat or perform additional preclinical studies or clinical trials or generate additional chemistry, manufacturing and controls data, including product stability data. In previous studies, ECG abnormalities were observed in our lonafarnib program. In addition, our Phase 3 LIMT-2 study of peginterferon lambda in patients with chronic HDV was terminated due to observations of hepatobiliary events that resulted in liver decompensation. The decision was based on the recommendation of the study's Data Safety Monitoring Board and FDA following a safety review. The FDA and similar foreign authorities could delay, limit or deny approval of a product candidate, and may
ultimately approve the product for narrower indications or with unfavorable labeling that would impede our commercialization of the product.
Approval procedures vary among countries and can involve additional product testing and additional administrative review periods, including obtaining reimbursement and pricing approval in select markets. The time required to obtain approval in other countries might differ from that required to obtain FDA approval. The regulatory approval process in other countries may include all of the risks associated with FDA approval as well as additional, presently unanticipated, risks. Regulatory approval in one country does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory approval in one country may negatively impact the regulatory process in others, including the risk that our product candidates may not be approved for all indications requested and that such approval may be subject to limitations on the indicated uses for which the product may be marketed.
Our business strategy is based upon obtaining and maintaining Orphan Drug designation for our products candidates, which is an uncertain process. The regulatory approval processes of the FDA and comparable foreign authorities are lengthy, time consuming, and inherently unpredictable. If we are unable to obtain or maintain Orphan Drug designation or regulatory approval for our product candidates, our business would be substantially harmed.
Our approach to identifying and developing product candidates depends, in large part, on our ability to obtain and maintain Orphan Drug designation from regulatory authorities in major markets. Without the potential protection of this regulatory exclusivity upon approval, many of our product candidates would otherwise not justify investment. While we assess the potential for obtaining Orphan Drug designation at the time that we contemplate the acquisition of product candidates and we intend to timely file for such designation, there can be no assurance that we will obtain Orphan Drug designation or be able to successfully meet the regulatory requirements to maintain that designation with the planned clinical trials for our product candidates. Failure to obtain and maintain Orphan Drug designation would make our product candidates significantly less competitive and potentially not viable investments for further development. Although we currently have Orphan Drug designation for some of our product candidates in multiple targeted indications, failure to demonstrate significant benefit over existing approved products in pivotal clinical trials may lead to marketing approval but without qualifying for Orphan Drug protection in some regions, such as in Europe.
The time required to obtain approval by the FDA and comparable foreign authorities is unpredictable, typically takes many years following the commencement of clinical studies and depends upon numerous factors. In addition, approval policies, regulations, or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s clinical development and may vary among jurisdictions, which may cause delays in the approval or the decision not to approve an application. We have obtained U.S. and EMA regulatory approval for one product, Zokinvy, and it is possible that none of our current product candidates or any future product candidates we may seek to develop will ever obtain regulatory approval.
Future applications for our product candidates could fail to receive regulatory approval for many reasons, including but not limited to the following:
•the FDA or comparable foreign regulatory authorities may disagree with the design, size or implementation of our clinical studies;
•the population studied in the clinical program may not be sufficiently broad or representative to assure safety in the full population for which we seek approval;
•the FDA or comparable foreign regulatory authorities may disagree with our interpretation of data from our development efforts;
•the data collected from clinical studies of our product candidates may not be sufficient or complete or meet the regulatory requirements to support the submission of an NDA, BLA, or other submission or to obtain regulatory approval in the United States or foreign jurisdictions;
•the FDA or comparable foreign regulatory authorities may find failures in our manufacturing processes, validation procedures and specifications, or facilities of our third-party manufacturers with which we contract for clinical and commercial supplies that may delay or limit our ability to obtain regulatory approval for our product candidates; and
•the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering our NDA, BLA or other submission insufficient for approval.
The lengthy and uncertain regulatory approval process, as well as the unpredictability of the results of clinical studies, may result in our failing to obtain additional regulatory approval to market any of our product candidates or to be significantly delayed from our expectations for potential approval, which would significantly harm our business, results of operations, and prospects. In addition, although we have obtained Orphan Drug designation for five of our development programs to date, there can be no assurance that the FDA or comparable foreign regulatory authorities will grant our similar status for our other proposed development indications or other product candidates in the future.
Although the FDA has granted Rare Pediatric Disease designation to avexitide for the treatment of congenital hyperinsulinism, NDA approval for this program may not meet the eligibility criteria for a priority review voucher.
Our avexitide compound has received Rare Pediatric Disease (RPD) designation from the FDA for the treatment of congenital hyperinsulinism (HI). The FDA defines a “rare pediatric disease” as a serious or life-threatening disease in which the serious or life-threatening manifestations primarily affect patients under the age of 18 years, that is, a disease or condition that affects fewer than 200,000 individuals in the United States, or a patient population greater than 200,000 in the United States when there is no reasonable expectation that the cost of developing and making available the product in the United States will be recovered from sales in the United States for that drug or biological product. Specifically, under this program, a sponsor who receives an approval for a drug or biologic for a “rare pediatric disease” may qualify for a voucher that can be redeemed to receive a priority review of a subsequent marketing application for a different product. The sponsor of a rare pediatric disease product receiving a priority review voucher may transfer (including by sale) the voucher to another sponsor. The voucher may be further transferred any number of times before the voucher is used, as long as the sponsor making the transfer has not yet submitted the application. The FDA may also revoke any priority review voucher if the rare pediatric disease product for which the voucher was awarded is not marketed in the U.S. for at least 12 months following the date of approval. In addition, the priority review voucher is only awarded to an NCE, thus if a compound is approved first for an indication that is not a rare pediatric disease the compound may not be eligible to receive the voucher. While we obtained and sold the priority review voucher issued upon approval of Zokinvy to reduce the risk of mortality in HGPS, and for treatment of PL with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, there can be no assurance that we will be successful in obtaining approval for avexitide for the treatment of HI, or that a priority review voucher will be issued at the time of any such approval.
Congress has only authorized the Rare Pediatric Disease Priority Review Voucher Program until September 30, 2024. However, if a product candidate receives RPD designation before September 30, 2024, it is eligible to receive a voucher if it is approved before September 30, 2026. Avexitide may not be approved by that date, or at all, and, therefore, we may not be in a position to obtain a priority review voucher prior to expiration of the program, unless Congress further reauthorizes the program.
There is no assurance we will receive a rare pediatric disease priority review voucher. Also, although priority review vouchers may be sold or transferred to third parties, there is no guarantee that we will be able to realize any value if we were to sell a priority review voucher.
Although we have received Breakthrough Therapy designations, this may not lead to a faster development, regulatory review or approval process, and it does not increase the likelihood of receiving marketing approval in the United States.
We have received Breakthrough Therapy designation for avexitide for the treatment of PBH and HI. A Breakthrough Therapy is defined as a therapy that is intended, alone or in combination with one or more other therapies, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the therapy may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. For therapies that have been designated as breakthrough therapies, interaction and communication between the FDA and the sponsor of the trial can help to identify the most efficient path for clinical development while minimizing the number of patients placed in ineffective control regimens. Therapies designated as breakthrough therapies by the FDA may also be eligible for priority review and accelerated approval.
Designation as a breakthrough therapy is within the discretion of the FDA. Accordingly, even if we believe one of our product candidates meets the criteria for designation as a breakthrough therapy, the FDA may disagree and instead determine not to make such designation. The Breakthrough Therapy designations we have obtained may not result in faster development processes, reviews or approvals compared to therapies considered for approval under conventional FDA procedures and does not assure ultimate approval by the FDA. In addition, the FDA may later decide that any of our development programs no longer meet the conditions for qualification or decide that the time period for FDA review or
approval will not be shortened. In addition, even if one or more of our product candidates qualify as breakthrough therapies, the FDA may later decide that the product no longer meets the conditions for qualification. Thus, even though we intend to seek Breakthrough Therapy designation for some or all of our future product candidates for the treatment of various conditions, there can be no assurance that we will receive any such Breakthrough Therapy designation.
We may submit an application for our product candidates under the Accelerated Approval Pathway. If we are unable to obtain approval or licensure of our product candidates through the Accelerated Approval Program in the United States, we may be required to conduct additional nonclinical and clinical studies and trials beyond those that we currently contemplate, which could increase the expense of obtaining, reduce the likelihood of obtaining and/or delay the timing of obtaining, necessary marketing approval. Even if we receive approval from the FDA through the Accelerated Approval Program, if any required confirmatory post-marketing trial does not verify clinical benefit, or if we do not comply with rigorous post-marketing requirements, the FDA may seek to withdraw the approval.
We may submit an NDA or BLA for our product candidates under the Accelerated Approval Pathway. If we are unable to obtain approval of our product candidates through the Accelerated Approval Program in the United States, we may be required to conduct additional nonclinical and clinical studies and trials beyond those that we currently contemplate, which could increase the expense of obtaining, reduce the likelihood of obtaining and/or delay the timing of obtaining, necessary marketing approval. Even if we receive approval from the FDA through the Accelerated Approval Program, if any required confirmatory post-marketing trial does not verify clinical benefit, or if we do not comply with rigorous post-marketing requirements, the FDA may seek to withdraw the approval.
For any approval to market a product, we must provide the FDA and foreign regulatory agencies with clinical data that adequately demonstrate the safety, efficacy, purity and/or potency of the product for the indication applied for in the NDA, BLA, or other respective regulatory filings. The Accelerated Approval Program is one of several approaches used by the FDA to make prescription drugs and biologics more rapidly available for the treatment of serious or life-threatening diseases. Section 506(c) of the Federal Food, Drug and Cosmetic Act (FDCA) provides that the FDA may grant accelerated approval to “a product for a serious or life-threatening condition upon a determination that the product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments.” Approval through the Accelerated Approval Program is typically subject, however, to the requirement that the applicant conduct additional post-marketing clinical trials to verify and describe the product’s clinical benefit. Typically, clinical benefit is verified when post-marketing clinical trials show that the product provides a clinically meaningful positive therapeutic effect, that is, an effect on how a patient feels, functions, or survives. The FDA may require that these studies be underway prior to Accelerated Approval pursuant to the Food and Drug Omnibus Reform Act of 2022 (FDORA). If such confirmatory post-marketing trial fails to confirm the product’s clinical profile or risks and benefits, the FDA may withdraw its approval of the product. The FDA has broad discretion with regard to approval through the Accelerated Approval Program, and even if we believe that the Accelerated Approval Program is appropriate for our product candidates, we cannot assure you that the FDA will ultimately agree. The FDA may also change its policies with respect over Accelerated Approval over time. For example, in March 2023, the FDA announced the availability of draft guidance on “Clinical Trial Considerations to Support Accelerated Approval of Oncology Therapeutics,” in which the agency outlined, and invited public comment on, its “preferred approach” of randomized controlled trials, including those that provide for longer term follow-up that could fulfill a post-marketing requirement to verify clinical benefit. In that draft guidance, the FDA acknowledged that historically, single-arm trial designs and response endpoints have most commonly been used in oncology, but noted that such trials have limitations. Furthermore, even if we do obtain approval through the Accelerated Approval Program, we may not experience a faster development process, review or approval compared to conventional FDA procedures.
Our lonafarnib-based regimens for the treatment of HDV has completed Phase 3 studies with endpoints intended to support accelerated approval. The primary endpoint for the D-LIVR study, the Phase 3 study of lonafarnib-based regimens, is a composite of a > 2 log reduction in HDV RNA and ALT normalization at end of 48 weeks of treatment and is intended to support accelerated approval. The study endpoints were previously achieved in Phase 2 studies and are consistent with FDA guidance on the development of treatments for HDV. While these proposed endpoints are designed to be consistent with FDA guidance, there is no assurance that approval will be granted on a timely basis, or at all. FDA may disagree that the design of, or results from, our studies supporting accelerated approval. Additionally, the FDA could require us to conduct further studies or trials prior to granting approval of any type, including by determining that approval through the Accelerated Approval Program is not appropriate and that our clinical trials may not be used to support approval through the conventional pathway. We might not be able to fulfill the FDA’s requirements in a timely manner, which would cause
delays, or approval might not be granted because our submission is deemed incomplete by the FDA. There also can be no assurance that after subsequent FDA feedback we will continue to pursue approval through the Accelerated Approval Program. A failure to obtain approval through the Accelerated Approval Program could result in a longer time period to obtain approval of our product candidates, could increase the cost of their development, could delay our ability to commercialize our products and could significantly harm our financial position and competitive position in the marketplace.
Even if we receive approval for one or more of our product candidates through the Accelerated Approval Program, we will be subject to rigorous post-marketing requirements, possibly including the completion of one or more confirmatory post-marketing trials as the FDA may require, to verify the clinical benefit of the product, and submission to the FDA of all promotional materials prior to their dissemination. The FDA could seek to withdraw the approval for multiple reasons, including if we fail to conduct any required confirmatory post-marketing trial with due diligence, our confirmatory post-marketing trial does not confirm the predicted clinical benefit, other evidence shows that the product is not safe or effective under the conditions of use, or we disseminate promotional materials that are found by the FDA to be false and misleading.
Moreover, Congress has recently enacted potential changes to the Accelerated Approval Program that could impact our ability to obtain an Accelerated Approval, or increase the burdens associated with post marketing requirements in the event we do obtain an Accelerated Approval. In particular, FDA must specify certain conditions for required post approval studies for products that receive Accelerated Approval, which may include enrollment targets and milestones, including the target date for study completion, by the time the product is approved. FDA may also require post approval studies to be underway at the time of Accelerated Approval or within a specified time period following Accelerated Approval for such products.
Any delay in obtaining, or inability to obtain, approval through the Accelerated Approval Program, or any issues in maintaining approval granted under the Accelerated Approval Program, would delay or prevent commercialization of our products, and would materially adversely affect our business, financial condition, results of operations, cash flows and prospects
Product development involves a lengthy and expensive process with an uncertain outcome, and results of earlier studies may not be predictive of future study results.
Clinical testing is expensive and generally takes many years to complete, and the outcome is inherently uncertain. Failure can occur at any time during the clinical study process. The results of preclinical studies and early clinical studies of our product candidates may not be predictive of the results of larger, later-stage controlled clinical studies. Product candidates that have shown promising results in early-stage clinical studies may still suffer significant setbacks in subsequent clinical studies. Our clinical studies to date have been conducted on a small number of patients in limited numbers of clinical sites and in academic settings or for other indications. We will have to conduct larger, well-controlled studies in our proposed indications to verify the results obtained to date and to support any regulatory submissions for further clinical development. A number of companies in the biopharmaceutical industry have suffered significant setbacks in advanced clinical studies due to lack of efficacy or adverse safety profiles despite promising results in earlier, smaller clinical studies. Moreover, clinical data are often susceptible to varying interpretations and analyses. We do not know whether any Phase 2, Phase 3, or other clinical studies we have conducted or may conduct will demonstrate consistent or adequate safety, efficacy, purity and/or potency with respect to the proposed indication for use sufficient to obtain regulatory approval to receive regulatory approval or market our product candidates. For example, in September 2023, we announced our decision to discontinue the Phase 3 LIMT-2 study of peginterferon lambda in patients with chronic HDV. The decision was based on the recommendation of the Data Safety Monitoring Board (DSMB) for the study following its quarterly safety review. In a communication dated September 7, 2023, the DSMB recommended, and FDA agreed, to the discontinuation of the LIMT-2 study due to observations of four patients with hepatobiliary events that resulted in liver decompensation.
We may find it difficult to enroll patients in our clinical studies given the limited number of patients who have the diseases for which our product candidates are being studied. Difficulty in enrolling patients could delay or prevent clinical studies of our product candidates.
Identifying and qualifying patients to participate in clinical studies of our product candidates is essential to our success. The timing of our clinical studies depends in part on the rate at which we can recruit patients to participate in clinical trials of our product candidates, and we may experience delays in our clinical studies if we encounter difficulties in enrollment.
The eligibility criteria of our planned clinical studies may further limit the available eligible study participants as we expect to require that patients have specific characteristics that we can measure or meet the criteria to assure their conditions are appropriate for inclusion in our clinical studies. We may not be able to identify, recruit, and enroll a sufficient number of patients to complete our clinical studies in a timely manner because of the perceived risks and benefits of the product candidate under study, the availability and efficacy of competing therapies and clinical studies, and the willingness of physicians to participate in our planned clinical studies. For example, early in the COVID-19 pandemic, certain clinical study sites that were scheduled to open were delayed in activating and other sites suspended randomization of subjects for a period of time. Future restrictions could result in delays of our clinical trials. If patients are unwilling to participate in our clinical studies for any reason, the timeline for conducting studies and obtaining regulatory approval of our product candidates may be delayed.
FDA may also modify or enhance trial requirements which may affect enrollment. In August 2023, FDA published a guidance document, Informed Consent, Guidance for IRBs, Clinical Investigators, and Sponsors, which supersedes past guidance and finalizes draft guidance on informed consent. Further, in December 2023, FDA published a final rule, Institutional Review Board Waiver or Alteration of Informed Consent for Minimal Risk Clinical Investigations, which allows exceptions from informed consent requirements when a clinical investigation poses no more than minimal risk to the human subject and includes appropriate safeguards to protect the rights, safety, and welfare of human subjects. FDA’s new guidance document and rule present evolving requirements for informed consent which may affect recruitment and retention of patients in clinical trials. Effects on recruitment and retention of patients may hinder or delay a clinical trial and could cause a significant setback to an applicable program.
If we experience delays in the completion of, or termination of, any clinical study of our product candidates, the commercial prospects of our product candidates could be harmed, and our ability to generate product revenue from any of these product candidates could be delayed or prevented. In addition, any delays in completing our clinical studies would likely increase our overall costs, impair product candidate development and jeopardize our ability to obtain regulatory approval relative to our current plans. Any of these occurrences may harm our business, financial condition, and prospects significantly.
Clinical studies are costly, time consuming and inherently risky, and we may fail to demonstrate safety, efficacy, purity and/or potency to the satisfaction of applicable regulatory authorities.
Clinical development is expensive, time consuming and involves significant risk. We cannot guarantee that any clinical studies will be conducted as planned or completed on schedule, if at all. A failure of one or more clinical studies can occur at any stage of development. Events that may prevent successful or timely completion of clinical development include but are not limited to:
•inability to generate satisfactory preclinical, toxicology, or other in vivo or in vitro data or diagnostics to support the initiation or continuation of clinical studies necessary for product approval;
•delays in reaching agreement on acceptable terms with contract research organizations (CROs) and clinical study sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and clinical study sites;
•delays in obtaining required Institutional Review Board (IRB) approval at each clinical study site;
•failure to permit the conduct of a study by regulatory authorities, after review of an investigational new drug (IND) or equivalent foreign application or amendment;
•delays in recruiting qualified patients, or patients dropping out of, in our clinical studies;
•feasibility of continuous trial execution in countries impacted by war, geopolitical conflict and other humanitarian crises, including conflicts in the Middle East;
•failure by clinical sites or our CROs or other third parties to adhere to clinical study requirements or report complete findings;
•failure to perform the clinical studies in accordance with good clinical practice (GCP) requirements, or applicable foreign regulatory guidelines;
•occurrence of adverse events associated with our product candidates;
•changes in regulatory requirements and guidance that require amending or submitting new clinical protocols;
•the cost of clinical studies of our product candidates;
•negative or inconclusive results from our clinical trials which may result in our deciding, or regulators requiring us, to conduct additional clinical studies or abandon development programs in other ongoing or planned indications for a product candidate; and
•delays in reaching agreement on acceptable terms with third-party manufacturers and the time for manufacture of sufficient quantities of our product candidates for use in clinical studies.
Any inability to successfully complete clinical development and obtain regulatory approval could result in additional costs to us or impair our ability to generate revenue. Further, requirements regarding clinical trial data may evolve which could cause delay or result in additional costs. In June 2023, FDA published a draft guidance, E6(R3) Good Clinical Practice (GCP), which seeks to unify standards for clinical trial data for ICH member countries and regions. Changes to data requirements may cause FDA or comparable foreign regulatory authorities to disagree with data from preclinical studies or clinical trials, and may require further studies.
Clinical study delays could also shorten any periods during which our products have patent protection and may allow competitors to develop and bring products to market before we do, which could impair our ability to obtain or maintain Orphan Drug designation exclusivity and to successfully commercialize our product candidates and may harm our business and results of operations.
The COVID-19 Public Health Emergency (PHE) expired in the U.S. and, while we intend to pursue similar temporary use authorizations in non-U.S. jurisdictions, we may not be able to receive such authorizations from foreign regulatory agencies. If we do receive a similar temporary use authorization, absent a full marketing authorization for that indication, our ability to sell our products may be limited.
Various regulatory pathways are available in jurisdictions outside the United States to make products available for emergency use. For example, regulatory authorities in certain EU Member States may temporarily authorize the distribution of an unauthorized drug in response to the suspected or confirmed spread of pathogenic agents such as the virus which is causing COVID-19. Obtaining such a temporary authorization is dependent upon a number of factors, which are not under our control. If such authorizations would be granted, they would only apply for a limited period of time. We might thus no longer be authorized to distribute our product under these authorizations if that time limit expired. The regulatory authorities in the EU or in other jurisdictions outside the U.S. may grant a conditional marketing authorization for medicinal products intended for the treatment of seriously debilitating or life-threatening diseases prior to the submission of comprehensive clinical data if that treatment is of major therapeutic advantage to the patients concerned or no other authorized treatment is available. In emergency situations, such a conditional marketing authorization may also be granted for these medicinal products where comprehensive preclinical or pharmaceutical data have not been supplied. These conditional marketing authorizations are subject to specific conditions (e.g., completing on-going studies or conducting new studies) which must be fulfilled within a timeline specified in the marketing authorization. These marketing authorizations are valid for a short period of time (e.g., one year) which can usually be renewed. If we would apply for such a conditional marketing authorization the regulatory authority concerned may reject our application because it considers that the benefit-risk balance of our product is not favorable or it judges it unlikely that we would be able to provide comprehensive data. If we would obtain such a conditional marketing authorization we may not be able to complete (timely) the studies which the regulatory authority imposed as a condition for the marketing authorization or the data collected in the course of these studies may indicate that our product does not have a favorable benefit-risk profile. As a result, we may not be able to continue distributing our product because the conditional marketing authorization has been revoked or not renewed, or the regulatory authority refused finally to grant a regular marketing authorization.
Regulatory authorities in the EU or other jurisdictions outside the EU may make a product available for compassionate reasons to a group of patients with a chronically or seriously debilitating disease or whose disease is considered to be life-threatening, and who cannot be treated satisfactorily with an authorized medicinal product provided that the product is subject of an application for marketing authorization or is undergoing clinical trials. If we would request a regulatory authority to make our product available to patient under these conditions the regulatory authority may reject our request if, for example, it considers that the patients concerned can be treated satisfactorily with other products that are already authorized.
Programs which make products available under the conditions mentioned above are usually authorized for a limited period of time (e.g., one year). Regulatory authorities may not renew expiring authorizations for these programs if we terminate prematurely a clinical trial with our product or decide not to submit or to withdraw an application for marketing authorization in the jurisdiction concerned. That may, for example, happen because pharmacovigilance data or other data collected during our clinical trials indicate that our product does not have the appropriate benefit-risk balance.
Our product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label, or result in significant negative consequences following marketing approval, if any.
Our avexitide product candidate has been studied in over 70 PBH patients and 39 HI patients, and the most common adverse events are injection site bruising/reaction, headache, diarrhea, nausea and dizziness. There is no guarantee that additional or more severe side effects will not be identified through other clinical studies of avexitide.
Our lonafarnib product candidate has been studied in thousands of oncology patients, and the most common non-hematologic adverse events of any grade were gastrointestinal system disorders (nausea, anorexia, diarrhea and vomiting), weight loss, fatigue and rash. Use of Zokinvy to reduce the risk of mortality in HGPS, and for treatment of processing-deficient progeroid laminopathies with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, has been reported to cause ECG abnormalities, but these ECG abnormalities have not resulted in a risk of mortality for these patients. There is no guarantee that additional or more severe side effects or other properties will not be identified through other clinical studies of lonafarnib.
For example, in September 2023, we announced our decision to discontinue the Phase 3 LIMT-2 study of peginterferon lambda in patients with HDV. The decision was based on the recommendation of the Data Safety Monitoring Board (DSMB) for the study following its quarterly safety review. In a communication dated September 7, 2023, the DSMB recommended, and FDA agreed, to the discontinuation of the LIMT-2 study due to observations of four patients with hepatobiliary events that resulted in liver decompensation. There is no guarantee that additional or more severe side effects will not be identified through future clinical studies for other uses of peginterferon lambda. Undesirable side effects, other properties, and negative results for other indications may negatively impact the development and potential for approval of our product candidates for our proposed indications.
Additionally, even if one or more of our current product candidates receives marketing approval, and we or others later may identify undesirable side effects caused by such products, potentially significant negative consequences could result, including but not limited to:
•regulatory authorities may withdraw approvals of such products;
•regulatory authorities may require additional warnings on the label;
•we may be required to create a Risk Evaluation and Mitigation Strategy (REMS) plan, which could include a medication guide outlining the risks of such side effects for distribution to patients, a communication plan for healthcare providers, and/or other elements to assure safe use;
•we could be sued and held liable for harm caused to patients; and
•our reputation may suffer.
Any of these events could prevent us from achieving or maintaining market acceptance of a product candidate, even if approved, and could significantly harm our business, results of operations, and prospects.
We are subject to ongoing regulatory requirements related to the U.S., EU, and Japan approvals of Zokinvy, and if we obtain additional regulatory approvals for a product candidate, we will be subject to additional ongoing regulatory requirements.
If our product candidates are approved, they will be subject to ongoing regulatory requirements with respect to manufacturing, labeling, packaging, storage, advertising, promotion, sampling, record-keeping, conduct of post-marketing studies, and submission of safety, efficacy and other post-approval information, including both federal and state requirements in the United States and requirements of comparable foreign regulatory authorities.
Manufacturers and manufacturers’ facilities are required to continuously comply with FDA and comparable foreign regulatory authority requirements, including ensuring that quality control and manufacturing procedures conform to current Good Manufacturing Practices (cGMP) regulations and corresponding foreign regulatory manufacturing requirements. As such, we and our contract manufacturers will be subject to continual review and inspections to assess compliance with cGMP and adherence to commitments made in any NDA, BLA, or MAA.
Any regulatory approvals that we receive for our product candidates may be subject to limitations on the approved indicated uses for which the product candidate may be marketed or to the conditions of approval, or contain requirements
for potentially costly post-marketing testing, including Phase 4 clinical trials, and surveillance to monitor the safety, efficacy, purity and/or potency of the product candidate. We will be required to report certain adverse reactions and production problems, if any, to the FDA and comparable foreign regulatory authorities.
Any new legislation addressing product safety issues could result in delays in product development or commercialization, or increased costs to assure compliance. If our original marketing approval for a product candidate was obtained through an accelerated approval pathway, we could be required to conduct a successful post-marketing clinical study to confirm the clinical benefit for our products. An unsuccessful post-marketing study or failure to complete such a study could result in the withdrawal of marketing approval.
If a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, or disagrees with the promotion, marketing, or labeling of a product, the regulatory agency may impose restrictions on that product or us, including requiring withdrawal of the product from the market. If we fail to comply with applicable regulatory requirements, a regulatory agency or enforcement authority may, among other things:
•issue warning letters;
•impose civil or criminal penalties;
•suspend or withdraw regulatory approval;
•suspend any of our ongoing clinical studies;
•refuse to approve pending applications or supplements to approved applications submitted by us;
•impose restrictions on our operations, including closing our contract manufacturers’ facilities; or
•require a product recall.
In addition, prescription drugs and biologics may be promoted only for the approved indications in accordance with the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label may be subject to significant liability. However, physicians may, in their independent medical judgment, prescribe legally available products for off-label uses. The FDA does not regulate the behavior of physicians in their choice of treatments, but the FDA does restrict manufacturer’s communications on the subject of off-label use of their products.
Any government investigation of alleged violations of law would be expected to require us to expend significant time and resources in response and could generate adverse publicity. Any failure to comply with ongoing regulatory requirements may significantly and adversely affect our ability to develop and commercialize our products and the value of us and our operating results would be adversely affected.
We rely on third parties to conduct our clinical studies, manufacture our product candidates and perform other services. If these third parties do not successfully perform and comply with regulatory requirements, we may not be able to successfully complete clinical development, obtain regulatory approval or commercialize our product candidates and our business could be substantially harmed.
We have relied upon and plan to continue to rely upon investigators and third-party CROs to conduct, monitor and manage our ongoing clinical programs. We rely on these parties for execution of clinical studies and manage and control only certain aspects of their activities. We remain responsible for ensuring that each of our studies is conducted in accordance with the applicable protocol, legal, regulatory, and scientific standards, and our reliance on the CROs does not relieve us of our regulatory responsibilities. We, our investigators, and our CROs and other vendors are required to comply with all applicable laws, regulations and guidelines, including those required by the FDA and comparable foreign regulatory authorities for all of our product candidates in clinical development. If we or any of our investigators, CROs or vendors fail to comply with applicable laws, regulations and guidelines, the results generated in our clinical studies may be deemed unreliable and the FDA or comparable foreign regulatory authorities may require us to perform additional studies before approving our marketing applications. We cannot assure you that our CROs and other vendors will meet these requirements, or that upon inspection by any regulatory authority, such regulatory authority will determine that efforts, including any of our clinical studies, comply with applicable requirements. Our failure to comply with these laws, regulations and guidelines may require us to repeat clinical studies or conduct larger additional studies, which would be costly and delay the regulatory approval process.
If any of our relationships with investigators or third-party CROs terminate, we may not be able to enter into arrangements with alternative CROs in a timely manner or do so on commercially reasonable terms. In addition, our CROs may not prioritize our clinical studies relative to those of other customers and any turnover in personnel or delays in the allocation of CRO employees by the CRO may negatively affect our clinical studies. If investigators or CROs do not successfully carry out their contractual duties or obligations or meet expected deadlines, our clinical studies may be delayed or terminated, and we may not be able to meet our current plans with respect to our product candidates. CROs may also involve higher costs than anticipated, which could negatively affect our financial condition and operations. In addition, we do not currently have, nor do we plan to establish, the capability to manufacture product candidates for use in the conduct of our clinical studies or in support of our commercialization of potential products, and we lack the resources and the capability to manufacture any of our product candidates on a clinical or commercial scale without the use of third-party manufacturers. We plan to rely on third-party manufacturers and their responsibilities will include purchasing from third-party suppliers the materials necessary to produce our product candidates for our clinical studies and regulatory approval. There are expected to be a limited number of suppliers for the active ingredients and other materials that we expect to use to manufacture our product candidates, and we may not be able to identify alternative suppliers to prevent a possible disruption of the manufacture of our product candidates for our clinical studies, and, if approved, ultimately for commercial sale. Although we generally do not expect to begin a clinical study unless we believe we have a sufficient supply of a product candidate to complete the study, any significant delay or discontinuity in the supply of a product candidate, or the active ingredient or other material components in the manufacture of the product candidate, could delay completion of our clinical studies and potential timing for regulatory approval of our product candidates, which would harm our business and results of operations. For example, in connection with Phase 3 start-up activities for avexitide, we observed low levels of product-related impurities in the finished drug product. Although not unusual for this class of compounds, we are working with our CMOs to control these impurities to a level agreed by FDA, and to ensure that an adequate supply of materials with a sufficient shelf-life can be released for Phase 3 studies. In the longer term, the impurities will be fully tox qualified to a higher level, and the product specifications will be updated to allow for a suitable commercial shelf-life.
With respect to our lonafarnib program, we procured an inventory of product from Merck to supply our initial clinical study needs. In 2016, we transferred the manufacturing of drug substance and drug product to our third-party contractors. The material used for ongoing progeria clinical studies, expanded access program, and commercial Zokinvy supply are sourced from CMOs. These same vendors are currently under development for commercial qualification. Materials used for our avexitide clinical trials are also sourced from CMOs. Our vendors have successfully made GMP batches for our clinical studies, however, our trials could be delayed if quality, stability, or other issues occur in relation to the manufacture of any unique batch. If these CMOs are not able to provide us with sufficient quantities of product for our clinical trials or in support of our commercialization of potential products on a timely basis, or at all, whether due to production shortages or other supply interruptions resulting from any delay, our clinical trials or regulatory approval may be delayed, or could impair our ability to generate revenue from the sale of such product candidate.
We rely and expect to continue to rely on third parties to manufacture our clinical product supplies, and if those third parties fail to obtain approval of government regulators, fail to provide us with sufficient quantities of product, or fail to do so at acceptable quality levels or prices our product candidates could be stopped, delayed, or made less profitable.
We do not currently have nor do we plan to acquire the infrastructure or capability internally to manufacture our clinical supplies for use in the conduct of our clinical trials, and we lack the resources and the capability to manufacture any of our product candidates on a clinical or commercial scale. We currently rely on outside vendors to source raw materials and manufacture our clinical supplies of our product candidates and plan to continue relying on third parties to manufacture our product candidates on a commercial scale, if approved.
The facilities used by our contract manufacturers to manufacture our product candidates will be subject to pre-approval inspection by the FDA that will be conducted after we submit our marketing applications to the FDA or comparable foreign regulatory authorities. We do not control the manufacturing process of, and are completely dependent on, our contract manufacturing partners for compliance with the regulatory requirements, known as cGMPs, for manufacture of our product candidates. If our contract manufacturers cannot successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the FDA, EMA or others, our future applications may not be approved by regulatory authorities, which would significantly delay our commercialization plans and increase our costs. We have no control over the ability of our contract manufacturers to maintain adequate quality control, quality assurance and qualified personnel, and in the past, we have experienced quality control issues with product manufactured by our contract manufacturers. If the FDA or a comparable foreign regulatory authority does not approve these facilities for the manufacture of our product candidates or if it withdraws any such approval in the future, we may need to find alternative manufacturing facilities,
which would significantly impact our ability to develop, obtain regulatory approval for or market our product candidates, if approved.
We do not yet have sufficient information to reliably estimate the cost of the commercial manufacturing of our product candidates, and the actual cost to manufacture our product candidates could materially and adversely affect the commercial viability of our product candidates. As a result, we may not be able to develop additional commercially viable products.
In addition, our reliance on third-party manufacturers exposes us to the following additional risks:
•We may be unable to identify manufacturers on acceptable terms or at all;
•Our third-party manufacturers might be unable to timely formulate and manufacture our product or produce the quantity and quality required to meet our clinical and commercial needs, if any;
•Contract manufacturers may not be able to execute our manufacturing procedures appropriately;
•Our future contract manufacturers may not perform as agreed or may not remain in the contract manufacturing business for the time required to supply our clinical trials or to successfully produce, store and distribute our products;
•Manufacturers are subject to ongoing periodic unannounced inspection by the FDA and corresponding state agencies to ensure strict compliance with cGMP and other government regulations and corresponding foreign standards. We do not have control over third-party manufacturers’ compliance with these regulations and standards;
•We may not own, or may have to share, the intellectual property rights to any improvements made by our third-party manufacturers in the manufacturing process for our product candidates; and
•Our third-party manufacturers could breach or terminate their agreement with us.
Each of these risks could delay our clinical trials, the approval of any of our product candidates by the FDA or comparable foreign regulatory authorities or the commercialization of our product candidates or result in higher costs or deprive us of potential product revenue. In addition, we rely on third parties to perform release testing on our product candidates prior to delivery to patients. If these tests are not conducted appropriately and test data is not reliable, patients could be put at risk of serious harm and could result in product liability suits.
The manufacturing of medical products is complex and requires significant expertise and capital investment, including the development of advanced manufacturing techniques and process controls. Manufacturers of biologic products often encounter difficulties in production, particularly in scaling up and validating initial production and absence of contamination. These problems include difficulties with production costs and yields, quality control, including stability of the product, quality assurance testing, operator error, shortages of qualified personnel, as well as compliance with strictly enforced federal, state and foreign regulations. Furthermore, if contaminants are discovered in our supply of our product candidates or in the manufacturing facilities, such manufacturing facilities may need to be closed for an extended period of time to investigate and remedy the contamination. We cannot assure you that any stability or other issues relating to the manufacture of our product candidates will not occur in the future. Additionally, our manufacturers may experience manufacturing difficulties due to resource constraints or as a result of labor disputes or unstable political environments. If our manufacturers were to encounter any of these difficulties, or otherwise fail to comply with their contractual obligations, our ability to provide our product candidates to patients in clinical trials would be jeopardized. Any delay or interruption in the supply of clinical trial supplies could delay the completion of clinical trials, increase the costs associated with maintaining clinical trial programs and, depending upon the period of delay, require us to commence new clinical trials at additional expense or terminate clinical trials completely.
If the market opportunities for our product candidates are smaller than we believe they are, we may not meet our revenue expectations and, even assuming approval of a product candidate, our business may suffer. Because the patient populations in the market for our product candidates may be small, we must be able to successfully identify patients and acquire a significant market share to achieve profitability and growth.
We focus our product development principally on treatments for rare and ultra-rare diseases. Given the small number of patients who have the diseases that we are targeting, our eligible patient population and pricing estimates may differ significantly from the actual market addressable by our product candidate. Our projections of both the number of people who have these diseases, as well as the subset of people with these diseases who have the potential to benefit from treatment with our product candidates, are based on our beliefs and estimates. These estimates have been derived from a
variety of sources, including the scientific literature, patient foundations, or market research, and may prove to be incorrect. Further, new studies may change the estimated incidence or prevalence of these diseases. The number of patients may turn out to be lower than expected. For example, although we believe that data associated with avexitide are supportive of activity in PBH, there can be no assurance that our clinical trials will successfully address this condition. Likewise, the potentially addressable patient population for each of our product candidates may be limited or may not be amenable to treatment with our product candidates, and new patients may become increasingly difficult to identify or gain access to, which would adversely affect our results of operations and our business. Moreover, we expect that the sales of Zokinvy to patients with progeria will have limited profits given the ultra-rare nature of these diseases.
We face intense competition and rapid technological change and the possibility that our competitors may develop therapies that are similar, more advanced, or more effective than ours, which may adversely affect our financial condition and our ability to successfully commercialize our product candidates.
The biotechnology and pharmaceutical industries are intensely competitive and subject to rapid and significant technological change. We are currently aware of various existing therapies that may compete with our product candidates. For example, we have competitors both in the United States and internationally, including multinational pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies. Some of the pharmaceutical and biotechnology companies we expect to compete with include Recordati, MBX Bioscience, Vogenx, Zealand Pharmaceuticals, Rezolute, and Hanmi Pharmaceuticals as well as other smaller companies or biotechnology start-ups and large multinational pharmaceutical companies. Many of our competitors have substantially greater financial, technical, and other resources, such as larger research and development staff and experienced marketing and manufacturing organizations. Additional mergers and acquisitions in the biotechnology and pharmaceutical industries may result in even more resources being concentrated in our competitors. As a result, these companies may obtain regulatory approval more rapidly than we are able to and may be more effective in selling and marketing their products as well. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies. Competition may increase further as a result of advances in the commercial applicability of technologies and greater availability of capital for investment in these industries. Our competitors may succeed in developing, acquiring, or licensing on an exclusive basis, products that are more effective or less costly than any product candidate that we may develop, or achieve earlier patent protection, regulatory approval, product commercialization, and market penetration than we do. Additionally, technologies developed by our competitors may render our potential product candidates uneconomical or obsolete, and we may not be successful in marketing our product candidates against competitors.
Although we commercially launched Zokinvy following its FDA approval, we have limited marketing and sales experience outside the U.S. If we are unable to establish sales and marketing capabilities or enter into agreements with third parties to market and sell our product candidates, we may be unable to generate any revenue.
Although certain of our employees may have marketed, launched and sold other pharmaceutical products in the U.S. in the past while employed at other companies, we have limited experience selling and marketing our product candidates outside the U.S.. To successfully commercialize Zokinvy and additional products that may result from our development programs, we will need to invest in and expand these capabilities, either on our own or with others, which would be expensive, difficult and time consuming. Any failure or delay in the timely development of our internal commercialization capabilities could adversely impact the potential for success of our products.
Further, given our, limited experience in marketing and selling biopharmaceutical products outside the U.S., we may rely on future collaborators to commercialize our products. If collaborators do not commit sufficient resources to commercialize our future products and we are unable to develop the necessary marketing and sales capabilities on our own, we will be unable to generate sufficient product revenue to sustain or grow our business. We may be competing with companies that currently have extensive and well-funded marketing and sales operations, in particular in the markets our product candidates are intended to address. Without appropriate capabilities, whether directly or through third-party collaborators, we may be unable to compete successfully against these more established companies. In addition, we have established an expanded access program in order to make Zokinvy available for patients with progeria, which requires additional resources and costs to support.
The commercial success of any of our current or future product candidates will depend upon the degree of market acceptance by physicians, patients, third-party payors, and others in the medical community.
Even with the approvals from the FDA and comparable foreign regulatory authorities, the commercial success of our products will depend in part on the health care providers, patients, and third-party payors accepting our product candidates
as medically useful, cost-effective, and safe. Any product that we bring to the market may not gain market acceptance by physicians, patients, third-party payors and other health care providers. The degree of market acceptance of any of our products will depend on a number of factors, including without limitation:
•the timing of our receipt of any marketing and commercialization licensures;
•the terms of any licensures and the countries in which licensures are obtained;
•the safety, efficacy, purity and/or potency of the product as demonstrated in clinical studies and potential advantages over competing treatments;
•the prevalence and severity of the disease and any side effects;
•the clinical indications for which approval is granted, including any limitations or warnings contained in a product’s approved labeling;
•the convenience and ease of administration;
•the cost of treatment;
•the willingness of the patients and physicians to accept these therapies or any new methods of administration;
•the marketing, sales and distribution support for the product;
•the publicity concerning our products or competing products and treatments;
•the success of our physician education programs;
•availability of alternative effective treatments for the disease indications our product candidates are intended to treat and the relative risks, benefits and costs of those treatments; and
•the pricing and availability of third-party insurance coverage and reimbursement.
Even if a product displays a favorable safety, efficacy, purity and/or potency profile upon approval, market acceptance of the product remains uncertain. Efforts to educate the medical community and third-party payors on the benefits of the products may require significant investment and resources and may never be successful. If our products fail to achieve an adequate level of acceptance by physicians, patients, third-party payors, and other health care providers, we will not be able to generate sufficient revenue to become or remain profitable.
Failure to obtain or maintain adequate reimbursement or insurance coverage for new or current products could limit our ability to market those products and decrease our ability to generate revenue.
We cannot be sure that reimbursement will be available for our products and, if coverage and reimbursement are available, what the level of reimbursement will be. Coverage may also be more limited than the indications for which the product is approved by the FDA or comparable foreign regulatory authorities. Reimbursement may impact the demand for, or the price of, our products.
The pricing, coverage and reimbursement of our products must be sufficient to support our commercial efforts and other development programs and the availability and adequacy of coverage and reimbursement by governmental and private payors are essential for most patients to be able to afford expensive treatments, particularly in Orphan Drug designated indications where the eligible patient population is small. Sales of our products will depend substantially, both domestically and abroad, on the extent to which the costs of our product candidates will be paid for or reimbursed by health maintenance, managed care, pharmacy benefit and similar healthcare management organizations, or government authorities, private health insurers, and other third-party payors. If coverage and reimbursement are not available, or are available only in limited amounts, we may have to subsidize products or we may not be able to successfully commercialize our products. For example, Zokinvy for patients with HGPS and processing-deficient PL is provided under an expanded access program may not result in reimbursement.
In addition, there is significant uncertainty related to the insurance coverage and reimbursement for newly approved products. In the United States, the principal decisions about coverage and reimbursement for new drugs are typically made by the Centers for Medicare & Medicaid Services (CMS), an agency within the U.S. Department of Health and Human Services (HHS), as CMS decides whether and to what extent a new drug will be covered and reimbursed under Medicare. Private payors often follow the coverage reimbursement policies established by CMS to a substantial degree. It is difficult to predict what CMS and other payors will decide with respect to coverage for products such as ours and what reimbursement our products may receive.
The process for determining whether a third-party payor will provide coverage for a drug product typically is separate from the process for setting the price of a drug product or for establishing the reimbursement rate that the payor will pay for the drug product once coverage is approved. Third-party payors may limit coverage to specific drug products on an approved list, also known as a formulary, which might not include all of the FDA-approved drugs for a particular indication. A decision by a third-party payor not to cover Zokinvy or any of our product candidates once approved could reduce utilization of such products and have a material adverse effect on our sales, results of operations and financial condition. Moreover, a third-party payor’s decision to provide coverage for a drug product does not imply that adequate reimbursement will be available. Adequate third-party payor reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development. Additionally, coverage and reimbursement for drug products can differ significantly from payor to payor. One third-party payor’s decision to cover a particular medical product or service does not ensure that other payors will also provide coverage for the medical product or service or will provide coverage at an adequate reimbursement rate. As a result, the coverage determination process will require us to provide scientific and clinical support for the use of our products to each payor separately and will be a time-consuming process.
Third-party payors are increasingly challenging the prices charged for medical products and services, examining the medical necessity and reviewing the cost-effectiveness of drug products and medical services, in addition to questioning safety and efficacy. If third-party payors do not consider our products to be cost-effective compared to other available therapies, they may not cover our products after FDA approval or, if they do, the level of payment may not be sufficient to allow us to sell our products at a profit. Additionally, coverage policies and third‑party reimbursement rates may change at any time. Therefore, even if favorable coverage and reimbursement status is attained for one or more of our products, less favorable coverage policies and reimbursement rates may be implemented in the future. If we are unable to obtain and maintain sufficient third‑party coverage and adequate reimbursement, the commercial success of our products may be greatly hindered and our financial condition and results of operations may be materially and adversely affected.
Outside the United States, international operations are generally subject to extensive governmental price controls and other price-restrictive regulations, and we believe the increasing emphasis on cost-containment initiatives in Europe, Canada, and other countries has and will continue to put pressure on the pricing and usage of products. In many countries, the prices of products are subject to varying price control mechanisms as part of national health systems. Price controls or other changes in pricing regulation could restrict the amount that we are able to charge for our products. Accordingly, in markets outside the United States, the potential revenue may be insufficient to generate commercially reasonable revenue and profits.
Moreover, increasing efforts by governmental and third-party payors in the United States and abroad to limit or reduce healthcare costs may result in restrictions on coverage and the level of reimbursement for new products and, as a result, inadequate coverage or payment for our products.
In the EU, coverage and reimbursement status of our products, if approved, are provided for by the national laws of the EU Member States. In the EU, pricing and reimbursement schemes vary widely from Member State to Member State. Some Member States provide that products can only be effectively marketed after a reimbursement price has been agreed. Some Member States may require the completion of additional studies in order to compare the cost-effectiveness of a particular drug candidate to currently available therapies (so called health technology assessment, or HTA) in order to obtain reimbursement or pricing approval. EU Member States may approve a specific price for a product or it may instead adopt a system of direct or indirect controls on the profitability of the Company placing the product on the market. Other Member States may allow companies to fix their own prices for products, but monitor and control prescription volumes and issue guidance to physicians to limit prescriptions. Recently, many countries in the EU have increased the amount of discounts required on pharmaceuticals and these efforts could continue as countries attempt to manage healthcare expenditures, especially in light of the severe fiscal and debt crises experienced by many countries in the EU. The downward pressure on healthcare costs in general, particularly prescription drugs, has become intense. As a result, increasingly high barriers are being erected to the entry of new products. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various EU Member States, and parallel trade (arbitrage between low-priced and high-priced Member States), can further reduce prices. There can be no assurance that any country that has price controls or reimbursement limitations for drug products will allow favorable reimbursement and pricing arrangements for any of our products, if approved in those countries.
We expect to experience pricing pressures in connection with products due to increased and continued efforts to limit or reduce healthcare spending. The downward pressure on healthcare costs in general, particularly prescription drugs, has and
is expected to continue to increase in the future. As a result, profitability of our products may be more difficult to achieve even if they receive regulatory approval.
We intend to rely on a combination of exclusivity from Orphan Drug designation as well as patent rights for our product candidates and any future product candidates. If we are unable to obtain or maintain exclusivity from the combination of these approaches, we may not be able to compete effectively in our markets.
Our business strategy is to focus on product candidates for which Orphan Drug designation may be obtained in the major markets of the world. In addition, we rely or will rely upon a combination of patents, trade secret protection, and confidentiality agreements to protect the intellectual property related to our technologies and product candidates. Our success depends in large part on our and our licensors’ ability to obtain regulatory exclusivity and maintain patent and other intellectual property protection in the United States and in other countries with respect to our proprietary technology and products.
Under the Orphan Drug Act, the FDA may designate a product as an Orphan Drug if it is intended to treat a rare disease or condition, defined as a patient population of fewer than 200,000 in the United States, or a patient population greater than 200,000 in the United States where there is no reasonable expectation that the cost of developing the product will be recovered from sales in the United States. In the EU, the EMA’s Committee for Orphan Medicinal Products (COMP) grants Orphan Drug designation to promote the development of products that are intended for the diagnosis, prevention, or treatment of a life-threatening or chronically debilitating condition affecting not more than five in 10,000 persons in the EU. Additionally, designation is granted for products intended for the diagnosis, prevention, or treatment of a life-threatening, seriously debilitating or serious and chronic condition when, without incentives, it is unlikely that sales of the drug in the EU would be sufficient to justify the necessary investment in developing the drug or biological product or where there is no satisfactory method of diagnosis, prevention, or treatment, or, if such a method exists, the medicine must be of significant benefit to those affected by the condition. In the United States, Orphan Drug designation entitles a party to financial incentives such as opportunities for grant funding towards clinical trial costs, tax advantages, and user-fee waivers. In addition, if a product receives the first FDA approval for the indication for which it has Orphan Drug designation, the product is entitled to Orphan Drug exclusivity, which means the FDA may not approve any other application to market the same product for the same indication for a period of seven years, except in limited circumstances, such as a showing of clinical superiority over the product with orphan exclusivity or where the manufacturer is unable to assure sufficient product quantity.
Congress is considering updates to the Orphan Drug provisions of the FDCA in response to a recent decision by the U.S. Court of Appeals for the 11th Circuit. Any changes to the Orphan Drug provisions could change our opportunities for, or likelihood of success in obtaining, Orphan Drug exclusivity and would materially adversely affect our business, financial condition, results of operations, cash flows and prospects.
In the EU, Orphan Drug designation entitles a party to financial incentives such as reduction of fees or fee waivers and ten years of market exclusivity following drug or biological product approval. This period may be reduced to six years if the Orphan Drug designation criteria are no longer met, including where it is shown that the product is sufficiently profitable not to justify maintenance of market exclusivity.
Because the extent and scope of patent protection for our products may in some cases be limited, Orphan Drug designation is especially important for our products for which Orphan Drug designation may be available. For eligible products, we plan to rely on the exclusivity period under the Orphan Drug Act to maintain a competitive position. If we do not obtain or maintain Orphan Drug exclusivity for our drug and biological products that do not have broad patent protection, our competitors may then sell the same drug to treat the same condition sooner than if we had obtained Orphan Drug exclusivity and our revenue will be reduced.
Even though we have Orphan Drug designations for each of our development programs in the United States and the EU, we may not be the first to obtain marketing approval for any particular orphan indication due to the uncertainties associated with developing pharmaceutical products. Further, even if we obtain Orphan Drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different products with different active moieties can be approved for the same condition. Even after an Orphan Drug is approved, the FDA or EMA can subsequently approve the same product with the same active moiety for the same condition if the FDA or EMA concludes that the later product is safer, more effective, or makes a major contribution to patient care. Orphan Drug designation neither shortens the development time or regulatory review time of a product candidate nor gives the product candidate any advantage in the regulatory review or approval process.
We have sought to protect our proprietary position by filing patent applications in the United States and abroad related to our product candidates that are important to our business. This process is expensive and time consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain and involves complex legal and factual questions for which legal principles remain unsolved. The patent applications that we own or in-licensed may fail to result in issued patents with claims that cover our product candidates in the United States or in other foreign countries. There is no assurance that all potentially relevant prior art relating to our patents and patent applications has been found, which can invalidate a patent or prevent a patent from issuing from a pending patent application. Even if patents do successfully issue, and even if such patents cover our product candidates, third parties may challenge their validity, enforceability, or scope, which may result in such patents being narrowed, found unenforceable or invalidated. Furthermore, even if they are unchallenged, our patents and patent applications may not adequately protect our intellectual property, provide exclusivity for our product candidates, or prevent others from designing around our claims. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business.
We, independently or together with our licensors, have filed several patent applications covering various aspects of our product candidates. We cannot offer any assurances about which, if any, patents will issue, the breadth of any such patent or whether any issued patents will be found invalid and unenforceable or will be threatened by third parties. Any successful opposition to these patents or any other patents owned by or licensed to us after patent issuance could deprive us of rights necessary for the successful commercialization of any product candidates that we may develop. Further, if we encounter delays in regulatory approvals, the period of time during which we could market a product candidate under patent protection could be reduced. Although we have licensed a number of patents covering methods of use and certain compositions of matter, we do not have complete patent protection for our product candidates. For example, in connection with our arbitration with The Progeria Research Foundation, Inc. (PRF) asserting two claims under a May 15, 2018 Collaboration and Supply Agreement with PRF, PRF is indirectly challenging the validity of our in-license of certain patents covering the methods of treating HGPS and progeroid laminopathies. Likewise, most of the patents or applications covering products that we have licensed in from Stanford have limited protection outside of the United States. Therefore, a competitor could develop the same or similar product that may compete with our product candidate.
Certain of our product licenses are limited to specified indications or therapeutic areas which may result in the same compound being developed and commercialized by a third party whom we have no control over or rights against. This may result in safety data, pricing or off-label uses from that third party’s product that may negatively affect the development and commercialization of our product candidates. If we cannot obtain and maintain effective protection of exclusivity from our regulatory efforts and intellectual property rights, including patent protection, for our product candidates, we may not be able to compete effectively, and our business and results of operations would be harmed.
The annual reassessment by the EMA of the risk-benefit balance, including information on the safe and effective use, for Zokinvy may not be positive, leading to a variation, suspension, revocation of our MA or fulfillment of additional specific obligations.
On July 20, 2022, we announced that the EC granted an MA under the exceptional circumstances procedure for Zokinvy to treat patients aged 12 months and older with HGPS and PL. The EC authorization follows the positive opinion granted by the Committee for Medicinal Products for Human Use (CHMP) in May 2022 which found that the risk-benefit balance for Zokinvy is favorable to recommend the granting of a MA although the rarity of the disease means that it was not possible to obtain complete information on Zokinvy during the assessment process. As a result, the MA was issued under the exceptional circumstances procedure and subject to the EMA’s continued review on an annual basis of new efficacy and safety information which may become available.
Under Article 14(8) Regulation (EC) No 726/2004, products for which the applicant can demonstrate that comprehensive data (in line with the requirements laid down in Annex I of Directive 2001/83/EC, as amended) cannot be provided (due to specific reasons foreseen in the legislation) might be eligible for marketing authorization under exceptional circumstances. This type of authorization is reviewed annually to reassess the risk-benefit balance. The fulfillment of any specific procedures/obligations imposed as part of the marketing authorization under exceptional circumstances is aimed at the provision of information on the safe and effective use of the product.
It is possible that the annual reassessment by the EMA of the risk-benefit balance including information on the safe and effective use for Zokinvy may not be positive. This could lead to the variation, suspension, revocation of our MA for Zokinvy in the EU, or lead to additional specific commitments or conditions being fulfilled.
We may not have sufficient patent term protections for our products to effectively protect our business.
Patents have a limited term. In the United States, the statutory expiration of a patent is generally 20 years after it is filed. Although various extensions may be available, the life of a patent, and the protection it affords, is limited. Even if patents covering our product candidates are obtained, once the patent life has expired for a product, we may be open to competition from generic medications. In addition, upon issuance in the United States any patent term can be adjusted based on certain delays caused by the applicant(s) or the United States Patent and Trademark Office (USPTO). For example, a patent term can be reduced based on certain delays caused by the patent applicant during patent prosecution.
Patent term extensions under the Hatch-Waxman Act in the United States and under supplementary protection certificates in the EU may be available to extend the patent or data exclusivity terms of products. With respect to lonafarnib-based regimens, peginterferon lambda and avexitide, a substantial portion of the potential commercial opportunity will likely rely on patent term extensions, and we cannot provide any assurances that any such patent term extensions will be obtained and, if so, for how long. As a result, we may not be able to maintain exclusivity for our products for an extended period after regulatory approval, which would negatively impact our business and results of operations. If we do not have sufficient patent terms or regulatory exclusivity to protect our products, our business and results of operations will be adversely affected.
Patent laws and rule changes could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of our patents or narrow the scope of our patent protection. The laws of foreign countries may not protect our rights to the same extent as the laws of the United States. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. We therefore cannot be certain that it or our licensors were the first to make the invention claimed in our owned and licensed patents or pending applications, or that we or our licensor were the first to file for patent protection of such inventions. Assuming the other requirements for patentability are met, in the United States prior to March 15, 2013, the first to make the claimed invention is entitled to the patent, while outside the United States, the first to file a patent application is entitled to the patent. After March 15, 2013, under the Leahy-Smith America Invents Act (the Leahy-Smith Act) enacted on September 16, 2011, the United States has moved to a first to file system. The Leahy-Smith Act also includes a number of significant changes that affect the way patent applications will be prosecuted and may also affect patent litigation. The effects of these changes are currently unclear as the USPTO must still implement various regulations, the courts have yet to address any of these provisions and the applicability of the act and new regulations on specific patents discussed herein have not been determined and would need to be reviewed. In general, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition.
If we are unable to maintain effective proprietary rights for our product candidates or any future product candidates, we may not be able to compete effectively in our markets.
In addition to the protection afforded by patents, we rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or that we elect not to patent, processes for which patents are difficult to enforce and any other elements of our product candidates' discovery and development processes that involve proprietary know-how, information or technology that is not covered by patents. However, trade secrets can be difficult to protect. We seek to protect our proprietary technology and processes, in part, by entering into confidentiality agreements with our employees, consultants, scientific advisors, and contractors. We also seek to preserve the integrity and confidentiality of our data and trade secrets by maintaining physical security of our premises and physical and electronic security of our information technology systems. While we have confidence in these individuals, organizations and systems, agreements or security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors.
Although we expect all of our employees and consultants to assign their inventions to us, and all of our employees, consultants, advisors, and any third parties who have access to our proprietary know-how, information, or technology to enter into confidentiality agreements, we cannot provide any assurances that all such agreements have been duly executed or that our trade secrets and other confidential proprietary information will not be disclosed or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. Misappropriation or unauthorized disclosure of our trade secrets could impair our competitive position and may have a material adverse effect on our business. Additionally, if the steps taken to maintain our trade secrets are deemed inadequate, we may have insufficient recourse against third parties for misappropriating the trade secret.
Third-party claims of intellectual property infringement may prevent or delay our development and commercialization efforts.
Our commercial success depends in part on our avoiding infringement of the patents and proprietary rights of third parties. There have been many lawsuits and other proceedings involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits, interferences, oppositions, and reexamination proceedings before the USPTO and corresponding foreign patent offices. Numerous U.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields in which we are developing product candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that our product candidates may be subject to claims of infringement of the patent rights of third parties.
Third parties may assert that we are using or exploiting their proprietary technology without authorization. There may be third-party patents or patent applications with claims to materials, formulations, methods of manufacture, or methods for treatment related to the use or manufacture of our product candidates. Even if we conduct freedom to operate analyses, we would expect to do so only with respect to certain of our product candidates as they move through development. Accordingly, there may be third-party patents that would impair our ability to commercialize product candidates and we cannot assure you that we could obtain a license, or even if available, whether such license might be obtained on commercially reasonable terms. Even in those situations where we conduct a freedom to operate analysis, there can be no assurance that we would identify all relevant or necessary patents and patent applications that may apply to the manufacture and commercialization of our product candidates. Because patent applications can take many years to issue, there may be currently pending patent applications that may later result in issued patents that our product candidates may infringe, and if patents issue with respect to any such application and we become aware of such issuance, we would have to determine its impact on our efforts to develop and commercialize our product candidates and the strategy for obtaining a license or contesting any such issued patent.
If any third-party patents were held by a court of competent jurisdiction to cover aspects of our formulations, the manufacturing process of any of our product candidates, methods of use, any molecules formed during the manufacturing process or any final product itself, the holders of any such patents may be able to block our ability to commercialize such product candidate unless we obtained a license under the applicable patents, or until such patents expire or are finally determined to be invalid or unenforceable. Such a license may not be available on commercially reasonable terms, or at all.
If we fail to obtain a license, then parties making claims against us may obtain injunctive or other equitable relief, which could effectively block our ability to further develop and commercialize one or more of our product candidates. Defense of these claims, regardless of their merit, would involve substantial litigation expense and would be a substantial diversion of employee resources from our business. In the event of a successful claim of infringement against us, we may have to pay substantial damages, including treble damages and attorneys’ fees for willful infringement, pay royalties, redesign our infringing products or obtain one or more licenses from third parties, which may be impossible or require substantial time and monetary expenditure.
We may not be successful in meeting our diligence obligations under our existing license agreements necessary to maintain our product candidates' licenses in effect. In addition, if required in order to commercialize our product candidates, we may be unsuccessful in obtaining or maintaining necessary rights to our product candidates through acquisitions and in-licenses.
We currently have rights to the intellectual property, through licenses from third parties and under patents that we do not own, to develop and commercialize our product candidates. Because our programs may require the use of proprietary rights held by third parties, the growth of our business will likely depend in part on our ability to maintain in effect these proprietary rights. For example, we have certain specified diligence obligations under our license agreement for lonafarnib. We may not be able to achieve the required diligence milestones in a timely manner, which may result in the license
agreement being terminated, and we may be unable to successfully negotiate an extension or waiver of those termination rights. Any termination of license agreements with third parties with respect to our product candidates would be expected to negatively impact our business prospects.
We may be unable to acquire or in-license any compositions, methods of use, processes, or other third-party intellectual property rights from third parties that we identify as necessary for our product candidates. The licensing and acquisition of third-party intellectual property rights is a competitive area, and a number of more established companies are also pursuing strategies to license or acquire third-party intellectual property rights that we may consider attractive. These established companies may have a competitive advantage over us due to their size, cash resources, and greater clinical development and commercialization capabilities. In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. Even if we are able to license or acquire third-party intellectual property rights that are necessary for our product candidates, there can be no assurance that they will be available on favorable terms.
We collaborate with U.S. and foreign academic institutions to identify product candidates, accelerate our research and conduct development. Typically, these institutions have provided us with an option to negotiate an exclusive license to any of the institution’s rights in the patents or other intellectual property resulting from the collaboration. Regardless of such option, we may be unable to negotiate a license within the specified timeframe or under terms that are acceptable to us. If we are unable to do so, the institution may offer the intellectual property rights to other parties, potentially blocking our ability to pursue a program of interest to us.
If we are unable to successfully obtain and maintain rights to required third-party intellectual property, we may have to abandon development of that product candidate or pay additional amounts to the third party, and our business and financial condition could suffer.
Our product candidates marketed under NDAs may be subject to generic competition.
Under the Hatch-Waxman Act, a pharmaceutical manufacturer may file an abbreviated new drug application (ANDA) seeking approval of a generic copy of an approved innovator product marketed under an NDA. Generally, in place of clinical studies intended to demonstrate safety or effectiveness, an ANDA applicant usually needs only to submit data demonstrating that its product has the same active ingredient(s), strength, dosage form, route of administration and that it is bioequivalent to the branded product. Under the Hatch-Waxman Act, a manufacturer may also submit an NDA under section 505(b)(2) that references the FDA’s finding of safety and effectiveness of a previously approved drug. A 505(b)(2) NDA product may be for a new or improved version of the original innovator product.
Innovative small molecule drugs may be eligible for certain periods of regulatory exclusivity. The FDCA provides a period of five years of non-patent exclusivity for a new drug containing a new chemical entity (NCE). During the exclusivity period, the FDA may not accept for review an ANDA or a 505(b)(2) NDA submitted by another company for another version of such product candidate where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement. The FDCA also provides three years of marketing exclusivity for an NDA, 505(b)(2) NDA or supplement to an approved NDA if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example, for new indications, dosages or strengths of an existing product candidate. This three-year exclusivity covers only the conditions associated with the new clinical investigations and does not prohibit the FDA from approving ANDAs for product candidates containing the original active agent for other conditions of use. Five-year and three-year exclusivity will not delay the submission or approval of a full NDA. However, an applicant submitting a full NDA would be required to conduct or obtain a right of reference to all of the nonclinical studies and adequate and well-controlled clinical trials necessary to demonstrate safety and effectiveness.
Manufacturers may seek to launch these generic drugs following the expiration of the marketing exclusivity period, even if we still have patent protection for our drug. Competition that our drug candidates may face from generic versions of our drug candidates could materially and adversely impact our future revenue, profitability and cash flows and substantially limit our ability to obtain a return on the investments we have made in those drug candidates. Our future revenues, profitability and cash flows could also be materially and adversely affected and our ability to obtain a return on the investments we have made in those drug candidates may be substantially limited if our drug candidates, if and when approved, are not afforded the appropriate periods of non-patent exclusivity.
In addition to the benefits of regulatory exclusivity, an innovator NDA holder may have patents claiming the active ingredient, product formulation or an approved use of the drug, which would be listed with the product in the FDA publication, “Approved Drug Products with Therapeutic Equivalence Evaluations,” known as the “Orange Book.” If there are patents listed in the Orange Book, a generic applicant that seeks to market its product before expiration of the patents must include in the ANDA or 505(b)(2) what is known as a “Paragraph IV certification,” challenging the validity or enforceability of, or claiming non-infringement of, the listed patent or patents. Notice of the certification must be given to the innovator, too, and if within 45 days of receiving notice the innovator sues to protect its patents, approval of the ANDA is stayed for 30 months, or as lengthened or shortened by the court.
If there are patents listed for our products in the Orange Book after approval by the FDA, ANDAs and 505(b)(2) NDAs with respect to those products would be required to include a certification as to each listed patent indicating whether the ANDA applicant does or does not intend to challenge the patent. We cannot predict whether any patents issuing from our pending patent applications will be eligible for listing in the Orange Book, how any generic competitor would address such patents, whether we would sue on any such patents, or the outcome of any such suit.
We may not be successful in securing or maintaining proprietary patent protection in the United States and/or in other countries for products and technologies we develop or license. Moreover, if any patents that are granted and listed in the Orange Book are successfully challenged by way of a Paragraph IV certification and subsequent litigation, the affected product could more immediately face generic competition and its sales would likely decline materially. Should sales decline, we may have to write off a portion or all of the intangible assets associated with the affected product and our results of operations and cash flows could be materially and adversely affected.
Our biological product candidates for which we intend to seek licensure may face competition from biosimilars.
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, collectively the ACA, includes a subtitle called the Biologics Price Competition and Innovation Act of 2009, or BPCIA, which created an abbreviated licensure pathway for biological products that are biosimilar to or interchangeable with an FDA-licensed reference biological product. Under the BPCIA, an application for a biosimilar product may not be submitted to the FDA until four years following the date that the reference product was first licensed by the FDA. In addition, the licensure of a biosimilar product may not be made effective by the FDA until 12 years from the date on which the reference product was first licensed. During this 12-year period of exclusivity, another company may still market a competing version of the reference product if the FDA approves a full BLA for the competing product containing the sponsor’s own preclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity and potency of their product. The law is complex and is still being interpreted and implemented by the FDA. As a result, its ultimate impact, implementation, and meaning are subject to uncertainty. While it is uncertain when such processes intended to implement BPCIA may be fully adopted by the FDA, any such processes could have an adverse effect on the future commercial prospects for our product candidates.
There is a risk that any product candidates we may develop that are licensed as a biological product under a BLA would not qualify for the 12-year period of exclusivity or that this exclusivity could be shortened due to congressional action or otherwise, or that the FDA will not consider any product candidates we may develop to be reference products for competing products, potentially creating the opportunity for competition sooner than anticipated. In addition, the approval of a biologic product biosimilar to one of our products could have a material adverse impact on our business as it may be significantly less costly to bring to market and may be priced significantly lower than our products. Moreover, the extent to which a biosimilar, once approved, will be substituted for any one of the reference products in a way that is similar to traditional generic substitution for non-biological products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing.
Other aspects of the BPCIA, some of which may impact the BPCIA exclusivity provisions, have also been the subject of recent litigation, including litigation challenging the constitutionality of the ACA. For example, in December 2018, a federal district court ruled that the ACA, without the “individual mandate” penalty (which was repealed by Congress as part of the Tax Cuts and Jobs Act), is unconstitutional in its entirety. In December 2019, the U.S. Court of Appeals for the 5th Circuit upheld the district court ruling that the individual mandate provisions are unconstitutional and remanded the case back to the district court for further analysis of whether such provisions could be severed from the remainder of the ACA. On June 17, 2021, the U.S. Supreme Court dismissed the case without specifically ruling on the constitutionality of the ACA. There may, however, be other efforts to challenge, repeal, or replace the ACA in the future. We continue to evaluate the effect that the ACA and its possible repeal and replacement has (or may have) on our business and exclusivity under the BPCIA. It is uncertain the extent to which any such changes may impact our business or financial condition.
The patent protection and patent prosecution for some of our product candidates is dependent on third parties.
While we normally seek and gain the right to fully prosecute the patents relating to our product candidates, there may be times when patents relating to our product candidates are controlled by our licensors. This is the case with our agreements with Stanford, of whom is primarily responsible for the prosecution of patents and patent applications licensed to us under the applicable collaboration agreements. If they or any of our future licensors fail to appropriately and broadly prosecute and maintain patent protection for patents covering any of our product candidates, our ability to develop and commercialize those product candidates may be adversely affected and we may not be able to prevent competitors from making, using, importing, and selling competing products. In addition, even where we now have the right to control patent prosecution of patents and patent applications, we have licensed from third parties, we may still be adversely affected or prejudiced by actions or inactions of our licensors in effect from actions prior to us assuming control over patent prosecution.
If we fail to comply with obligations in the agreements under which we license intellectual property and other rights from third parties or otherwise experience disruptions to our business relationships with our licensors, we could lose license rights that are important to our business.
We are a party to a number of intellectual property license and supply agreements that are important to our business and expects to enter into additional license agreements in the future. Our existing agreements impose, and we expect that future license agreements will impose, various diligence, milestone payment, royalty, purchasing, supply and other obligations on us. If we fail to comply with our obligations under these agreements, including those relating to our entering the Chapter 11 Cases, our agreements may be subject to termination by the licensor, in which event we would not be able to develop, manufacture or market products covered by the license or subject to supply commitments.
Although we are not currently involved in any intellectual property litigation, we may be involved in lawsuits to protect or enforce our patents or the patents of our licensors, which could be expensive, time consuming, and unsuccessful.
Competitors may infringe our patents or the patents of our licensors. Although we are not currently involved in any intellectual property litigation, if we or one of our licensing partners were to initiate legal proceedings against a third party to enforce a patent covering one of our product candidates, the defendant could counterclaim that the patent covering our product candidates is invalid and/or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity and/or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness, or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement, during prosecution. The outcome following legal assertions of invalidity and unenforceability is unpredictable.
Interference proceedings provoked by third parties or brought by us or declared by the USPTO may be necessary to determine the priority of inventions with respect to our patents or patent applications or those of our licensors. An unfavorable outcome could require us to cease using the related technology or to attempt to license rights to it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license on commercially reasonable terms. Our defense of litigation or interference proceedings may fail and, even if successful, may result in substantial costs and distract our management and other employees. In addition, the uncertainties associated with litigation could have a material adverse effect on our ability to raise the funds necessary to continue our clinical trials, continue our research programs, license necessary technology from third parties, or enter into development partnerships that would help us bring our product candidates to market.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions, or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our common stock.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties or that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
We employ individuals who were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we have written agreements and make every effort to ensure that our employees, consultants and independent contractors do not use the proprietary information or
intellectual property rights of others in their work forums, and we are not currently subject to any claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties, we may in the future be subject to such claims. Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel, which could adversely impact our business. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting, and defending patents on product candidates in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States can be less extensive than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and may also export infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States. These products may compete with our products and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, trade secrets, and other intellectual property protection, particularly those relating to biotechnology products, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions, whether or not successful, could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate, and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
Risks Related to our Business Operations
Our future success depends in part on our ability to attract, retain, and motivate qualified personnel.
On December 14, 2022, David Cory resigned as our President and CEO and a member of our Board, and on January 3, 2023, Sriram Ryali notified the Company of his resignation as the Company’s CFO, effective January 20, 2023, and on February 17, 2023, Erik Atkisson resigned as our General Counsel, Corporate Secretary and Chief Compliance Officer. The Company has also experienced recent turnover in its financial reporting function. Additionally, effective February 2, 2024, Eldon Mayer resigned as the Company’s Chief Commercial Officer.
In June 2023, the Board appointed Dr. David Apelian as the Company’s CEO. We are highly dependent on Dr. Apelian, who has remained a member of the Board following his appointment. On April 13, 2023, Eiger announced the appointments of William G. Kachioff as Chief Financial Officer, and James A. Vollins as General Counsel, Chief Compliance Officer and Corporate Secretary, to the Company's management team.
Recruiting and retaining other qualified employees, consultants, and advisors for our business, including scientific and technical personnel, will also be critical to our success. There is currently a shortage of highly qualified personnel in our industry, which is likely to continue. As a result, competition for personnel is intense and the turnover rate can be high. We may not be able to attract and retain personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for individuals with similar skill sets. In addition, failure to succeed in development and commercialization of our product candidates may make it more challenging to recruit and retain qualified personnel. The inability to recruit and retain qualified personnel, the loss of the services of Dr. Apelian or other members of our management team, or the failure to hire other qualified management personnel, may impede the progress of our research, development, and commercialization objectives and would negatively impact our ability to succeed.
Failure to comply with existing or future laws and regulations related to privacy or data security could lead to government enforcement actions (which could include civil or criminal fines or penalties), private litigation, other liabilities, and/or adverse publicity. Compliance or the failure to comply with such laws could increase the costs of our products and services, could limit their use or adoption, and could otherwise negatively affect our operating results and business.
Regulation of data processing is evolving as federal, state, and foreign governments continue to adopt new, or modify existing, laws and regulations addressing data privacy and security, and the collection, processing, storage, transfer, and use of data. We and our partners may be subject to current, new, or modified federal, state, and foreign data protection laws and regulations (e.g., laws and regulations that address data privacy and data security including, without limitation, health data). These new or proposed laws and regulations are subject to differing interpretations and may be inconsistent among jurisdictions, and guidance on implementation and compliance practices are often updated or otherwise revised, which adds to the complexity of processing personal data. These and other requirements could require us or our partners to incur additional costs to achieve compliance, limit our competitiveness, necessitate the acceptance of more onerous obligations in our contracts, restrict our ability to use, store, transfer, and process data, impact our or our partners’ ability to process or use data in order to support the provision of our products or services, affect our or our partners’ ability to offer our products and services in certain locations, or cause regulators to reject, limit or disrupt our clinical trial activities.
In the United States, numerous federal and state laws and regulations, including state data breach notification laws, state health information privacy laws, and federal and state consumer protection laws (e.g., Section 5 of the Federal Trade Commission Act) that govern the collection, use, disclosure, and protection of health-related and other personal information apply to our operations or the operations of our partners. In addition, we may receive unintended health information in error from third parties (including research institutions from which we may obtain clinical trial data) that are subject to privacy and security requirements under the federal Health Insurance Portability and Accountability Act of 1996 (HIPAA) as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (HITECH). Depending on the facts and circumstances, we could be subject to significant penalties if we obtain, use, or disclose individually identifiable health information in a manner that is not authorized or permitted by HIPAA. On December 10, 2020, the Office of Civil Rights within the Department of Health and Human Services issued proposed revisions to the HIPAA Privacy Rule aimed at reducing regulatory burdens that may exist in discouraging coordination of care and patient access to their health information, among other changes. While a final rule has not yet been issued, if adopted, these proposed changes may require us to update our policies and procedures to comply with the new requirements. In particular, several state laws have recently been passed or amended to significantly expand privacy rights and obligations of businesses that process personal information, including identifiable sensitive health information. For example, in June 2018, California enacted the California Consumer Privacy Act (CCPA), which went into effect on January 1, 2020. The CCPA gives California residents expanded rights to access and require deletion of their personal information, opt out of certain personal information sharing, and receive detailed information about how their personal information is used. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches that may increase data breach litigation. The California Privacy Rights Act of 2020 (CPRA), which expands the CCPA, was passed in the election on November 3, 2020. The CPRA will, among other things, give consumers the ability to limit use of information deemed to be sensitive, increase the maximum penalties for violations concerning consumers under age 16, and establish the California Privacy Protection Agency to implement and enforce the new law and impose administrative fines. Following the CPRA, Virginia and Colorado have enacted similar, but not completely consistent, comprehensive privacy legislation that will also go into effect in January and July 2023, respectively. Many other states are considering similar legislation.
Aspects of these new state privacy laws, and their interpretation and enforcement, remain uncertain. The potential effects of these new and evolving state privacy laws are far-reaching and may require us to modify our data processing practices and policies and to incur substantial costs and expenses in an effort to comply. Although the California, Virginia and Colorado laws include exemptions for certain clinical trials data and HIPAA protected health information, the law may increase our compliance costs and potential liability with respect to other personal information we collect about California residents, and those health-data related exceptions may evolve through amendment or regulatory interpretation. The state privacy law developments, moreover, have prompted a number of additional proposals for new federal and state privacy legislation that, if passed, could increase our potential liability, increase our compliance costs and adversely affect our business.
Foreign data protection laws, including, without limitation, the EU GDPR that took effect in May 2018, and EU Member State data protection legislation, may also apply to health-related and other personal information obtained from individuals. The EU GDPR has been transposed into the national laws of the United Kingdom by virtue of section 3 of the EU (Withdrawal) Act 2018 as the UK GDPR (together, the EU GDPR and the UK GDPR, the GDPR). The EU GDPR has direct effect in all EU Member States and has extraterritorial effect where organizations outside of the EU process personal information of individuals in the EU in relation to the offering of goods or services to those individuals (targeting test) or
the monitoring of their behavior (monitoring test). The UK GDPR has a similar extraterritorial test for organizations outside of the UK processing personal information of individuals in the UK in relation to the offering of goods or services to those individuals or the monitoring of their behavior. As such, the GDPR applies to us to the extent we are established in an EU Member State or the UK or we meet the requirements of either the targeting test or the monitoring test. These laws impose strict obligations on businesses, including to: (i) implement administrative, physical, technical, and organizational safeguards to protect personal information; (ii) establish an appropriate and valid legal basis for processing personal information; (iii) comply with accountability transparency requirements regarding the processing of personal information, which require controllers to demonstrate and record compliance with the GDPR and to provide more detailed information to data subjects regarding processing; (iv) comply with data protection rights of data subjects including a right of access to and rectification of personal information, a right to obtain restriction of processing or to object to processing of personal information, a right to ask for a copy of personal information to be provided to a third party in a useable format and erasing personal information in certain circumstances; (v) report certain personal data breaches to the relevant supervisory authority without undue delay (and no later than 72 hours, where feasible); (vi) obtain explicit consent for collection of sensitive personal information such as health data; and (vii) consider data protection as any new products or services are developed and to limit the amount of personal information processed.
The EU GDPR restricts the transfer of personal information from the European Economic Area (EEA) to the United States and other countries that the European Commission does not recognize as having “adequate” data protection laws unless the parties to the transfer have implemented an appropriate data transfer mechanism in accordance with the EU GDPR. The UK GDPR has similar restrictions on transfers of personal information from the United Kingdom to countries that the UK does not recognize as having “adequate” data protection laws in the United Kingdom (as discussed below) and Switzerland impose similar restrictions. One of the primary mechanisms allowing United States companies to import personal information from Europe had been certification to the EU-U.S. Privacy Shield and Swiss-U.S. Privacy Shield frameworks administered by the United States Department of Commerce. However, in July 2020, the Court of Justice of the EU (CJEU) invalidated the EU-U.S. Privacy Shield, and subsequent regulatory guidance required additional compliance efforts to analyze international data flows and take steps to ensure adequate protections for personal data transferred to the United States and other certain jurisdictions, including by implementing supplementary measures that provide privacy protections in addition to those provided under the Standard Contractual Clauses (SCCs). Moreover, new versions of the European Commission’s Standard Contractual Clauses, now the primary mechanism for the lawful transfer of personal information transfers from Europe and/or the United Kingdom to the United States or other countries, have been released requiring additional compliance and implementation efforts. The United Kingdom is also expected to publish its own set of SCCs in early 2022 for transfers of personal data outside of the United Kingdom. Similarly, the Swiss Federal Data Protection and Information Commissioner announced that the Swiss-U.S. Privacy Shield Framework is inadequate for personal information transfers from Switzerland to the United States in light of the CJEU’s July 2020 decision, and also raised questions about the viability of the older version of the Standard Contractual Clauses. As such, any transfers by us or our vendors of personal information from Europe may not comply with European data protection law, may increase our exposure to the EU GDPR’s heightened sanctions for violations of its cross-border data transfer restrictions and may reduce demand from companies subject to European data protection laws.
Moreover, where we rely on SCCs, we must now evaluate and implement supplementary measures that provide privacy protections additional to those provided under SCCs. This evaluation will, in particular, include an assessment as to whether the types of personal data transferred pursuant to SCCs may be subject to government surveillance in the data importer’s country and an assessment as to whether the data importer can meet its contractual obligations under the SCCs. This may have implications for our cross-border data flows and may result in compliance costs. Inability to import personal information from the EEA, United Kingdom or Switzerland may also restrict our clinical trial activities in Europe; limit our ability to collaborate with CROs, service providers, contractors and other companies subject to European data protection laws; and require us to increase our data processing capabilities in Europe at significant expense.
Additionally, other countries outside of Europe continue to enact or are considering enacting similar cross-border data transfer restrictions and laws requiring local data residency, which could increase the cost and complexity of delivering our services and operating our business. For example, Brazil enacted the General Data Protection Law (Lei Geral de Proteção de Dados Pessoais or LGPD) (Law No. 13,709/2018), which broadly regulates the processing of personal information and imposes compliance obligations and penalties comparable to those of the EU GDPR.
Under the EU GDPR, regulators may impose substantial fines and penalties for non-compliance. Companies that violate the EU GDPR can face fines of up to the greater of 20 million Euros or 4% of their consolidated worldwide annual turnover (revenue) and restrictions or prohibitions on data processing. The EU also confers a private right of action on data subjects and consumer associations to lodge complaints with supervisory authorities, seek judicial remedies and obtain
compensation for damages resulting from violations of the EU GDPR. The EU GDPR has increased our responsibility and liability in relation to personal information that we process, requiring us to put in place additional mechanisms to ensure compliance with the EU GDPR and other EU and international data protection rules. There may also be a risk that the measures will not be implemented correctly or that individuals within the business will not be fully compliant with the required procedures. Similarly, the UK GDPR introduces fines of up to the greater of £17.5 million or up to 4% of their consolidated worldwide annual turnover (revenue).
Failure to comply with U.S. and international data protection laws and regulations could result in government enforcement actions (which could include civil or criminal penalties, fines or sanctions), private litigation, and/or adverse publicity and could negatively affect our operating results and business. Moreover, patients about whom we or our partners obtain information, as well as the providers who share this information with us, may contractually limit our ability to use and disclose the information. Claims that we have violated individuals’ privacy rights, failed to comply with data protection laws, or breached our contractual obligations related to security or privacy, even if we are not found liable, could be expensive and time-consuming to defend and could result in adverse publicity that could harm our business. Compliance with data protection laws may be time-consuming, require additional resources and could result in increased expenses, reduce overall demand for our products and services and make it more difficult to meet expectations of or commitments to customers or partners.
Any of these matters could materially adversely affect our business, financial condition, or operational results.
Failure in our information technology and storage systems or our security measures, including without limitation, data breaches, or inadequacy of our business continuity and disaster recovery plans and procedures, could significantly disrupt the operation of our business.
Our ability to execute our business plan and maintain operations depends on the continued and uninterrupted performance of our, and our third-party vendors’, information technology (IT) systems, and the availability of data related to our products, services and operations. IT systems and data are vulnerable to risks and damages from a variety of sources, including catastrophe or natural disaster, telecommunications or network failures, malicious human acts, breaches of security, cyber-attacks, loss of power or other natural or man-made events. Moreover, despite network security and back-up measures, we and our vendors frequently defend against and respond to data security attacks and incidents, and vendors’ servers are potentially vulnerable to physical or electronic break-ins, computer viruses, software vulnerabilities, ransomware attacks and similar disruptive problems. If our business continuity and disaster recovery plans and procedures were disrupted, inadequate or unsuccessful in the event of a problem, we could experience a material adverse interruption of our operations.
Specifically, data security breaches, whether inadvertent or intentional, by employees or others, may expose proprietary information, trade secrets, personal information, clinical trial data or other sensitive data to unauthorized persons, impact the integrity, availability or confidentiality of our IT systems or data (including, but not limited to, data loss), or disrupt or interrupt our IT systems or operations. Our partners and vendors face similar risks and any security breach of their systems could adversely affect our security posture. Malicious attacks by third parties are of ever-increasing sophistication and can be made by groups and individuals with a wide range of motives (including, but not limited to, industrial espionage and financial motivation prompted by the enormous growth in ransomware over the past several years) and expertise, including organized criminal groups, “hacktivists,” nation states and others. Foreign, federal, and state laws or regulations allows for the imposition of civil liability, fines and/or corrective action on entities that improperly use or disclose the personal information of individuals, including through a data security breach. Accordingly, data security breaches experienced by us, our collaborators or contractors could lead to significant fines, required corrective action, loss of trade secrets or other intellectual property, or could lead to the public exposure of personally identifiable information (including sensitive personal information) of our employees, collaborators, clinical trial patients, and others. A data security breach or privacy violation that leads to disclosure or modification of or prevents access to personal information, including personally identifiable information, patient information or protected health information, could result in civil liability, harm our reputation, compel us to comply with federal and/or state breach notification laws, subject us to mandatory corrective action, require us to verify the correctness of database contents and otherwise subject us to liability under laws and regulations that protect personal data, resulting in increased costs or loss of revenue. If we are unable to adequately prevent, detect or respond to data security breaches or privacy violations, or implement satisfactory remedial measures in the wake of a data security incident, our operations could be disrupted, and we may suffer civil liability to our customers or individuals, loss of reputation, financial loss and other regulatory penalties because of lost or misappropriated information, including sensitive patient data, or our clinical trials may be adversely impacted from data loss resulting in delayed regulatory approvals or other operational impacts. In addition, these breaches and other inappropriate access events can be
difficult to detect, and any delay in identifying and responding to them may lead to increased harm of the type described above. Moreover, the prevalent use of mobile devices or other remote working activity that access and process confidential information remotely increases the risk of data security breaches, which could lead to the loss of confidential information, trade secrets or other intellectual property. While we have implemented security measures designed to protect our data security and information technology systems, no set of security measures is infallible, and these measures may not prevent such events.
For example, in March 2021, we learned we were the victim of a business email compromise during which an unauthorized party gained access to the email account of an employee in our finance department. The incident resulted in a net loss of approximately $0.3 million. Based on our investigation, the incident was financially motivated and impacted a single email account. In response to the incident, we conducted a review of our corporate information technology and email policies and have implemented additional security measures.
Despite precautionary measures to prevent anticipated and unanticipated problems, including data breaches, there can be no assurance that our efforts to protect our data and information technology systems will prevent breakdowns or breaches in our systems (or that of our third-party providers). Such events could affect our IT systems, sustained or repeated system failures that interrupt our ability to generate, use and maintain data or our IT systems could adversely affect our ability to operate our business and result in increased costs or loss of revenue, other financial and reputational harm to us, theft of trade secrets and other proprietary information, legal claims or proceedings, liability under laws that protect the privacy of personal information and regulatory penalties.
The financial exposure from the events referenced above could either not be insured against or not be fully covered through any insurance that we maintain. There can be no assurance that the limitations of liability in our contracts would be enforceable or adequate or would otherwise protect us from liabilities or damages as a result of the events referenced above.
We may not be successful in any efforts to identify, license, discover, develop or commercialize additional product candidates.
Although a substantial amount of our effort will focus on the continued clinical testing, potential approval, and commercialization of our existing product candidates, the success of our business is also expected to depend in part upon our ability to identify, license, discover, develop, or commercialize additional product candidates. Research programs to identify new product candidates require substantial technical, financial, and human resources. We may focus our efforts and resources on potential programs or product candidates that ultimately prove to be unsuccessful. Our research programs or licensing efforts may fail to yield additional product candidates for clinical development and commercialization for a number of reasons, including but not limited to the following:
•our research or business development methodology or search criteria and process may be unsuccessful in identifying potential product candidates;
•we may not be able or willing to assemble sufficient resources to acquire or discover additional product candidates;
•our product candidates may not succeed in preclinical or clinical testing;
•our potential product candidates may be shown to have harmful side effects or may have other characteristics that may make the products unmarketable or unlikely to receive marketing approval;
•competitors may develop alternatives that render our product candidates obsolete or less attractive;
•product candidates we develop may be covered by third parties’ patents or other exclusive rights;
•the market for a product candidate may change during our program so that such a product may become unreasonable to continue to develop;
•a product candidate may not be capable of being produced in commercial quantities at an acceptable cost, or at all; and
•a product candidate may not be accepted as safe and effective by patients, the medical community, or third-party payors.
If any of these events occur, we may be forced to abandon our development efforts for a program or programs, or we may not be able to identify, license, discover, develop, or commercialize additional product candidates, which would have a material adverse effect on our business and could potentially cause us to cease operations.
Healthcare legislative reform measures may have a material adverse effect on our business and results of operations.
In the United States and other jurisdictions, there have been, and we expect there will continue to be, a number of legislative initiatives and regulatory changes to reform the delivery and payment for healthcare items and services, which could affect our ability to profitably sell our products. For example, in March 2010, the Patient Protection and Affordable Care Act (ACA) was passed, which substantially changed the way health care is financed by both governmental and private insurers, and significantly impacts the U.S. pharmaceutical industry. This law was designed to expand access to health insurance coverage for uninsured and underinsured individuals while at the same time containing overall healthcare costs. The framework of the ACA and other healthcare reforms continues to evolve as a result of executive, legislative, regulatory, and administrative developments. For example, for calendar years 2021 and 2022, the American Rescue Plan Act of 2021 temporarily increased premium tax credit assistance established under the ACA to help eligible individuals cover premiums for health insurance purchased through the health insurance marketplace and removed the 400% federal poverty level limit that otherwise applies for purposes of eligibility to receive premium tax credits. The Inflation Reduction Act of 2022 (IRA) extended this increased tax credit assistance and removal of the 400% federal poverty limit through 2025. There have also been legal challenges to certain aspects of the ACA. On June 17, 2021, the U.S. Supreme Court dismissed a judicial challenge to the ACA brought by several states without specifically ruling on the constitutionality of the ACA. It is unclear how the healthcare reform measures of the Biden administration or other efforts, if any, to challenge, repeal, amend or replace the ACA will impact the ACA and our business.
Also, there has been heightened governmental scrutiny over pharmaceutical pricing practices in light of the rising cost of prescription drugs and biologics. Such scrutiny has resulted in Congressional and federal agency inquiries regarding pricing and related practices, as well as proposed and enacted federal and state legislation and regulation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for products. For example, the American Rescue Plan Act of 2021 included among its provisions a sunset of the ACA’s cap on pharmaceutical manufacturers’ rebate liability under the Medicaid Drug Rebate Program. Previously, under the ACA, manufacturers’ rebate liability was capped at 100% of the average manufacturer price for a covered outpatient drug. Effective January 1, 2024, manufacturers’ Medicaid Drug Rebate Program rebate liability is no longer capped, potentially resulting in a manufacturer paying more in Medicaid Drug Rebate Program rebates than it receives on the sale of certain covered outpatient drugs.
The Biden Administration has continued to support legislative reforms to lower prescription drug prices in other ways. In August 2022, for instance, President Biden signed into law the IRA, which implements substantial changes to the Medicare program, including drug pricing reforms and changes to the Medicare Part D benefit design. Among other reforms, the IRA of 2022 imposes inflation rebates on drug manufacturers for products reimbursed under Medicare Parts B and D if the prices of those products increase faster than inflation; implements changes to the Medicare Part D benefit that, beginning in 2025, will cap patient annual out-of-pocket spending at $2,000, while imposing new discount obligations for pharmaceutical manufacturers and payors; and, beginning in 2026, establishes a “maximum fair price” for a fixed number of pharmaceutical and biological products covered under Medicare Parts B and D following a price negotiation process with the CMS. The IRA explicitly excludes from price negotiation orphan drugs designated for only one rare disease or condition and for which the only active approved indication is for such disease or condition. Those drugs with multiple orphan designations are not explicitly excluded from drug price negotiation.
Since the IRA was enacted, CMS has taken various steps to implement the drug pricing provisions of the law. This includes, without limitation, on June 30, 2023 issuing guidance detailing the requirements and parameters of the first round of price negotiations, to take place during 2023 and 2024, for products subject to the “maximum fair price” provision that would become effective in 2026, on August 29, 2023, releasing the initial list of 10 drugs subject to price negotiations; and on
December 14, 2023 releasing a list of 48 Medicare Part B products that had adjusted coinsurance rates based on the inflationary rebate provisions of the IRA for the time period of January 1, 2024 to March 31, 2024. While it remains to be seen how the drug pricing provisions imposed by the IRA will affect the broader pharmaceutical industry (including orphan drug development), several pharmaceutical manufacturers and other industry stakeholders have challenged the law, including through lawsuits brought against the HHS, the Secretary of HHS, CMS, and the CMS Administrator challenging the constitutionality and administrative implementation of the IRA’s drug price negotiation provisions. We cannot predict
the likelihood, nature or extent of other healthcare or drug pricing reforms that may arise from future legislation or other administration action nor can we predict how, or to what extent, the drug pricing policies or future healthcare reforms will affect our products. However, we expect these initiatives to increase pressure on drug pricing.
At the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. Any legislation at the state or federal level aimed at further healthcare reform may have the effect of limiting the amounts that government agencies will pay for healthcare products and services. For example, an emerging trend at the state level is the establishment of prescription drug affordability boards, some of which will prospectively permit certain states to establish upper payment limits for drugs that the state has determined to be “high-cost”. In addition, policy changes, including potential modification or repeal of all or parts of the ACA or the implementation of new health care legislation, could result in significant changes to the health care system, which may prevent us from being able to generate revenue, attain profitability or commercialize our drugs. We expect that additional state and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in reduced demand or lower pricing for our product candidates, or additional pricing pressures.
In the United States, the EU and other potentially significant markets for our product candidates, government authorities and third-party payors are increasingly attempting to limit or regulate the price of medical products and services, particularly for new and innovative products and therapies, which has resulted in lower average selling prices. The increased emphasis on managed healthcare in the United States and on country and regional pricing and reimbursement controls in the EU will put additional pressure on product pricing, reimbursement and usage, which may adversely affect our future product sales and results of operations. These pressures can arise from rules and practices of managed care groups, judicial decisions and governmental laws and regulations related to Medicare, Medicaid and healthcare reform, pharmaceutical reimbursement policies and pricing in general.
The proposed revision of the European legislation on pharmaceuticals could lead to uncertainties over the regulatory framework that will be applicable to medicinal products in the EU, including orphan medicinal products.
In April 2023, the European Commission published proposals to revise the existing European legislation on medicinal products (“EU Pharma Law Review”). The revision consists of two proposals, a new directive and a new regulation (“EU Pharma Law Proposal”), that would amend and/or repeal and replace the relevant legislation concerning medicinal products for human use, including legislation concerning orphan medicinal products and medicinal products for paediatric use. The EU Pharma Law Review could have a significant impact on the RDP available for to innovative medicinal products in the EU. If adopted in current form, the EU Pharma Law Proposal would reduce the current baseline for data exclusivity from eight to six years, extensible under certain conditions. Such RDP reduction could lead to a faster access to the EU market for generics and biosimilars. The EU Pharma Law Review could have a significant impact on the designation of and incentives offered to orphan medicinal products in the EU. If adopted in current form, the EU Pharma Law Proposal would introduce the possibility for the Commission, by way of delegated acts, to derogate from the current prevalence criterion, and introduce specific criteria for certain conditions, due to the characteristics of such conditions or other scientific reasons. The EU Pharma Law Proposal also proposes changes to the current orphan market exclusivity (“OME”) approach. If adopted in the current form, the EU Pharma Law Proposal would in most cases reduce the duration of the OME, and replace the current system with separate OME periods for each new indication, to a system with a single OME period for each active substance. The EU Pharma Law Review could have a significant impact on paediatric medicinal products. If adopted in the current form, the EU Pharma Law Proposal would introduce a new evolutionary PIPs system for certain types of developments, allowing the submission of an initial high level PIP, to be subsequently completed at precise stages in the development. The greater flexibility granted by this step-wise approach could be offset by the more stringent scrutiny over PIPs by the EMAw. In addition, the EU Pharma Law Proposal could introduce the obligation to place medicinal products authorized for a paediatric indication on the market of all Member States where the medicinal product is already placed on the market.
We may be subject, directly or indirectly, to foreign, federal and state healthcare fraud and abuse laws, false claims laws, and health information privacy and security laws. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties, sanctions or other liability.
Our operations may be subject to various foreign, federal and state fraud and abuse laws, including, without limitation, the federal Anti-Kickback Statute, the federal False Claims Act, physician sunshine laws, the EU GDPR and/or the UK GDPR
and other regulations. These laws may impact, among other things, our research, sales, marketing, education and patient assistance programs. In addition, we may be subject to patient privacy regulation by foreign, federal, and state governments in which we conduct our business. The laws that may affect our ability to operate include:
•the federal Anti-Kickback Statute, which prohibits, among other things, persons from knowingly and willfully soliciting, receiving, offering or paying anything of value as remuneration, directly or indirectly, to induce, or in return for, the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs;
•federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third-party payors that are false or fraudulent;
•HIPAA, which created new federal criminal statutes that prohibit executing a scheme to defraud any healthcare benefit program and making false statements relating to healthcare matters;
•HIPAA and its implementing regulations impose certain requirements on certain covered entity healthcare providers, health plans, and healthcare clearinghouse and their business associates that perform certain services involving the use or disclosure of individually identifiable health information as well as their covered subcontractors, relating to the privacy, security, and transmission of individually identifiable health information;
•The Physician Payments Sunshine Act, which requires, among others, manufacturers of certain drugs, devices, biologics, and medical supplies for which payment is available under Medicare, Medicaid, or the Children’s Health Insurance Program to report annually to CMS information related to certain payments and other transfers of value to U.S.-licensed physicians (defined to include doctors, dentists, optometrists, podiatrists, and licensed chiropractors), teaching hospitals, physician assistants, nurse practitioners, clinical nurse specialists, anesthesiologist assistants, certified registered nurse anesthetists, and certified nurse midwives, as well as tracking and reporting of ownership and investment interests held by U.S.-licensed physicians and their immediate family members and applicable group purchasing organizations;
•the Federal Food, Drug, and Cosmetic Act (FDCA), which, among other things, prohibits the adulteration and misbranding of drugs and biological products;
•state law equivalents of each of the above federal laws, such as state anti-kickback and false claims laws that may apply to items or services reimbursed by any third-party payors, including commercial insurers, state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, or otherwise restrict payments that may be made to healthcare providers and other potential referral sources; state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; state and local laws requiring the registration of pharmaceutical sales representatives; state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts; and federal and state laws requiring the disclosure of certain drug pricing information; and
•the EU GDPR and/or the UK GDPR (together, GDPR) and other EU Member State or English data protection legislation, which require data controllers and processors to (i) implement administrative, physical, technical, and organizational safeguards to protect personal information; (ii) establish an appropriate and valid legal basis for processing personal information (iii) comply with accountability transparency requirements regarding the processing of personal information, which require controllers to demonstrate and record compliance with the GDPR and to provide more detailed information to data subjects regarding processing; (iv) comply with data protection rights of data subjects including a right of access to and rectification of personal information, a right to obtain restriction of processing or to object to processing of personal information, a right to ask for a copy of personal information to be provided to a third party in a useable format and erasing personal information in certain circumstances; (v) report certain personal data breaches to the relevant supervisory authority without undue delay (and no later than 72 hours, where feasible); (vi) obtain explicit consent for collection of sensitive personal information such as health data; and (vii) consider data protection as any new products or services are developed and to limit the amount of personal information processed. In addition, the GDPR prohibits the international transfer of personal information outside of the EU and/or the UK including to the U.S., unless made to a country deemed to have adequate data privacy laws by the European Commission and/or the UK or a data transfer mechanism in accordance with the GDPR (as applicable) has been put in place.
Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws. In addition, recent health care reform legislation has strengthened these laws.
If our operations are found to be in violation of any of the laws described above or any other governmental regulations that apply, we may be subject to penalties, including significant civil, criminal and administrative penalties, damages, disgorgement, fines, sanctions, exclusion from participation in government health care programs, such as Medicare and Medicaid, imprisonment, corporate integrity oversight and reporting obligations, contractual damages, reputational harm, diminished profits and future earnings, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
If we fail to comply with our reporting and payment obligations under the Medicaid Drug Rebate Program or other governmental pricing programs in the US, we could be subject to additional rebate or discount requirements, fines, sanctions and exposure under other laws which could have an adverse effect on our business, results of operations and financial condition.
The Company’s product is subject to government mandated rebates under the Medicaid Drug Rebate Program and other government health care programs in the United States. These programs generally require us to pay rebates or otherwise provide discounts to government payors in connection with drugs that are dispensed to beneficiaries of these programs. In some cases, such as with the Medicaid Drug Rebate Program, rebates are calculated based on pricing metrics we are required to report on a monthly and quarterly basis to the government agencies that administer the programs. These pricing metrics are complex and may vary across products and programs. Additionally, the requirements of government health care programs change frequently and are often subject to interpretation by regulatory agencies and the courts. Tracking these updates and maintaining compliance will be costly and time consuming. Further, errors in the submission of our pricing data, failure to make necessary pricing disclosures, and/or overcharging government payors (or failing to identify overpayments) could result in allegations against us under the federal False Claims Act and other laws and regulations, as well as subject us to potential termination of our rebate agreement with CMS. Responding to government investigations or enforcement actions and/or making necessary refunds to government payors would be expensive and time consuming and could have an adverse effect on our business, results of operations and financial condition.
Additionally, we may be affected by developments relating to the 340B Drug Pricing Program. Recently, multiple manufacturers have implemented policies to reduce diversion and inappropriate claims for discounts and rebates by contract pharmacies affiliated with 340B-eligible entities. The Department of Health and Human Services has sent several of these manufacturers letters claiming that the policies violate the 340B statue and referring the manufacturers for potential enforcement action. Manufacturers have challenged these letters in federal court, and the U.S. Court of Appeals for the Third Circuit has ruled in favor of several manufacturers; other challenges are still pending. Further, Arkansas and Louisiana recently enacted laws requiring manufacturers to ship 340B drugs to certain contract pharmacies and imposing penalties on manufacturers that do not comply. Both laws have been challenged in federal court. Other states are considering similar laws, and federal legislation has been proposed as well. It is unclear how this pending litigation, recent and proposed legislation, or future administrative action relating to the 340B program will impact our business.
Following Brexit, legal, political and economic uncertainty surrounding the exit of the U.K. from the EU may be a source of instability in international markets, create significant currency fluctuations, adversely affect our operations in the U.K. and pose additional risks to our business, revenue, financial condition and results of operations.
In June 2016, the electorate in the UK voted in favor of leaving the EU (commonly referred to as “Brexit”). Thereafter, in March 2017, the country formally notified the EU of its intention to withdraw pursuant to Article 50 of the Lisbon Treaty and the UK formally left the EU on January 31, 2020. A transition period began on February 1, 2020, during which EU pharmaceutical law remained applicable to the UK. This transition period ended on December 31, 2020. The EU-UK Trade and Cooperation Agreement (TCA) was agreed on December 24, 2020. The UK and EU agreed that the TCA would apply provisionally from January 1, 2021, the TCA was ratified on April 30, 2021 and came into force on May 1, 2021.
Since the regulatory framework in the UK covering the quality, safety and efficacy of pharmaceutical products, clinical trials, marketing authorization, commercial sales and distribution of pharmaceutical products is derived from EU Directives and Regulations, Brexit could materially impact the future regulatory regime which applies to products and the approval of product candidates in the UK, as UK legislation now has the potential to diverge from EU legislation. It remains to be seen how Brexit will fully impact regulatory requirements for product candidates and products in the UK in the long-term.
At present, the regulatory framework for medicines that existed before the end of the transition period has effectively been preserved in UK domestic legislation as ‘retained EU law’ which has prevented substantial divergence to the regulation of medicines so far. However, the Retained EU Law (Revocation and Reform) Bill 2022 which intends to end the “special status” of retained EU law under UK law was enacted into law on June 29, 2023. It facilitates the departure from retained EU law, and may lead to greater regulatory divergence between the EU and UK in the future. In comparison, as matters currently stand, under the terms of the Northern Ireland Protocol (the NIP), implemented by the EU (Withdrawal Agreement) Act 2020, Northern Ireland is treated for the same purposes as if it were still an EU Member State, and must remain aligned to the EU single market and customs rules. The UK government has also introduced a bill ‘Northern Ireland Protocol Bill’ which if enacted into law would enable the government to unilaterally disapply parts of the NIP which may lead to changes to the regulatory environment in Northern Ireland, and may trigger retaliatory measures against the UK by the EU. However, on February 27, 2023, the UK government and the EU Commission reached political agreement on a package of proposed amendments to the NIP (Windsor Framework). These proposals were adopted on March 24, 2023 and include, amongst other measures a “carve-out from EU rules”. Indeed, the UK medicines regulator (MHRA) will be responsible for approving medicines for the whole UK market, meaning that medicinal products available in Northern Ireland will not have to comply with EU law but rather with UK law, so medicinal products would be able to freely circulate between Northern Ireland and Great Britain. It is expected that these new measures put in place by the Windsor Framework will take effect from January 1, 2025. The EU-UK Trade and Cooperation Agreement allows for future deviation from the current regulatory framework and it is not known if and/or when any deviations may occur, which may have an impact on Eiger’s business.
Companies now need to comply with a separate UK regulatory legal framework in order to commercialize medicinal products in Great Britain (namely, England, Wales and Scotland, as EU law currently applies in Northern Ireland). For instance, Great Britain will no longer be covered by centralized marketing authorizations (as matters stand, under the Northern Irish Protocol, centralized marketing authorizations will continue to be recognized in Northern Ireland). All medicinal products with a current centralized marketing authorization were automatically converted to Great Britain marketing authorizations on January 1, 2021. For a period of three years from January 1, 2021, the MHRA may rely on a decision taken by the European Commission on the approval of a new marketing authorization in the centralized procedure, in order to more quickly grant a new Great Britain marketing authorization. A separate application will, however, still be required. The MHRA has also introduced new procedures aimed at accelerating regulatory approvals and time to market, including ‘rolling reviews’ and the Innovative Licensing and Access Pathway (ILAP) although eligibility requirements apply.
The TCA and the proposed Bills allow for future deviation from the current regulatory framework and it is not known if and/or when any deviations may occur, which may have an impact on development, manufacture, marketing authorization, commercial sales and distribution of pharmaceutical products.
Our employees, principal investigators, consultants and commercial partners may engage in misconduct or other improper activities, including non-compliance with regulatory standards and requirements and insider trading.
We are exposed to the risk of fraudulent conduct or other illegal activity by our employees, independent contractors, principal investigators, consultants, commercial partners and vendors. Misconduct by these parties could include intentional, reckless and/or negligent conduct that fails to: comply with the regulations of the FDA and non-U.S. regulators, provide accurate information to the FDA and non-U.S. regulators, comply with healthcare fraud and abuse laws in the United States and abroad, report financial information or data accurately or disclose unauthorized activities to us. In particular, promotion, sales, marketing and certain business arrangements in the healthcare industry are subject to extensive laws intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Such misconduct could also involve the improper use of information obtained in the course of patient recruitment or clinical trials, which could result in regulatory sanctions and cause serious harm to our reputation. We have adopted a code of business conduct and ethics applicable to all of our employees, but it is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant fines or other sanctions.
We face potential product liability, and, if successful claims are brought against us, we may incur substantial liability and costs. If the use or misuse of our product candidates harm patients or is perceived to harm patients even when such harm is unrelated to our product candidates, our regulatory approvals could be revoked or otherwise negatively impacted, and we could be subject to costly and damaging product liability claims.
The use or misuse of our product candidates in clinical trials and the sale of any products for which we obtain marketing approval exposes us to the risk of product liability claims. Product liability claims might be brought against us by consumers, healthcare providers, pharmaceutical companies or others selling or otherwise coming into contact with our products. There is a risk that our product candidates may induce adverse events. If we cannot successfully defend against product liability claims, we could incur substantial liability and costs. In addition, regardless of merit or eventual outcome, product liability claims may result in:
•impairment of our business reputation;
•initiation of investigations by regulators;
•withdrawal of clinical trial participants;
•costs due to related litigation;
•distraction of management’s attention from our primary business;
•substantial monetary awards to patients or other claimants;
•the inability to commercialize our product candidates;
•product recalls, withdrawals or labeling, marketing or promotional restrictions; and
•decreased demand for our product candidates, if approved for commercial sale.
We believe our current product liability insurance coverage is appropriate in light of our clinical programs; however, we may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability. If and when we obtain marketing approval for product candidates, we intend to increase our insurance coverage to include the sale of commercial products; however, we may be unable to obtain product liability insurance on commercially reasonable terms or in adequate amounts. On occasion, large judgments have been awarded in class action lawsuits based on drugs or medical treatments that had unanticipated adverse effects. A successful product liability claims or series of claims brought against us could cause our stock price to decline and, if judgments exceed our insurance coverage, could adversely affect our results of operations and business.
Patients with the diseases targeted by our product candidates are often already in severe and advanced stages of disease and have both known and unknown significant pre-existing and potentially life-threatening health risks. During the course of treatment, patients may suffer adverse events, including death, for reasons that may or may not be related to our product candidates. Such events could subject us to costly litigation, require us to pay substantial amounts of money to injured patients, delay, negatively impact or end our opportunity to receive or maintain regulatory approval to market our products, or require us to suspend or abandon our commercialization efforts. Even in a circumstance in which we do not believe that an adverse event is related to our products, the investigation into the circumstance may be time-consuming or inconclusive. These investigations may interrupt our sales efforts, delay our regulatory approval process in other countries, or impact and limit the type of regulatory approvals our product candidates receive or maintain. As a result of these factors, a product liability claim, even if successfully defended, could have a material adverse effect on our business, financial condition or results of operations.
If we fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have a material adverse effect on the success of our business.
Our research and development activities and our third-party manufacturers’ and suppliers’ activities involve the controlled storage, use, and disposal of hazardous materials, including the components of our product candidates and other hazardous compounds. We and our manufacturers and suppliers are subject to laws and regulations governing the use, manufacture, storage, handling, and disposal of these hazardous materials. In some cases, these hazardous materials and various wastes resulting from their use are stored at our and our manufacturers’ facilities pending their use and disposal. We cannot eliminate the risk of contamination, which could cause an interruption of our commercialization efforts, research and development efforts and business operations, environmental damage resulting in costly clean-up and liabilities under applicable laws and regulations governing the use, storage, handling, and disposal of these materials and specified waste products. Although we believe that the safety procedures utilized by our licensors and our third-party manufacturers for
handling and disposing of these materials generally comply with the standards prescribed by these laws and regulations, we cannot guarantee that this is the case or eliminate the risk of accidental contamination or injury from these materials. In such an event, we may be held liable for any resulting damages and such liability could exceed our resources and state or federal or other applicable authorities may curtail our use of certain materials and/or interrupt our business operations. Furthermore, environmental laws and regulations are complex, change frequently, and have tended to become more stringent. We cannot predict the impact of such changes and cannot be certain of our future compliance. We do not currently carry biological or hazardous waste insurance coverage.
We are currently conducting and will continue to conduct clinical trials in foreign countries, which could expose us to risks that could have a material adverse effect on the success of our business and the delivery of clinical trial data.
We have conducted in the past clinical trials in the United States, Canada, Australia, Turkey, Germany, Pakistan, New Zealand, Mongolia, Spain, France, Bulgaria, Romania, Taiwan, Sweden, Italy, Belgium, Switzerland, United Kingdom, Greece, Moldova, Ukraine, Russia, and Israel, and accordingly, we are subject to risks associated with doing business globally, including commercial, political, and financial risks. Emerging regions, such as Eastern Europe, Latin America, Asia, and Africa, as well as more developed markets, such as the United Kingdom, France, Germany, and Australia, provide clinical study opportunities for us. In addition, we are subject to potential disruption caused by military conflicts; potentially unstable governments or legal systems; civil or political upheaval or unrest; local labor policies and conditions; possible expropriation, nationalization, or confiscation of assets; problems with repatriation of foreign earnings; economic or trade sanctions; closure of markets to imports; anti-American sentiment; terrorism or other types of violence in or outside the United States; health pandemics; and a significant reduction in global travel. Certain countries previously closed their borders due to COVID-19 preventing activation of clinical sites. Actions taken by the Russian Federation in Ukraine and surrounding areas have destabilized the region and caused the adoption of comprehensive sanctions by, among others, the EU, the United States and the UK, which restrict a wide range of trade and financial dealings with Russia and Russian persons, as well as certain regions in Ukraine, including by imposing stricter export controls, prohibiting dealings with major Russian banks and credit institutions, and prohibiting trade with the Donetsk, Luhansk, Kherson and Zaporizhzhia regions of Ukraine. In addition, clinical site initiation and patient enrollment may be delayed, and we may not be able to access sites for initiation, monitoring and data collection in regions affected by conflict in the Middle East and the Russian invasion of Ukraine, including due to the prioritization of hospital resources away from clinical trials or as a result of government-imposed curfews, warfare, violence or other governmental action or events that restrict movement. Some patients may not be able to comply with clinical trial protocols if the conflict impedes patient movement or interrupts healthcare services. We could also experience disruptions in our supply chain or limits to our ability to obtain sufficient materials for our products in certain regions. Our success will depend, in part, on our ability to overcome the challenges we encounter with respect to these risks and other factors affecting U.S. companies with global operations. If our global clinical trials were to experience significant disruption due to these risks or for other reasons, it could have a material adverse effect on our operations and prospects.
We or the third parties upon whom we depend may be adversely affected by earthquakes, natural epidemics or other natural disasters and our business continuity and disaster recovery plans may not adequately protect us from a serious disaster.
Earthquakes, health epidemics or other natural disasters could severely disrupt our operations and have a material adverse effect on our business. If a natural disaster, power outage or other event occurred that prevented us from using all or a significant portion of our headquarters, damaged critical infrastructure, such as the manufacturing facilities of our third-party CMOs, or otherwise disrupted operations, it may be difficult or, in certain cases, impossible for us to continue our business for a substantial period of time. For example, in December 2019, an outbreak of a novel strain of coronavirus (COVID-19) originated in Wuhan, China. Since certain starting materials of certain of our products obtained from third-party chemical suppliers are manufactured in China and Japan, an outbreak of communicable diseases in the region, or the perception that such an outbreak could occur, and the measures taken by the governments of countries affected, could adversely affect our business, financial condition or results of operations by limiting our ability to manufacture product within or outside certain countries, forcing temporary closure of facilities that we rely upon or increasing the costs associated with obtaining starting materials and then clinical supplies of our product candidates. The extent to which the coronavirus or another PHE impacts our results will depend on future developments, which are highly uncertain and cannot be accurately predicted, including new information which may emerge concerning the severity of the coronavirus or other PHEs and the actions to contain the coronavirus or treat its impact, among others. In addition, our corporate headquarters is located in the San Francisco Bay Area, which has in the past experienced severe earthquakes, other natural disasters, and an outbreak of COVID-19. We do not carry earthquake insurance. We have limited disaster recovery and business continuity plans in place currently and our business would be impaired in the event of a serious disaster or similar event. We may
incur substantial expenses to develop and implement any disaster recovery and business continuity plans, which could have a material adverse effect on our business.
Our business has been adversely affected by and could be materially adversely affected in the future by the effects of disease outbreaks, epidemics and pandemics including the continuing effects of the COVID-19 outbreak. We had a significant number of clinical trial sites, and continue to depend on manufacturing operations for various stages of our supply chain, in countries that were directly affected by COVID-19.
Our business has been adversely affected by COVID-19 and could be materially and adversely impacted by COVID-19 or other health epidemics in regions where we have significant manufacturing facilities, concentrations of clinical trial sites or other business operations. The effects of COVID-19 or other epidemics and government and other responses thereto, including any resumption of shelter-in-place orders or work-from-home policies, may continue to negatively impact productivity, disrupt our business and delay our clinical programs and timelines, the magnitude of which would depend, in part, on the length and severity of any restrictions and other limitations on our ability to conduct our business in the ordinary course. These and similar, and perhaps more severe, disruptions in our operations could negatively impact our business, operating results and financial condition.
Further, as a result of other PHEs, including any resurgence of the COVID-19 pandemic, we may be required to develop and implement additional clinical study policies and procedures designed to help protect study participants from illness, which may include using telemedicine visits, remote monitoring of patients and clinical sites, and measures to ensure that data from clinical studies that may be disrupted as result of the pandemic are collected pursuant to the study protocol and consistent with GCPs, with any material protocol deviation reviewed and approved by the site IRB.
The FDA and EMA have in the past suspended, may continue to suspend, prioritize or delay certain foreign inspections, and if suspensions or delays in inspections occur, our product application reviews and potential approvals could be impacted or delayed. Further, prior surges in COVID-19 case numbers have contributed to interruptions in FDA’s surveillance capabilities, and such disruptions may persist in the future as a result of future surges or other PHEs. We cannot predict whether, and when, FDA will decide to pause or resume inspections due to a pandemic.
Regulatory authorities outside the United States may adopt additional restrictions or other policy measures in response to PHEs, including providing guidance regarding the conduct of clinical trials. If global health concerns prevent the FDA or other regulatory authorities from conducting their regular inspections, or impact reviews or other regulatory activities, it could significantly impact the ability of the FDA or other foreign regulatory authorities to timely review and process our regulatory submissions, which could have a material adverse effect on our business.
COVID-19 may continue to adversely affect our business operations, and the extent of the impact on our clinical development and regulatory efforts and the value of and market for our common stock will depend on future developments that are highly uncertain and cannot be predicted with confidence at this time. In addition, to the extent COVID-19 or another health epidemic adversely affects our business and results of operations, it may also have the effect of heightening many of the other risks and uncertainties described elsewhere in this ‘‘Risk Factors’’ section.
Risks Related to Ownership of our Common Stock
The market price of our common stock has been and may continue to be highly volatile, and you may not be able to resell some or all of your shares at a desired market price.
The market price of our common stock has been and is likely to continue to be volatile. Our stock price could be subject to wide fluctuations in response to a variety of factors, including the following:
•results or delays in preclinical studies or clinical trials;
•our decision to initiate a clinical trial, not to initiate a clinical trial or to terminate an existing clinical trial;
•unanticipated serious safety concerns related to the use of any of our product candidates;
•reports of adverse events in other gene therapy products or clinical trials of such products;
•inability to obtain additional funding;
•any delay in filing an IND, NDA, BLA, or MAA for any of our product candidates and any adverse development or perceived adverse development with respect to the FDA’s review of that IND, NDA, or BLA (or the EMA’s review of that MAA);
•our ability to obtain regulatory approvals for our product candidates, and delays or failures to obtain such approvals;
•failure of any of our product candidates, if approved, to achieve commercial success;
•failure to obtain or maintain Orphan Drug designation;
•failure to maintain our existing third-party license and supply agreements;
•failure by our licensors to prosecute, maintain, or enforce our intellectual property rights;
•changes in laws or regulations applicable to our product candidates;
•any inability to obtain adequate supply of our product candidates or the inability to do so at acceptable prices;
•adverse regulatory authority decisions;
•introduction of new products, services, or technologies by our competitors;
•failure to meet or exceed financial and development projections we may provide to the public;
•failure to meet or exceed the financial and development projections of the investment community;
•the perception of the pharmaceutical industry by the public, legislatures, regulators, and the investment community;
•announcements of significant acquisitions, strategic partnerships, joint ventures, or capital commitments by us or our competitors;
•disputes or other developments relating to proprietary rights, including patents, litigation matters, and our ability to obtain patent protection for our technologies;
•additions or departures of key personnel;
•significant lawsuits, including patent or stockholder litigation;
•if securities or industry analysts do not publish research or reports about our business, or if they issue adverse or misleading opinions regarding our business and stock;
•changes in the market valuations of similar companies;
•general market or macroeconomic conditions;
•sales of our common stock by us or our stockholders in the future;
•trading volume of our common stock;
•announcements by commercial partners or competitors of new commercial products, clinical progress or the lack thereof, significant contracts, commercial relationships or capital commitments;
•adverse publicity relating to the hepatitis market generally, including with respect to other products and potential products in such markets;
•the introduction of technological innovations or new therapies that compete with potential products of ours;
•changes in the structure of health care payment systems; and
•period-to-period fluctuations in our financial results.
Moreover, the stock markets in general have experienced substantial volatility that has often been unrelated to the operating performance of individual companies, including very recently in connection with global macroeconomic and geopolitical conditions, which volatility has resulted in decreased stock prices for many companies notwithstanding the lack of a fundamental change in their underlying business models or prospects. These broad market and industry factors, including potentially worsening economic conditions and other adverse effects or developments, may also adversely affect the trading price of our common stock.
In the past, following periods of volatility in the market price of a company’s securities, stockholders have often instituted class action securities litigation against those companies. Such litigation, if instituted, could result in substantial costs and diversion of management attention and resources, which could significantly harm our profitability and reputation.
We will incur costs and demands upon management as a result of complying with the laws and regulations affecting public companies.
We have incurred and will continue to incur significant legal, accounting and other expenses associated with public company reporting requirements. We also incur costs associated with corporate governance requirements, including requirements under the Sarbanes-Oxley Act, as well as rules implemented by the SEC. These rules and regulations impose significant legal and financial compliance costs and make some activities more time-consuming and costly. For example, our management team consists of certain executive officers who have not previously managed and operated a public company. These executive officers and other personnel will need to devote substantial time to gaining expertise regarding operations as a public company and compliance with applicable laws and regulations. In addition, it may be more difficult for us to attract and retain qualified individuals to serve on our board of directors or as executive officers, which may adversely affect investor confidence and could cause our business or stock price to suffer.
As a result of the Chapter 11 Cases, our common stock will be delisted from Nasdaq, which could have a material adverse effect on the liquidity of our common stock and our stock price.
Following our filing of the Chapter 11 Cases, we received written notice from the Listing Qualifications Department of the Nasdaq Stock Market LLC notifying us that, as a result of the Chapter 11 Cases and in accordance with Nasdaq Listing Rules 5101, 5110(b) and IM-5101-1, Nasdaq had determined that the Company’s common stock will be delisted from Nasdaq. Trading of our common stock will be suspended at the opening of business on April 11, 2024. Our common stock is expected to start being quoted on the over-the-counter ("OTC") market under the symbol EIGRQ.
The lack of an active market may impair the ability of holders of our common stock to sell their shares at the time they wish to sell them or at a price that they consider reasonable. The lack of an active market may also reduce the fair market value of the shares of our common stock. Furthermore, because of the limited market and generally low volume of trading in our common stock, the price of our common stock could be more likely to be affected by broad market fluctuations, general market conditions, fluctuations in our operating results, changes in the markets’ perception of our business, and announcements made by us, parties with whom we have business relationships or third parties with interests in the Chapter 11 Cases. Additionally, the delisting may adversely impact the perception of the Company’s financial condition and cause reputational harm with investors and parties conducting business with the Company. The perceived decreased value of employee equity incentive awards may reduce their effectiveness in encouraging performance and retention. Each of these occurrences could have a material adverse effect on our business.
Anti-takeover provisions in our charter documents and under Delaware law could make an acquisition of us more difficult and may prevent attempts by our stockholders to replace or remove our management.
Provisions in our certificate of incorporation and bylaws may delay or prevent an acquisition or a change in management. These provisions include a classified board of directors, a prohibition on actions by written consent of our stockholders and the ability of the board of directors to issue preferred stock without stockholder approval. In addition, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which prohibits stockholders owning in excess of 15% of our voting stock from merging or combining with us. Although we believe these provisions collectively will provide for an opportunity to receive higher bids by requiring potential acquirers to negotiate with our board of directors, they would apply even if the offer may be considered beneficial by some stockholders. In addition, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove then current management by making it more difficult for stockholders to replace members of the board of directors, which is responsible for appointing the members of management.
We do not anticipate that we will pay any cash dividends in the foreseeable future.
We expect to retain our future earnings to fund the development and growth of our business. As a result, capital appreciation, if any, of our common stock will be the sole source of gain for our stockholders, if any, for the foreseeable future.
Future sales of shares by existing stockholders could cause our stock price to decline.
If existing stockholders sell, or indicate an intention to sell, substantial amounts of our common stock in the public market after legal restrictions on resale lapse, the trading price of our common stock could decline. Certain of our existing stockholders, including Columbia Threadneedle Investments, Arkin, 683 Capital Management, The Vanguard Group,
Telemetry Investments, and their respective affiliated entities, own substantial ownership interest in our common stock and any decision to sell a significant number of shares may negatively impact the price of our common stock.
The ownership of our common stock is highly concentrated, and it may prevent stockholders from influencing significant corporate decisions and may result in conflicts of interest that could cause our stock price to decline.
Our executive officers, directors and 5% stockholders and their affiliates beneficially own or control a significant portion of the outstanding shares of our common stock. Accordingly, these executive officers, directors, 5% stockholders and their affiliates, acting as a group, have substantial influence over the outcome of corporate actions requiring stockholder approval, including the election of directors, any merger, consolidation or sale of all or substantially all of our assets or any other significant corporate transactions. These stockholders may also delay or prevent a change of control of us, even if such a change of control would benefit our other stockholders. The significant concentration of stock ownership may adversely affect the trading price of our common stock due to investors’ perception that conflicts of interest may exist or arise.
Our net operating loss carryforwards and certain other tax attributes are now subject to limitations.
Our federal and state net operating loss (NOL) carry-forwards will begin to expire, if not utilized, beginning in 2030 for federal income tax purposes and 2028 for California state income tax purposes. These NOL carry-forwards could expire unused and be unavailable to offset future income tax liabilities. Under legislation enacted in 2017, informally titled the Tax Cuts and Jobs Act, as modified by the Coronavirus Aid, Relief, and Economic Security Act (CARES Act), U.S. federal net operating loss carryforwards generated in tax years beginning after December 31, 2017 may be carried forward indefinitely but, in the case of tax years beginning after December 31, 2020, may only be used to offset 80% of taxable income annually. In addition, California has enacted A.B. 85 which imposed limits on the usability of California state net operating losses and certain tax credits in tax years beginning after 2019 and before 2023. Such limitations could result in the expiration of portions of our net operating loss and tax credit carryforwards before utilization. On February 9, 2022, Senate Bill No. 113 was signed into California law and reinstates the net operating loss deduction, and removes the above-described temporary limitation on allowable credits, for taxable years beginning on or after January 1, 2022. Moreover, if a corporation undergoes an ownership change within the meaning of Section 382 of the Code (Section 382) the corporation’s NOL carryforwards and certain other tax attributes arising from before the ownership change are subject to limitations on use after the “ownership change.” In general, an ownership change occurs if there is a cumulative change in the corporation’s equity ownership by certain stockholders that exceeds fifty percentage points over a rolling three-year period. Similar rules may apply under state tax laws. Our merger with Celladon resulted in such an ownership change and, accordingly, Celladon’s NOL carryforwards and certain other tax attributes will be subject to further limitations on their use. In addition, we assessed whether Eiger had an ownership change, as defined by Section 382 of the Code, as a result of the Merger and other stock issuances that occurred from our formation through December 31, 2020. Based upon this assessment, we have experienced ownership changes on April 20, 2016, October 18, 2018 and December 31, 2020. Due to these ownership changes, reductions were made to our NOL and tax credit carryforwards under these rules. Additional ownership changes in the future could result in additional limitations on our net operating loss and tax credit carryforwards. Consequently, even if we achieve profitability, we may not be able to utilize a material portion of our NOL carryforwards and other tax attributes, which could have a material adverse effect on cash flow and results of operations. A full valuation allowance has been provided for the entire amount of our remaining net operating loss.
ITEM 1B. Unresolved Staff Comments
Not applicable.
ITEM 1C. Cybersecurity
As cyber-attacks become more prevalent, we’ve taken action to mitigate the threat to our business. As described in more detail below and as part of our risk management program, we have established policies and processes for assessing, identifying, and managing material risks from cybersecurity threats. Cybersecurity is a critical element of this program.
Risk Management and Strategy
Management along with the support of a third-party IT firm are responsible for the day-to-day administration of our risk management program and our cybersecurity policies, processes, and practices.
Identification and Reporting
We implemented a cross-functional approach to assessing, identifying, and managing material cybersecurity threats and incidents. We have put in place controls and procedures to identify, classify, and escalate certain cybersecurity incidents to provide management visibility and obtain direction from management as to the public disclosure and reporting of material incidents in a timely manner. Incidents are reported and tracked through our third-party IT firm's online support system.
Technical Safeguards
We implemented technical safeguards that are designed to protect our information systems from cybersecurity threats, including firewalls, intrusion prevention and detection systems, anti-malware functionality, and access controls, which are evaluated and improved through quarterly vulnerability assessments and cybersecurity threat intelligence, as well as outside periodic audits and certifications.
Incident Response and Recovery Planning
We have established and maintain comprehensive incident response, business continuity, and disaster recovery plans designed to address our response to a cybersecurity incident.
Third-Party Risk Management
We maintain a risk-based approach to identifying and overseeing material cybersecurity threats presented by third parties, including vendors, service providers, and other external users of our systems, as well as the systems of third parties that could adversely impact our business in the event of a material cybersecurity incident affecting those third-party systems, including any outside auditors or consultants who advise on our cybersecurity systems.
Education and Awareness
We provide mandatory training for all employees and consultants regarding cybersecurity threats. The goal of the training is to equip our employees with tools and to raise their awareness of cybersecurity risks the Company faces. We conduct random training campaigns via email to test their knowledge and responses. We regularly communicate tips and current events to keep cybersecurity top of mind.
Governance
Board Oversight
Our Board of Directors, in coordination with our Audit Committee, oversees our risk management program, including the management of cybersecurity threats. Our Audit Committee receive quarterly updates on developments in the cybersecurity space, including risk management practices, recent developments, evolving standards, vulnerability assessments, third-party and independent reviews, the threat environment, technological trends, and information security issues encountered by our peers and third parties.
In the event of a material incident, our Board of Directors and our Audit Committee would receive from management prompt and timely information regarding any cybersecurity risk that meet reporting thresholds, as well as ongoing updates regarding any such risk.
Management’s Role
Management along with the support of a third-party IT firm are responsible for the day-to-day administration of our risk management program and our cybersecurity policies, processes, and practices.
On a monthly basis, the cross functional team meets to discuss current trends in cybersecurity threats and the response required to eliminate the threats.
As of the date of this report, we have not identified any risks from cybersecurity threats, including as a result of any previous cybersecurity incidents, that have materially affected us, our business strategy, results of operation or financial condition. For more information about the risks we face from cybersecurity incidents, please refer to Part I, Item 1A. “Risk Factors” of this report, including under the caption “Failure in our information technology and storage systems or our security measures, including without limitation, data breaches, or inadequacy of our business continuity and disaster recovery plans and procedures, could significantly disrupt the operation of our business.”
ITEM 2. Properties
Our corporate headquarters are located at 2155 Park Blvd in Palo Alto, California 94306 in a facility we lease encompassing 8,029 square feet of office space. The lease commenced on March 1, 2018 and was to expire five years after the commencement date. The lease had one three-year renewal option prior to expiration and included rent escalation clauses through the lease term. In February 2023, we amended the lease to extend the lease term for an additional year from March 1, 2023 to February 28, 2024. In February 2024, we amended the lease to reduce the space to 1,881 square feet of office space and extend the lease term for an additional month from March 1, 2024 to April 1, 2024 with a month to month option to renew.
ITEM 3. Legal Proceedings
We do not list all routine litigation matters with which we are a party. We discuss below certain pending matters. In determining whether to discuss a pending matter, we consider both quantitative and qualitative factors to assess materiality, such as, among others, the amount of damages alleged and the nature of other relief sought, if specified; our view of the merits of the claims and of the strength of our defenses; and whether the action purports to be, or is, a class action the jurisdiction in which the proceeding is pending.
Schoen v. Eiger BioPharmaceuticals, Inc., et al., Case No. 22-cv-06985
On November 8, 2022 a putative securities class action complaint was filed in the United States District Court for the Northern District of California alleging that the company and two former executives violated Sections 10(b) and 20(a) of the Securities Exchange Act and SEC Rule 10b-5. The complaint alleged generally that between March 2021 and October 2022, material misstatements and omissions were made to shareholders regarding the TOGETHER study of peginterferon lambda for the treatment of COVID-19 as well as the likelihood of FDA approval of an Emergency Use Authorization for peginterferon lambda. The Court appointed a lead plaintiff on March 2, 2023. On April 10, 2023, the lead plaintiff filed a notice of voluntary dismissal without prejudice.
The Progeria Research Foundation, Inc. v. Eiger BioPharmaceuticals, Inc. Arbitration
On November 15, 2022, the Company received a demand for arbitration (Demand) from claimant, The Progeria Research Foundation, Inc. (PRF), asserting two claims under a May 15, 2018 Collaboration and Supply Agreement (the PRF Collaboration Agreement) between the parties. PRF has alleged that the Company breached an obligation to supply quantities of a drug as requested by PRF. PRF also has a claim for declaratory relief regarding the grant of licenses under the PRF Collaboration Agreement. On January 18, 2023, the Company filed a response to the Demand denying PRF’s claims, contesting the arbitrability of PRF’s claim for declaratory relief, and asserting a counterclaim for declaratory relief related to the contractual provision underlying PRF’s original drug supply claim. To give the parties an opportunity to discuss a potential negotiated resolution of their dispute, the arbitration was suspended through the end of 2023. As a result, all arbitration activities were on hold, and the final hearing, originally scheduled for May 9 - 12 , 2023, in Boston, Massachusetts, was cancelled. On February 29, 2024, the Company and PRF negotiated and entered into an amended and restated agreement. The amended and restated collaboration agreement with PRF confirms the Company's exclusive right to commercialize Zokinvy subject to terms and conditions of the Agreement. On March 1, 2024, the arbitration was dismissed with prejudice by both parties.
ITEM 4. Mine Safety Disclosures
Not applicable.
PART II
ITEM 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information
On March 22, 2016, Celladon and Private Eiger completed the Merger. Immediately prior to the Merger, Celladon completed a 15-for-1 reverse stock split. Following the Merger, we changed the name of the combined company to Eiger BioPharmaceuticals, Inc. and changed the symbol to “EIGR.” Our common stock originally began trading on The Nasdaq Global Market on January 30, 2014. Prior to January 30, 2014, there was no public market for our common stock.
In January 2024, following approval by the Company’s stockholders at a special meeting of stockholders held in December 2023, our board of directors approved a certificate of amendment to the Company’s amended and restated certificate of incorporation to effect a reverse split of shares of our common stock on a 1-for-30 basis (Reverse Stock Split), which was effected on January 5, 2024. All share data and per share data amounts included in this Annual Report on Form 10-K have been retrospectively adjusted to reflect the effect of the Reverse Stock Split.
On April 2, 2024, the Company received written notice from the Listing Qualifications Department of the Nasdaq Stock Market LLC (Nasdaq) notifying the Company that, as a result of the Chapter 11 Cases and in accordance with Nasdaq Listing Rules 5101, 5110(b) and IM-5101-1, Nasdaq had determined that the Company’s common stock will be delisted from Nasdaq. The Delisting Notice also advises the Company of its right to appeal Nasdaq’s determination pursuant to procedures set forth in Nasdaq Listing Rule 5800 Series. The Company does not intend to pursue an appeal.
Trading of the Company's common stock will be suspended at the opening of business on April 11, 2024. Nasdaq will file a Form 25-NSE with the Securities and Exchange Commission, which will remove the Company's common stock from listing and registration on Nasdaq. As a result, the Company's common stock is expected to begin trading on the over-the counter ("OTC") market on April 11, 2024. On the OTC market, shares of the Company's common stock, which previously traded on the Nasdaq under the symbol EIGR, are expected to trade under the symbol EIGRQ.
Holders of Record
As of April 4, 2024, there were approximately 17 stockholders of record of our common stock. Certain shares are held in “street” name and accordingly, the number of beneficial owners of such shares is not known or included in the foregoing number.
Dividend Policy
We have never declared or paid any dividends on our common stock. We currently intend to retain all available funds and future earnings, if any, to fund the development and expansion of our business, and we do not anticipate paying any cash dividends in the foreseeable future. Any future determination to pay dividends, if permitted, will be made at the discretion of our board of directors.
Securities Authorized for Issuance under Equity Compensation Plans
The following table summarizes our compensation plans under which our equity securities are authorized for issuance as of December 31, 2023:
| | | | | | | | | | | | | | | | | | | | |
| | | (a) Number of Securities to be Issued Upon Exercise of Outstanding Options, Warrants and Rights | | (b) Weighted-Average Exercise Price of Outstanding Options ($/share) | | (c) Number of Shares Remaining Available For Issuance Under Equity Compensation Plans (Excluding Securities Reflected in Column (a)) |
| Plan Category | | (1) | | (2) | | |
Equity compensation plans approved by stockholders(3) | | 186,149 | | $ | 167.68 | | | 228,478 |
Equity compensation plans not approved by stockholders | | — | | — | | | — |
| Total | | 186,149 | | $ | 167.68 | | | 228,478 |
(1)The amount shown in column (a) includes 180,151 outstanding options and 5,998 restricted and performance stock units.
(2)The weighted average exercise price in column (b) includes options only as restricted and performance stock units do not have an exercise price.
(3)As of December 31, 2023, the number of securities remaining available for issuance in column (c) includes 203,891 shares available for future issuance in the form of options or restricted stock, restricted or performance stock units, or restricted stock awards under our Amended and Restated 2013 Equity Incentive Plan and 2021 Inducement Plan and 24,587 shares remained available for future issuance under our 2013 Employee Stock Purchase Plan (ESPP).
Unregistered Sales of Equity Securities
None
Issuer Purchases of Equity Securities
None
ITEM 6. Reserved
ITEM 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis together with our financial statements and related notes included elsewhere in this Annual Report. The following discussion contains forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those expressed or implied in any forward-looking statements as a result of various factors, including those set forth under the caption “Item 1A. Risk Factors.”
Unless otherwise indicated, references to the terms the “combined company,” “Eiger,” the “Company,” “we,” “our” and “us” refer to Eiger BioPharmaceuticals, Inc. (formerly known as Celladon Corporation) and its subsidiaries after the merger described herein. The term “Private Eiger” refers to privately-held Eiger BioPharmaceuticals, Inc. prior to its merger with Celladon Merger Sub, Inc. a wholly-owned subsidiary of Celladon Corporation. The term “Celladon” refers to Celladon Corporation and its subsidiaries prior to the Merger.
Overview
In June 2023, in connection with an extensive portfolio prioritization review, we announced that the Company is focusing its clinical development efforts on advancing avexitide in hyperinsulinemic hypoglycemia (HH) indications. We will continue to commercialize Zokinvy for the treatment of Hutchinson-Gilford progeria syndrome (HGPS) and processing-deficient progeroid laminopathies (PL). In addition, we are evaluating strategic partnering options for our virology assets, lonafarnib and peginterferon lambda. During 2023, we have also undertaken certain cost savings initiatives, including a 55% reduction in workforce to date and efforts to reduce spending related to our hepatitis delta (HDV) development program. In June 2023, we also announced that we had appointed David Apelian, MD, PhD, MBA, who had served as interim CEO since December 2022, as our next CEO.
Avexitide is a well characterized, first-in-class glucagon-like peptide-1 (GLP-1) antagonist and is in development for the treatment of post-bariatric hypoglycemia (PBH) and other forms of hyperinsulinemic hypoglycemia (HH) arising after gastrointestinal surgeries. These disorders are characterized by exaggerated secretion of GLP-1 after meals, dysregulated secretion of insulin, and a rapid drop in blood sugar. Avexitide is the only drug in development for PBH with Breakthrough Therapy designation by the U.S. Food and Drug Administration (FDA). Avexitide is also in development for congenital hyperinsulinism (HI), an ultra-rare, life-threatening, pediatric disorder of persistent hypoglycemia that results in irreversible brain damage in up to 50% of children with the condition. Avexitide has completed Phase 2 for both PBH and HI, and Phase 3 study start-up activities for PBH and HI were initiated. However, further Phase 3 activities have been paused until additional funding is secured. In connection with our Phase 3 start-up activities, we have observed low levels of product-related impurities in the finished drug product. Although not unusual for this class of compounds, we are working with our Contract Manufacturing Organizations (CMOs) to control these materials to ensure that an adequate supply of materials with a sufficient shelf-life can be released for Phase 3 studies. On November 20, 2020, the FDA approved our first commercial product, Zokinvy, to reduce the risk of mortality of HGPS and for treatment of processing-deficient PL with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations. Collectively known as progeria, these are ultra-rare and rapidly fatal genetic conditions of accelerated aging in children. On July 20, 2022, we announced that the EC granted MA under exceptional circumstances for Zokinvy for the treatment of progeria through the centralized procedure. The MA is valid for 5 years from date of authorization in all 27 EU member states plus Iceland, Liechtenstein, and Norway and is subject to annual reassessment of the benefit/risk profile based on the requirements set forth in the MA under exceptional circumstances. In May 2022, the Pharmaceutical Division at The Ministry of Health of Israel granted regulatory approval for Zokinvy in Israel. In August 2022, marketing authorization of Zokinvy was granted by the UK's Medicine and Healthcare products Regulatory Agency (MHRA) as part of the UK's European Commission Decision Reliance Procedure. In January 2024, Eiger and its partner AnGes, Inc. received marketing approval in Japan from the Ministry of Health, Labour and Welfare for Zokinvy for the treatment of HGPS and processing-deficient progeria laminopathies.
We commercially launched Zokinvy in the U.S. in January 2021 and in Europe in November 2022 and started to record product revenue in the first quarter of 2021. Our first European sales were recognized in the fourth quarter of 2022.
We continue to evaluate strategic partnering options for our virology assets: lonafarnib, a first-in-class, oral prenylation inhibitor for HDV, and peginterferon lambda, a potential first-in-class interferon for respiratory viruses such as COVID-19, influenza, and RSV.
For lonafarnib, in June 2023, we completed and reported end of study data from the single, pivotal Phase 3 D-LIVR study in patients with chronic HDV. The end of treatment Week 48 data demonstrated that the primary endpoint was achieved in both the oral and combination lonafarnib treatment arms. 24-week post-treatment data demonstrated that both lonafarnib
arms showed a statistically significant difference in composite response rate compared to placebo. Our pre-New Drug Application (pre-NDA) meeting with FDA was supportive of a potential path to an NDA approval for both oral and combination lonafarnib regimens as finite therapies.
For peginterferon lambda, in September 2023, we announced our decision to discontinue the Phase 3 LIMT-2 study of peginterferon lambda in patients with chronic HDV. The decision was based on the recommendation of the Data Safety Monitoring Board (DSMB) for the study following its quarterly safety review. The DSMB recommended the discontinuation of the LIMT-2 study due to observations of four patients with hepatobiliary events that resulted in liver decompensation. Potential remains for peginterferon lambda for development in respiratory diseases such as COVID-19 and influenza, as well as other virology indications.
We have historically incurred operating losses in each year since inception and we expect to incur losses for the foreseeable future. We had a net loss of $75.0 million and $96.8 million for the years ended December 31, 2023 and 2022, respectively. As of December 31, 2023, we had an accumulated deficit of $512.2 million. Substantially all of our operating losses have resulted from expenses incurred in connection with our research and development programs and from selling, general and administrative costs associated with our operations.
We expect to incur significant expenses and increasing operating losses for at least the next several years as we initiate and continue the clinical development of, and seek regulatory approval for, and potentially commercialize our product candidates, which will require the addition of new personnel and upgrades to our information technology systems. We expect that our operating losses will fluctuate significantly from quarter to quarter and year to year due to timing of clinical development programs and efforts to achieve additional regulatory approvals. We have completed a program prioritization assessment.
Voluntary Filing Under Chapter 11
As previously reported in our Current Report on Form 8-K filed with the U.S. Securities and Exchange Commission (SEC) on April 1, 2024, we commenced voluntary proceedings under Chapter 11 of the United States Bankruptcy Code in the United States Bankruptcy Court for the Northern District of Texas (Bankruptcy Court). The Chapter 11 proceedings are jointly administered under the caption In re Eiger BioPharmaceuticals, Inc., et al, Case No. 24-80040. (Chapter 11 Cases). We continue to operate our business in accordance with the applicable provisions of the Bankruptcy Code and orders of the Bankruptcy Court.
On March 31, 2024, we entered into a “stalking horse” asset purchase agreement (Asset Purchase Agreement) with Sentynl Therapeutics, Inc. (Purchaser), pursuant to which the Purchaser has agreed to acquire substantially all of the rights of the Company to its Zokinvy® program, including the Company’s in-license from Merck Sharp & Dohme Corp. (successor-in-interest of Schering Corporation) (Transferred Assets). The acquisition of the Transferred Assets by the Purchaser pursuant to the Asset Purchase Agreement is subject to approval of the Bankruptcy Court and one or more auctions, if necessary, to solicit higher or otherwise better bids. On April 1, 2024, we filed a motion (Bidding Procedures Motion) seeking approval of, among other things, certain bidding procedures (Bidding Procedures), which will establish procedures for the selection of the highest or otherwise best bids for the sale of the Transfer Assets and other assets. Other interested bidders would be permitted to participate in the auction if they submit qualifying bids that are higher or otherwise better than the Asset Purchase Agreement. The Asset Purchase Agreement acts as a baseline for competitive bids for the acquisition of the Transferred Assets. The Bidding Procedures Motion additionally seeks the Bankruptcy Court’s approval of the Asset Purchase Agreement and designation of the Purchaser as the “stalking horse” bidder for the Transferred Assets.
Under the Asset Purchase Agreement, the Purchaser has agreed, subject to the Bankruptcy Court’s approval and absent any higher or otherwise better bid, to acquire the Transferred Assets from the Debtors for $26 million, subject to certain adjustments, including per diem reductions if the sale closes after April 24, 2024, in accordance with the terms and conditions of the Asset Purchase Agreement, plus the assumption of specified liabilities related to the Transferred Assets. The Asset Purchase Agreement includes customary representations and warranties and various customary covenants under the circumstances that are subject to certain limitations, including, without limitation, a termination fee, expense reimbursement and the right to designate executory contracts and unexpired leases to assume or reject. On April 3, 2024, the Purchaser increased the purchase price to $30 million, subject to certain adjustments, including per diem reductions if the sale closes after April 24, 2024.
Recent Developments
Reverse Stock Split
In January 2024, following approval by the Company’s stockholders at a special meeting of stockholders held in December 2023, our board of directors approved a certificate of amendment to the Company’s amended and restated certificate of incorporation to effect a reverse split of shares of our common stock on a 1-for-30 basis (Reverse Stock Split), which was effected on January 5, 2024. All share data and per share data amounts included in this Annual Report on Form 10-K have been retrospectively adjusted to reflect the effect of the Reverse Stock Split.
Financial Operations Overview
Product Revenue, Net
Our product revenue, net consists of sales of Zokinvy® (lonafarnib) for HGPS and processing-deficient progeroid laminopathies in the United States and under a reimbursed early access program, or cohort ATU program, in France. In November 2022, sales of Zokinvy launched commercially in Europe through our wholly owned subsidiary in Ireland.
Other Revenue
Other revenue consists of milestone payments from the MDA with AnGes, Inc., which was executed in May 2022.
Cost of Sales
Cost of sales consists primarily of direct and indirect costs related to the manufacturing of Zokinvy for commercial sale, including third-party manufacturing costs, third party logistics costs, write down of inventories, and other period costs.
Research and Development Expenses
Research and development expenses represent costs incurred to conduct research and development, such as the development of our product candidates. We recognize all research and development costs as they are incurred. Research and development expenses consist primarily of the following:
•expenses incurred under agreements with consultants, contract research organizations and clinical trial sites that conduct research and development activities on our behalf;
•laboratory and vendor expenses related to the execution of clinical trials;
•contract manufacturing expenses, primarily for the production of clinical trial supplies;
•license fees associated with our license agreements; and
•internal costs that are associated with activities performed by our research and development organization and generally benefit multiple programs. These costs are not separately allocated by product candidate. Unallocated internal research and development costs consist primarily of:
◦personnel costs, which include salaries, benefits and stock-based compensation expense;
◦allocated facilities and other expenses, which include expenses for rent and maintenance of facilities and depreciation expense; and
◦regulatory expenses and technology license fees related to development activities.
The largest component of our operating expenses has historically been the investment in clinical trials, including contract manufacturing arrangements, clinical trial material related costs and other research and development activities. However, we do not allocate internal research and development costs, such as salaries, benefits, stock-based compensation expense
and indirect costs to product candidates on a program-specific basis. The following table shows our research and development expenses for the years ended December 31, 2023 and 2022 (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Product candidates: | | | |
| Lonafarnib | $ | 21,816 | | | $ | 25,643 | |
| Peginterferon Lambda | 16,688 | | | 21,563 | |
| Avexitide | 6,178 | | | 9,596 | |
| Internal research and development costs | 17,649 | | | 18,480 | |
| Total research and development expense | $ | 62,331 | | | $ | 75,282 | |
We expect research and development expenses will continue to be significant and may increase in the future as we advance our product candidates into and through later stage clinical trials and pursue regulatory approvals, which will require a significant investment in regulatory support and contract manufacturing and clinical trial material related costs. In addition, in the future we may evaluate opportunities to acquire or in-license other product candidates and technologies, which may result in higher research and development expenses due to license fees and/or milestone payments.
The process of conducting clinical trials necessary to obtain regulatory approval is costly and time consuming. We may never succeed in timely developing and achieving regulatory approval for our product candidates. The probability of success of our product candidates may be affected by numerous factors, including clinical data, competition, intellectual property rights, manufacturing capability and commercial viability. As a result, we are unable to determine the duration and completion costs of our development projects or when and to what extent we will generate revenue from the commercialization and sale of any of our product candidates.
Selling, General and Administrative Expenses
Selling, general and administrative expenses consist of personnel costs, allocated expenses, expenses for outside professional services, including legal, audit, accounting services, insurance costs and costs associated with being a public company, and commercial related expenses. Personnel costs consist of salaries, benefits and stock-based compensation. Allocated expenses consist of facilities and other expenses, which include direct and allocated expenses for rent and maintenance of facilities, depreciation expense and other supplies. Our expenses include costs related to compliance with the rules and regulations of the SEC, insurance, investor relations, banking fees and other administrative expenses and professional services. We expect our selling, general and administrative expenses to increase in the future due to sales and marketing activities from the commercialization of our product candidates.
Interest Expense
Interest expense consists of interest on our long-term borrowings.
Interest Income
Interest income consists of interest earned on our investments in debt securities and cash equivalents.
Other (expense) income, net
Other (expense) income, net primarily consists of foreign currency translation loss from the significant volatility of the foreign currency exchange rates during the year ended December 31, 2023. Other (expense) income, net during the year ended December 31, 2022 primarily consists of the loss on extinguishment of debt.
Critical Accounting Policies and Estimates
Our management’s discussion and analysis of financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with generally accepted accounting principles in the United States. The preparation of these consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities revenues, and expenses. On an ongoing basis, we evaluate these estimates and
judgments. We base our estimates on historical experience and on various assumptions that we believe to be reasonable under the circumstances. These estimates and assumptions form the basis for making judgments about the carrying values of assets and liabilities and the recording of revenues and expenses that are not readily apparent from other sources. Actual results may differ materially from these estimates. We believe that the accounting policies discussed below are critical to understanding our historical and future performance, as these policies relate to the more significant areas involving management’s judgments and estimates.
Accrued Research and Development Costs
We accrue for estimated costs of research and development activities conducted by third-party service providers, which include the conduct of preclinical and clinical studies, and contract manufacturing activities. We record the estimated costs of research and development activities based upon the estimated services provided but not yet invoiced and include these costs in accrued liabilities in the consolidated balance sheets and within research and development expenses in the consolidated statements of operations. We accrue for these costs based on factors such as estimates of the work completed made by our internal personnel and third-party service providers and in accordance with agreements established with our third-party service providers. We make judgments and estimates in determining the accrued liabilities balance in each reporting period and although we do not expect our estimate to be materially different from amounts actually incurred, as actual costs become known, we adjust our accrued liabilities. Through December 31, 2023, there have been no material differences from our accrued estimated expenses to the actual clinical trial and manufacturing development expenses.
Product Revenue
We recognize product revenue when a customer obtains control of our product, which occurs at a point in time, typically upon delivery to a customer as the delivery of the product is our only performance obligation. Shipping and handling activities are fulfillment activities rather than a separate performance obligation and are recorded in cost of sales.
Product revenue is recorded at the net sales price (transaction price), which includes estimates of variable consideration resulting from rebates, prompt payment discounts, co-payment assistance, and returns. Amounts related to such items are estimated at contract inception and updated at the end of each reporting period as additional information becomes available. The amount of variable consideration may be constrained and is included in the transaction price only to the extent it is probable that a significant reversal of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is resolved. Product revenue is recorded after considering the impact of the following variable consideration amounts along with the constraint at the time of revenue recognition:
Rebates: Our product is subject to government mandated rebates for Medicaid Drug Rebate Program, Medicare Part D Prescription Drug Benefit Program, and other government health care programs in the United States, and other countries. Rebate amounts are based upon contractual agreements or legal requirements with public sector benefit providers. We use the expected-value method for estimating these rebates based on statutory discount rates and expected utilization. The expected utilization of rebates is estimated based on expected coverage of identified patients. Estimates for these rebates are adjusted quarterly to reflect the most recent information. We record an accrued liability for unpaid rebates related to products for which control has been transferred to a customer.
Prompt payment discounts: We provide a discount to a customer if it pays for purchases within 30 days. We expect that our customers will earn prompt payment discounts and use the most likely amount method for estimating such discounts. As a result, when revenues are recognized, we deduct the full amount of the prompt payment discounts from total product revenues and record these discounts as a reduction of accounts receivable.
Co-payment assistance: We provide co-payment assistance to patients who have commercial insurance and meet certain eligibility requirements. We use the expected-value method for estimating co-payment assistance based on estimates of program redemption using data provided by third-party administrators. Estimates for the co-payment assistance are adjusted quarterly to reflect actual experience. We record an accrued liability for unredeemed co-payment assistance related to products for which control has been transferred to a customer.
Product returns: We offer limited product return rights and generally allow for the return of product that is damaged or defective, or within a few months prior to and up to a few months after the product expiration date. We consider several factors in the estimation of potential product returns, including expiration dates of the product shipped, the limited product return rights, third-party data in monitoring channel inventory levels, shelf life of the product, and other relevant factors.
Stock-Based Compensation
We recognize compensation costs related to stock options and restricted stock units based on the estimated fair value of the awards on the grant date. We estimate the grant date fair value of stock options, and the resulting stock-based compensation expense, using the Black-Scholes option-pricing model. The grant date fair value of the stock-based awards is generally recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the respective awards. We record forfeitures when they occur.
The Black-Scholes option-pricing model includes the following assumptions:
Expected Term. The expected term represents the number of years that we estimate, based primarily on historical experience, that the options will be outstanding prior to exercise.
Expected Volatility. The expected volatility for stock options is based on the Company's historical stock price volatility.
Risk-Free Interest Rate. The risk-free interest rate is based on the U.S. Treasury zero coupon issues in effect at the time of grant for periods corresponding with the expected term of option.
Expected Dividend. We have never paid dividends on our common stock and have no plans to pay dividends on our common stock. Therefore, we used an expected dividend yield of zero.
We use the contractual term to determine the non-employee awards’ fair value at the grant date. The contractual term of options granted under the Plan is 10 years. Our Board of Directors determined the fair value of each share of underlying common stock based on the closing price of our common stock as reported by the Nasdaq Global Market on the date of grant.
We estimate the fair value of restricted stock units based on the closing market price of our common stock on the date of grant and the resulting stock-based compensation expense is recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the awards.
The estimated fair value of equity awards that contain performance conditions is expensed over the term of the award once we have determined that it is probable that performance conditions will be satisfied.
Results of Operations
Comparison of the Years Ended December 31, 2023 and 2022
The following table summarizes results of operations for the years ended December 31, 2023 and 2022 (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | $
Change | | %
Change |
| 2023 | | 2022 | | |
| Product revenue, net | $ | 15,523 | | | $ | 12,734 | | | $ | 2,789 | | | 22 | % |
| Other revenue | 250 | | | 750 | | | (500) | | | (67) | % |
| Total revenue | 15,773 | | | 13,484 | | | 2,289 | | | 17 | % |
| Costs and operating expenses: | | | | | | | |
| Cost of sales | 24 | | | 1,837 | | | (1,813) | | | (99) | % |
| Research and development | 62,331 | | | 75,282 | | | (12,951) | | | (17) | % |
| Selling, general and administrative | 25,204 | | | 29,105 | | | (3,901) | | | (13) | % |
| Total costs and operating expenses | 87,559 | | | 106,224 | | | (18,665) | | | (18) | % |
| Loss from operations | (71,786) | | | (92,740) | | | 20,954 | | | (23) | % |
| Interest expense | (5,466) | | | (4,132) | | | (1,334) | | | 32 | % |
| Interest income | 2,208 | | | 1,082 | | | 1,126 | | | 104 | % |
| Other (expense) income, net | 87 | | | (963) | | | 1,050 | | | (109) | % |
| Loss before provision for income taxes | (74,957) | | | (96,753) | | | 21,796 | | | (23) | % |
| Provision for income taxes | 6 | | | 23 | | | (17) | | | (74) | % |
| Net loss | $ | (74,963) | | | $ | (96,776) | | | $ | 21,813 | | | (23) | % |
*Percentage not meaningful or not material. Product revenue, net
Product revenue, net increased by $2.8 million to $15.5 million for the year ended December 31, 2023, from $12.7 million for the same period in 2022. The increase was primarily due to an increase of $1.9 million in sales under the ATU program in France and a $1.2 million increase in product sales in Germany during the period. It was partially offset by a decrease of $0.5 million in U.S. sales.
Other revenue
Other revenue decreased by $0.5 million to $0.3 million for the year ended December 31, 2023, from $0.8 million for the same period in 2022. The decrease was primarily due to the timing of certain upfront payments received under the terms of the MDA with AnGes.
Cost of sales
Cost of sales decreased by $1.8 million to $24,000 for the year ended December 31, 2023, from $1.8 million for the same period in 2022. The decrease was primarily due to a non-conforming batch of inventory that was written off during the year ended December 31, 2022 and a reversal of an inventory accrual related to a non-conforming batch of product where we were notified in the second quarter of 2023 that we were no longer obligated to pay for such product.
Research and development expenses
Research and development expenses decreased by $13.0 million to $62.3 million for the year ended December 31, 2023, from $75.3 million for the same period in 2022. The decrease was primarily due to a $5.0 million decrease in milestone expenses related to the Phase 3 LIMT-2 study of peginterferon lambda for HDV under our License Agreement with Bristol-Myers Squibb Company, which occurred in March 2022, a $3.5 million decrease in outside services across programs including consulting and advisory services due to a decline in spending on peginterferon lambda programs, a $3.3 million decrease in clinical and contract manufacturing expenditures due to decline in manufacturing costs for peginterferon lambda and avexitide, and a $1.0 million decrease in compensation and personnel related expenses, including
$0.9 million in severance payouts for terminated employees resulting from a reduction in workforce. It was partially offset by a $0.3 million increase in regulatory expenses.
Selling, general and administrative expenses
Selling, general and administrative expenses decreased by $3.9 million to $25.2 million for the year ended December 31, 2023, from $29.1 million for the same period in 2022. The decrease was primarily due to a $3.7 million decrease in compensation and personnel related expense, including severance payouts for terminated employees resulting from a reduction in workforce and a $0.3 million decrease in sales and marketing expense. This was partially offset by a $0.1 million increase in outside services, including consulting, advisory and accounting services.
Interest expense
Interest expense increased by $1.3 million compared to the same period in 2022. The increase was primarily due to an increase in the outstanding principal balance and increase in the effective interest rate of our long-term borrowings related to the Innovatus Loan.
Interest income
Interest income increased by $1.1 million compared to the same period in 2022. The increase was primarily due to an increase in interest rates.
Other (expense) income, net
Other (expense) income, net changed by $1.1 million compared to the same period in 2022. The change was primarily due to a loss on early extinguishment of the Oxford loan in 2022.
Provision for income taxes
Provision for income taxes decreased by $17,000 compared to the same period in 2022. The change was primarily due to the tax expense related to state taxes.
Liquidity and Capital Resources
Voluntary Filing Under Chapter 11
We expect to continue operations in the normal course for the duration of the Chapter 11 Cases. To ensure ordinary course operations, we have obtained approval from the Bankruptcy Court for certain “first day” motions to continue our ordinary course operations after the filing date. We will continue to fund current operations using our current cash balance. However, for the duration of our Chapter 11 Cases, our operations and our ability to develop and execute our business plan, our financial condition, our liquidity and our continuation as a going concern are subject to a high degree of risk and uncertainty associated with our Chapter 11 Cases. The outcome of the Chapter 11 Cases is dependent upon factors that are outside of our control, including actions of the Bankruptcy Court.
Sources of Liquidity
As of December 31, 2023, we had $25.4 million of cash and cash equivalents, and an accumulated deficit of $512.2 million.
On June 1, 2022, we entered into the Innovatus Loan with Innovatus, providing for up to $75.0 million funded in three tranches with a maturity date of August 31, 2027. We were funded $40.0 million in June 2022 at the closing, of which approximately $33.5 million was used to pay off the Oxford Loan. We recorded a loss of $1.1 million on early extinguishment of the debt which was recognized as a component of Other (expense) income, net in the consolidated statement of operations and comprehensive loss. As part of the agreement with Innovatus, we are required to maintain a cash balance of not less than 5% of the aggregate principal amount of funded and outstanding term loans at all times. The Innovatus Loan provides that upon the filing of the Bankruptcy Petitions, the principal amount together with accrued and unpaid fees and interest thereon shall become immediately due and payable. However, any efforts to enforce such payment
obligations under the Innovatus Loan are automatically stayed as a result of the Bankruptcy Petitions, and the creditors’ rights of enforcement in respect of the Innovatus Loan are subject to the applicable provisions of the Bankruptcy Code.
On March 25, 2022, we entered into an Open Market Sale AgreementSM with Jefferies, pursuant to which we can sell up to a maximum of $50.0 million of our common stock in offerings that are deemed “at the market” offerings as defined in Rule 415 under the Securities Act, under Eiger’s currently effective shelf registration statement (the 2022 ATM Facility). In April 2022, we completed offerings from the 2022 ATM facility for a total of 89,539 shares (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) of our common stock resulting in net proceeds of $20.8 million, after deducting commissions costs. No additional offerings have been completed since April 2022. We did not sell shares under the 2022 ATM Facility during 2023. The registration statement registering the offer and sale of shares pursuant to the 2022 ATM Facility expired in December 2023. To the extent that we raise additional capital through the sale of equity, including offerings of debt or other securities convertible into equity, stockholder ownership interest will be diluted, and the terms of these new securities may include liquidation or other preferences that adversely affect the rights of holders of our common stock. In addition, based on our public float as of the filing of this Annual Report on Form 10-K, we are only permitted to file a shelf registration statement subject to Instruction I.B.6 to Form S-3, which is referred to as the “baby shelf” rule. For so long as our public float is less than $75.0 million, we may not sell more than the equivalent of one-third of our public float during any 12 consecutive months pursuant to the baby shelf rules.
Based on our recurring losses from operations incurred since inception, expectation of continuing operating losses and negative cash flows for the foreseeable future, and need to raise additional capital to finance our future operations, management has concluded that there is substantial doubt regarding Eiger's ability to continue as a going concern beyond twelve months after the date that our consolidated financial statements are issued, which was a principal reason for our decision to seek bankruptcy protection under Chapter 11.
Our primary uses of cash are to fund operating expenses, including research and development expenditures and selling, general and administrative expenditures. Cash used to fund operating expenses is impacted by the timing of when we pay these expenses, as reflected in the change in outstanding accounts payable and accrued expenses.
Future Funding Requirements
Prior to 2021, we had not generated any product revenue. We launched our first commercial product, Zokinvy, in January 2021. We expect our expenses to increase in connection with our ongoing development and manufacturing activities, particularly as we continue the research, development, manufacture and clinical trials of, and seek regulatory approval for our product candidates.
Our primary uses of capital are, and we expect will continue to be, sales and marketing costs for commercialization of Zokinvy, compensation and related expenses, operational, manufacturing, financial, and management personnel, and costs associated with operating as a public company. We expect to incur substantial expenditures in the foreseeable future for the development and potential commercialization of our product candidates. We have reduced our workforce by 55% to date and have reduced spending related to our HDV development program in an effort to extend our cash runway. We are actively seeking additional capital to fund losses from operations and capital funding needs through public or private equity offerings and/or debt financings, including through potential additional collaborations or strategic partnerships with other companies, or through a combination of the foregoing. As a result of economic conditions, general global economic uncertainty, political change and other factors, we do not know whether additional capital will be available when needed, or that, if available, we will be able to obtain additional capital on reasonable terms. If we are unable to raise additional capital due to the volatile global financial markets, general economic uncertainty or other factors, we may need to curtail planned development or commercialization activities. The sale of additional equity, or convertible debt could result in additional dilution to our stockholders. The incurrence of indebtedness would result in debt service obligations and could result in operating and financing covenants that would restrict our operations. We can provide no assurance that financing will be available in the amounts we need or on terms acceptable to us, if at all. If we are not able to secure adequate additional funding we may be forced to delay, make reductions in spending, extend payment terms with suppliers, liquidate assets where possible, and/or suspend or curtail planned programs. Any of these actions could materially harm our business.
Contractual Obligations and Other Commitments
Leases
In October 2017, we entered into a non-cancelable operating facility lease agreement commencing on March 1, 2018 and expiring in February 2023. In February 2023, we amended the lease to extend the lease by one year with a one year renewal option. The extended lease commenced on March 1, 2023 and expired on February 28, 2024. In February 2024, we amended the lease to extend the lease term for an additional month from March 1, 2024 to April 1, 2024 with a month to month option to renew. We also have additional operating leases that are not considered significant for disclosure.
Asset and License Agreements
We are obligated to make future payments to third parties under asset purchase and license agreements, including royalties and payments that become due and payable on the achievement of certain development and commercialization milestones. Refer to Note 5 and Note 6 in our consolidated financial statements included within Item 8 of this Annual Report on Form 10-K for a description of our contractual obligations as of December 31, 2023.
Product Development Agreements
On August 11, 2018, we entered into a Product Development Agreement and a First Project Agreement (the Product Agreements), pursuant to which we will receive development program support services for the HDV program. The services are to be provided from July 1, 2018 through the new drug application (NDA) filing. The Product Agreements can be terminated by either party due to material breach or by us without cause with 90 days prior written notice. Refer to Note 5 of our consolidated financial statements included within Item 8 of this Annual Report on Form 10-K for additional details.
Innovatus Term Loan
On June 1, 2022, we entered the Innovatus Loan, providing for up to $75.0 million funded in three tranches with a maturity date of August 31, 2027. The floating per annum interest rate of the Innovatus Loan is equal to the sum of (a) the greater of (i) the Prime Rate published in the Money Rates section of the Wall Street Journal (or any successor thereto) and (ii) 3.5%, plus (b) 3.75%; provided that, at the election of the Borrower, up to 2.25% of such rate shall be payable in-kind until the third anniversary of the closing date. We received $40.0 million in June 2022 on the closing date under Tranche A. Given the event of default was triggered upon our bankruptcy petition filing, no additional tranches are available to the Company.
Additionally, in connection with entering into the Innovatus Loan, we entered into a Stock Purchase Agreement with Innovatus for the sale of common stock with an aggregate value of $5.0 million. On June 1, 2022, we issued 24,967 shares of common stock to Innovatus at a per share purchase price of $200.25, the preceding five-day volume weighted average price per share (in each case as adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024).
A portion of the loan proceeds was used to repay in full the approximately $33.5 million of aggregate principal amount, unpaid interest, and exit fees in connection with our loans then outstanding and owed to Oxford Finance LLC. Refer to Note 7 of our consolidated financial statements included within Item 8 of this Annual Report on Form 10-K for additional details.
Off-Balance Sheet Arrangements
During the periods presented, we did not have, nor do we currently have, any off-balance sheet arrangements as defined under the rules of the SEC and do not have any holdings in variable interest entities.
Cash Flows
The following table summarizes our cash flows for the periods indicated (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Net cash used in operating activities | $ | (74,548) | | | $ | (83,010) | |
| Net cash provided by investing activities | 74,228 | | | 9,632 | |
| Net cash provided by financing activities | 41 | | | 76,955 | |
| Effect of foreign exchange on cash and cash equivalents | (79) | | | — | |
Net (decrease) increase in cash and cash equivalents | $ | (358) | | | $ | 3,577 | |
Cash flows from operating activities
Cash used in operating activities for the year ended December 31, 2023 was $74.5 million, which primarily consisted of a net loss of $75.0 million, stock-based compensation expense of $5.0 million, non-cash interest expense of $1.5 million, amortization of debt securities discount of $1.1 million, reduction in the carrying amount of right-of-use assets of $0.5 million, and depreciation and amortization of $0.3 million. Additionally, cash used in operating activities reflected changes in net operating assets due to a decrease of $13.0 million in accounts payable and accrued liabilities primarily due to the timing of payments and a decrease of $0.5 million in operating lease liabilities partially offset by a decrease of $4.2 million in prepaid expenses and other current assets, a decrease of $1.9 million in other assets and a decrease of $1.6 million in inventories.
Cash used in operating activities for the year ended December 31, 2022 was $83.0 million, which primarily consisted of a net loss of $96.8 million, stock-based compensation expense of $8.3 million, loss on extinguishment of long-term debt of $1.1 million, inventory write down of $1.0 million, non-cash interest expense of $1.2 million, amortization of debt securities discount of $0.7 million, amortization of operating lease right-of-use assets of $0.6 million and depreciation and amortization of $0.3 million. Additionally, cash used in operating activities reflected changes in net operating assets due to an increase of $3.2 million in accounts payable and accrued liabilities primarily due to the timing of payments and a decrease of $0.8 million in accounts receivable partially offset by an increase of $1.4 million in inventories, an increase of $1.3 million in other assets, a decrease of $0.6 million in operating lease liabilities and an increase of $0.2 million in prepaid expenses and other current assets.
Cash flows from investing activities
Cash provided by investing activities for the year ended December 31, 2023 was $74.2 million, primarily consisting of $93.9 million of proceeds from maturities of debt securities, which were partially offset by $19.4 million of purchases of debt securities and $0.2 million of purchases of property and equipment.
Cash provided by investing activities for the year ended December 31, 2022 was $9.6 million, primarily consisting of $85.1 million of proceeds from maturities of debt securities, which were partially offset by $75.1 million of purchases of debt securities.
Cash flows from financing activities
Cash provided by financing activities for the year ended December 31, 2023 was $41,000, primarily consisting of $56,000 proceeds from issuance of common stock upon ESPP purchases, which were partially offset by $15,000 payment of common stock offering costs.
Cash provided by financing activities for the year ended December 31, 2022 was $77.0 million, which primarily consisted of $66.4 million of proceeds net of commissions from the issuance of common stock under the 2020 and 2022 ATM Facilities, $39.8 million of proceeds from debt, $5.0 million of proceeds from issuance of common stock to lender, $0.4 million proceeds from issuance of common stock upon stock option exercises and ESPP purchases, which were partially offset by the $33.3 million repayment of debt, $1.1 million payment of debt issuance costs and $0.3 million of common stock offering costs.
Recent Accounting Pronouncements
Refer to Note 2 to our consolidated financial statements included within Item 8 of this Annual Report on Form 10-K for a description of recent accounting pronouncements adopted and not yet adopted for the year ended December 31, 2023.
ITEM 7A. Quantitative and Qualitative Disclosures about Market Risk
We are a smaller reporting company as defined by Rule 12b-2 of the Securities Exchange Act of 1934 and are not required to provide the information under this item.
ITEM 8. Financial Statements and Supplementary Data
INDEX TO FINANCIAL STATEMENTS
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors
Eiger BioPharmaceuticals, Inc.:
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of Eiger BioPharmaceuticals, Inc. and subsidiaries (the Company) as of December 31, 2023 and 2022, the related consolidated statements of operations, comprehensive loss, stockholders’ (deficit) equity, and cash flows for each of the years in the two-year period ended December 31, 2023, and the related notes (collectively, the consolidated financial statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the years in the two-year period ended December 31, 2023, in conformity with U.S. generally accepted accounting principles.
Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company’s recurring losses from operations, expectation of continuing operating losses and negative cash flows, and the need to raise additional capital to finance its future operations, raises substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
Critical audit matters are matters arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. We determined that there are no critical audit matters.
We have served as the Company’s auditor since 2015.
San Francisco, California
April 8, 2024
Eiger BioPharmaceuticals, Inc.
Consolidated Balance Sheets
(In thousands)
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Assets | | | |
| Current assets: | | | |
| Cash and cash equivalents | $ | 25,440 | | | $ | 25,798 | |
| Short-term debt securities | — | | | 73,150 | |
| Accounts receivable, net | 1,682 | | | 1,749 | |
Inventories, net | 1,556 | | | 2,853 | |
| Prepaid expenses and other current assets | 9,161 | | | 13,985 | |
| Total current assets | 37,839 | | | 117,535 | |
| Property and equipment, net | 627 | | | 696 | |
| Other assets | 360 | | | 1,908 | |
| Total assets | $ | 38,826 | | | $ | 120,139 | |
Liabilities and Stockholders’ (Deficit) Equity | | | |
| Current liabilities: | | | |
| Accounts payable | $ | 2,115 | | | $ | 8,975 | |
| Accrued liabilities | 9,966 | | | 15,655 | |
| Current portion of operating lease liabilities | 84 | | | 491 | |
Current portion of long-term debt | 41,119 | | | — | |
| Total current liabilities | 53,284 | | | 25,121 | |
Debt, net | — | | | 39,625 | |
| Operating lease liabilities | — | | | 83 | |
| Total liabilities | 53,284 | | | 64,829 | |
Commitments and contingencies (Note 12) | | | |
Stockholders’ (deficit) equity: | | | |
Common stock, $0.001 par value, 200,000,000 shares authorized as of December 31, 2023 and 2022; 1,479,483 and 1,469,149 shares issued and outstanding as of December 31, 2023 and 2022, respectively | 1 | | | 1 | |
| Additional paid-in capital | 497,866 | | | 492,802 | |
| Accumulated other comprehensive loss | (169) | | | (300) | |
| Accumulated deficit | (512,156) | | | (437,193) | |
Total stockholders’ (deficit) equity | (14,458) | | | 55,310 | |
Total liabilities and stockholders’ (deficit) equity | $ | 38,826 | | | $ | 120,139 | |
* All periods presented have been retroactively adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024. Refer to Note 1 for further information.
The accompanying notes are an integral part of these consolidated financial statements.
Eiger BioPharmaceuticals, Inc.
Consolidated Statements of Operations
(In thousands, except share and per share amounts)
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Product revenue, net | $ | 15,523 | | | $ | 12,734 | |
| Other revenue | 250 | | | 750 | |
| Total revenue | 15,773 | | | 13,484 | |
| Costs and operating expenses: | | | |
| Cost of sales | 24 | | | 1,837 | |
| Research and development | 62,331 | | | 75,282 | |
| Selling, general and administrative | 25,204 | | | 29,105 | |
| Total costs and operating expenses | 87,559 | | | 106,224 | |
| Loss from operations | (71,786) | | | (92,740) | |
| Interest expense | (5,466) | | | (4,132) | |
| Interest income | 2,208 | | | 1,082 | |
| Other (expense) income, net | 87 | | | (963) | |
| Loss before provision for income taxes | (74,957) | | | (96,753) | |
| Provision for income taxes | 6 | | | 23 | |
| Net loss | $ | (74,963) | | | $ | (96,776) | |
Net loss per common share: | | | |
Basic and diluted | $ | (50.78) | | | $ | (69.74) | |
Weighted-average common shares outstanding: | | | |
Basic and diluted | 1,476,247 | | | 1,387,632 | |
* All periods presented have been retroactively adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024. Refer to Note 1 for further information
The accompanying notes are an integral part of these consolidated financial statements.
Eiger BioPharmaceuticals, Inc.
Consolidated Statements of Comprehensive Loss
(In thousands)
| | | | | | | | | | | |
| Year Ended December 31, |
| | 2023 | | 2022 |
| Net loss | $ | (74,963) | | | $ | (96,776) | |
| Other comprehensive loss: | | | |
Unrealized gain (loss) on available-for-sale debt securities, net | 200 | | | (51) | |
| Foreign currency translation adjustment | (69) | | | (100) | |
| Comprehensive loss | $ | (74,832) | | | $ | (96,927) | |
The accompanying notes are an integral part of these consolidated financial statements.
Eiger BioPharmaceuticals, Inc.
Consolidated Statements of Stockholders’ (Deficit) Equity
(In thousands, except share amounts)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Common Stock | | Additional Paid-In Capital | | Accumulated Other Comprehensive Income (Loss) | | Accumulated Deficit | | Total Stockholders’ (Deficit) Equity |
| | Shares | | Amount | | | | |
Balance at December 31, 2021 | 1,152,350 | | | $ | 1 | | | $ | 412,964 | | | $ | (149) | | | $ | (340,417) | | | $ | 72,399 | |
Issuance of common stock upon offering at-the-market, net of $2,068 of issuance costs | 284,262 | | | — | | | 66,110 | | | — | | | — | | | 66,110 | |
| Issuance of common stock upon exercise of stock options | 1,573 | | | — | | | 234 | | | — | | | — | | | 234 | |
| Issuance of common stock to lender | 24,967 | | | — | | | 5,000 | | | — | | | — | | | 5,000 | |
| Vesting of common stock issued under Product Development Agreement | — | | | — | | | 19 | | | — | | | — | | | 19 | |
| Issuance of common stock upon ESPP purchase | 1,594 | | | — | | | 158 | | | — | | | — | | | 158 | |
| Issuance of common stock upon release of restricted stock units | 4,403 | | | — | | | — | | | — | | | — | | | — | |
| Stock-based compensation expense | — | | | — | | | 8,317 | | | — | | | — | | | 8,317 | |
Unrealized loss on available-for-sale debt securities, net | — | | | — | | | — | | | (51) | | | — | | | (51) | |
| Cumulative translation adjustment | — | | | — | | | — | | | (100) | | | — | | | (100) | |
| Net loss | — | | | — | | | — | | | — | | | (96,776) | | | (96,776) | |
Balance at December 31, 2022 | 1,469,149 | | | 1 | | | 492,802 | | | (300) | | | (437,193) | | | 55,310 | |
| Issuance of common stock upon ESPP purchase | 1,709 | | | — | | | 56 | | | — | | | — | | | 56 | |
Issuance of common stock upon release of restricted stock units and performance stock units | 8,625 | | | — | | | — | | | — | | | — | | | — | |
| Stock-based compensation expense | — | | | — | | | 5,008 | | | — | | | — | | | 5,008 | |
Unrealized gain on available-for-sale debt securities, net | — | | | — | | | — | | | 200 | | | — | | | 200 | |
| Cumulative translation adjustment | — | | | — | | | — | | | (69) | | | — | | | (69) | |
| Net loss | — | | | — | | | — | | | — | | | (74,963) | | | (74,963) | |
Balance at December 31, 2023 | 1,479,483 | | | $ | 1 | | | $ | 497,866 | | | $ | (169) | | | $ | (512,156) | | | $ | (14,458) | |
* All periods presented have been retroactively adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024. Refer to Note 1 for further information
The accompanying notes are an integral part of these consolidated financial statements
Eiger BioPharmaceuticals, Inc.
Consolidated Statements of Cash Flows
(In thousands)
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Operating activities | | | |
| Net loss | $ | (74,963) | | | $ | (96,776) | |
| Adjustments to reconcile net loss to net cash used in operating activities: | | | |
| Depreciation and amortization | 275 | | | 292 | |
| Inventory write down | 8 | | | 1,043 | |
| Net amortization of premiums and accretion of discounts on investments | (1,112) | | | 682 | |
| Loss on extinguishment of debt | — | | | 1,144 | |
| Non-cash interest expense | 1,494 | | | 1,238 | |
| Reduction in the carrying amount of right-of-use assets | 475 | | | 550 | |
| Common stock issued under Product Development Agreement | — | | | 19 | |
| Stock-based compensation | 5,008 | | | 8,317 | |
| Change in operating assets and liabilities: | | | |
| Accounts receivable | 84 | | | 828 | |
| Inventories | 1,626 | | | (1,367) | |
| Prepaid expenses and other current assets | 4,169 | | | (196) | |
| Other assets | 1,857 | | | (1,265) | |
| Accounts payable | (6,835) | | | 1,152 | |
| Accrued liabilities | (6,145) | | | 1,957 | |
| Operating lease liabilities | (489) | | | (628) | |
| Net cash used in operating activities | (74,548) | | | (83,010) | |
| Investing activities | | | |
| Purchase of debt securities available-for-sale | (19,388) | | | (75,101) | |
| Proceeds from maturities of debt securities available-for-sale | 93,850 | | | 85,073 | |
| Purchase of property and equipment | (234) | | | (340) | |
| Net cash provided by investing activities | 74,228 | | | 9,632 | |
| Financing activities | | | |
| Issuance of common stock upon offering at-the-market, net of commissions | — | | | 66,402 | |
| Proceeds from issuance of common stock to lender | — | | | 5,000 | |
| Proceeds from debt | — | | | 39,841 | |
| Repayment of debt | — | | | (33,277) | |
| Proceeds from issuance of common stock upon stock option exercises | — | | | 234 | |
| Proceeds from issuance of common stock upon ESPP purchase | 56 | | | 157 | |
| Payment of debt issuance costs | — | | | (1,116) | |
| Common stock offering costs | (15) | | | (286) | |
| Net cash provided by financing activities | 41 | | | 76,955 | |
Effect of foreign exchange on cash and cash equivalents | (79) | | | — | |
Net (decrease) increase in cash and cash equivalents | (358) | | | 3,577 | |
| Cash and cash equivalents at beginning of the year | 25,798 | | | 22,221 | |
| Cash and cash equivalents at end of the year | $ | 25,440 | | | $ | 25,798 | |
| | | |
| Supplemental disclosure of cash flow information: | | | |
| Interest paid | $ | 3,972 | | | $ | 2,885 | |
| Income taxes paid | $ | 269 | | | $ | 64 | |
Property and equipment purchases included in accounts payable and accrued liabilities | $ | 7 | | | $ | 36 | |
Common stock offering costs included in accounts payable and accrued liabilities | $ | 135 | | | $ | 6 | |
| Right-of-use assets obtained in exchange for lease liabilities | $ | — | | | $ | 458 | |
The accompanying notes are an integral part of these consolidated financial statements.
Eiger BioPharmaceuticals, Inc.
Notes to the Consolidated Financial Statements
1. Description of Business
Eiger BioPharmaceuticals, Inc. (the Company or Eiger) was incorporated in the State of Delaware on November 6, 2008. Eiger is a commercial-stage biopharmaceutical company focused on the development of innovative therapies for rare metabolic diseases.
Eiger’s lead product candidate, avexitide, is a well-characterized, first-in-class glucagon-like peptide-1 (GLP-1) antagonist and is in development for the treatment of post-bariatric hypoglycemia (PBH) and other forms of hyperinsulinemic hypoglycemia (HH) arising after gastrointestinal surgeries. These disorders are characterized by exaggerated secretion of GLP-1 after meals, dysregulated secretion of insulin, followed by a rapid drop in blood sugar. Avexitide is the only drug in development for PBH with Breakthrough Therapy designation by the U.S. Food and Drug Administration (FDA). Avexitide is also in development for congenital hyperinsulinism (HI), an ultra-rare, life-threatening, pediatric disorder of persistent hypoglycemia that results in irreversible brain damage in up to 50% of children with the condition. Avexitide has completed Phase 2 for both PBH and HI, and Phase 3 study start-up activities for PBH and HI were initiated. However further Phase 3 activities have been paused until additional funding is secured.
The FDA approved the Company’s first commercial product, Zokinvy® (lonafarnib), to reduce risk of mortality of Hutchinson-Gilford progeria syndrome (HGPS) and for treatment of processing-deficient progeroid laminopathies (PL), with either heterozygous LMNA mutation with progerin-like protein accumulation, or homozygous or compound heterozygous ZMPSTE24 mutations, on November 20, 2020. Collectively known as progeria, HGPS and PL are ultra-rare, fatal, genetic premature aging diseases that accelerate mortality in young patients. In July 2022, the Company announced that the European Commission (EC) granted marketing authorization (MA) under exceptional circumstances for Zokinvy through the centralized procedure. The EC's MA is valid in all 27 European Union (EU) member states plus Iceland, Liechtenstein, and Norway. In May 2022, the Pharmaceutical Division at the Ministry of Health of Israel granted regulatory approval for Zokinvy in Israel. In August 2022, the Medicine and Healthcare products Regulatory Agency (MHRA) granted approval in the UK.
The Company commercially launched Zokinvy in the U.S. in January 2021 and started to recognize product revenue in the first quarter of 2021. The first European sales were recognized in the fourth quarter of 2022.
In June 2023, the Company announced that it is focusing its clinical development efforts on advancing avexitide in HH indications, including PBH. The Company will continue to commercialize Zokinvy (lonafarnib) for the treatment of HGPS and processing-deficient PL. In addition, Eiger is evaluating strategic partnering options for its virology assets, lonafarnib and peginterferon lambda. In June 2023, the Company also announced that it has appointed David Apelian, MD, PhD, MBA, who has served as interim Chief Executive Officer (CEO) since December 2022, as the Company's next CEO.
The Company’s principal operations are based in Palo Alto, California, with subsidiaries in Delaware, Ireland, England and Wales. The Company operates in one segment.
Reverse Stock Split
In January 2024, following approval by the Company’s stockholders at a special meeting of stockholders held in December 2023, the Company's board of directors approved a certificate of amendment to the Company’s amended and restated certificate of incorporation to effect a reverse split of shares of the Company’s common stock on a 1-for-30 basis, which was effected on January 5, 2024. All share data and per share data amounts for all periods presented in the consolidated financial statements and notes thereto have been retrospectively adjusted to reflect the effect of the Reverse Stock Split.
Liquidity
As previously reported in the Company's Current Report on Form 8-K, filed with the U.S. Securities and Exchange Commission on April 1, 2024, and as discussed in Note 13, the Company and its wholly owned subsidiaries commenced voluntary proceedings under Chapter 11 of the United States Bankruptcy Code. See Note 13 for additional details. The
Company expects to be delisted from Nasdaq on April 11, 2024, which will have a negative impact on the Company's ability to raise capital.
As of December 31, 2023, the Company had $25.4 million of cash and cash equivalents. The Company had an accumulated deficit of $512.2 million and negative cash flows from operating activities as of December 31, 2023. As the Company continues to incur losses, its transition to profitability will depend on the successful development, approval, and commercialization of product candidates and on the achievement of sufficient revenues to support its cost structure. The Company may never achieve profitability, and until it does, the Company will need to continue to raise additional capital. Based on its recurring losses from operations incurred since inception, expectation of continuing operating losses and negative cash flows for the foreseeable future, need to raise additional capital to finance its future operations, and given the current cash, cash equivalents and short-term securities balance, the Company has concluded that there is substantial doubt regarding the Company’s ability to continue as a going concern beyond twelve months after the date that these consolidated financial statements are issued, which was a principal reason for our decision to seek bankruptcy protection under Chapter 11.
The registration statement registering the offer and sale of shares pursuant to the 2022 ATM Facility expired in December 2023. To the extent that the Company raises additional capital through offerings of debt or other securities convertible into equity, stockholder ownership interest will be diluted, and the terms of these new securities may include liquidation or other preferences that adversely affect the rights of holders of our common stock. The Company has not raised any proceeds under the 2022 ATM Facility during 2023. As a result, the Company has concluded that management’s plans do not alleviate substantial doubt about the Company’s ability to continue as a going concern. The consolidated financial statements have been prepared assuming the Company will continue as a going concern, which contemplates the continuity of operations, the realization of assets and the satisfaction of liabilities and commitments in the normal course of business. The Company reclassified its long term debt to current liabilities on its consolidated balance sheet as of December 31, 2023, in connection with its recent filing of Chapter 11, as discussed in Note 7 and Note 13.
2. Summary of Significant Accounting Policies
Basis of Presentation
The consolidated financial statements include the accounts of Eiger BioPharmaceuticals, Inc. and its wholly owned subsidiaries, EBPI Merger Inc., EB Pharma LLC, Eiger BioPharmaceuticals Europe Limited, and EigerBio Europe Limited, have been prepared in accordance with accounting principles generally accepted in the United States of America, (U.S. GAAP) and follow the requirements of the Securities and Exchange Commission (SEC) for annual reporting. All intercompany balances and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the reporting period. On an ongoing basis, the Company evaluates its estimates including, but not limited to, those related to the estimated useful lives of long-lived assets, clinical trial accruals, fair value of assets and liabilities, fair value of investments, and stock-based compensation. The Company bases its estimates on historical experience and on various other market-specific and relevant assumptions that the Company believes to be reasonable under the circumstances. Actual results could differ from those estimates.
Concentrations of Risk
Financial instruments that potentially subject the Company to a concentration of credit risk consist of cash, cash equivalents and investments. The Company’s cash is held by financial institutions in the United States and Ireland. Amounts on deposit may at times exceed federally insured limits. The Company is exposed to credit risk in the event of default by the financial institutions holding its cash, cash equivalents and investments and issuers of investments. The Company manages its credit risk by holding its cash, cash equivalents and investments in large financial institutions within the U.S. and Ireland. In addition, the Company’s investment policy limits investments to certain types of instruments such as money market funds, debt securities issued by the U.S. government and its agencies, corporate debt securities, commercial paper as well as asset-backed securities, and places restrictions on the credit ratings, maturities and concentration by type and issuer. The Company has not experienced any losses on its deposits of cash, cash equivalents and investments. The Company relies on one supply chain for each of its product candidates. If any of the single source
suppliers in any of the supply chains fail to satisfy the Company’s requirements on a timely basis, the Company could suffer delays in its clinical development programs and activities which could adversely affect its operating results.
Three customers accounted for approximately 48 percent, 37 percent and 15 percent of the Company’s accounts receivable as of December 31, 2023. Two customers accounted for approximately 73 percent and 26 percent of product revenue during the year ended December 31, 2023. Two customers accounted for approximately 58 percent and 42 percent of the Company’s accounts receivable as of December 31, 2022. One customer accounted for approximately 93 percent of product revenue during the year ended December 31, 2022.
Foreign Currency Exchange
Foreign Currency Transaction Risk
The foreign currency transaction risk relates to changes in exchange rates on monetary assets, liabilities, revenues and expenses held at Eiger BioPharmaceuticals Europe Limited. Gains and losses on foreign currency transactions result primarily from monetary assets, liabilities, revenues and expenses denominated in Euro. Aggregated transaction gains for were $0.1 million for the years ended December 31, 2023 and 2022. The Company expects the foreign currency gain/loss to continue to fluctuate as long as the Company continues to hold monetary assets and liabilities at its subsidiaries in Ireland, England and Wales. Market uncertainty could potentially lead to significant volatility with foreign currency exchange rates, which could result in additional foreign currency gain/loss.
Foreign Currency Translation Risk
The foreign currency translation risk relates to the translation of the foreign consolidated subsidiaries' assets, liabilities, revenues and expenses from the subsidiaries’ functional currency to the U.S. dollar at each reporting date. Fluctuations in exchange rates may impact the amount of assets, liabilities, revenues and expenses reported on the consolidated balance sheets and consolidated statements of operations. The financial statements of the Company’s foreign subsidiaries, which have a functional currency other than the U.S. dollar, are translated into U.S. dollars using a current exchange rate. Gains and losses resulting from this translation are recognized as a foreign currency translation adjustment within accumulated other comprehensive loss, which is a component of stockholders' (deficit) equity and comprehensive loss. Aggregate translation losses, net of tax, were $0.1 million for the years ended December 31, 2023 and 2022.
Cash and Cash Equivalents
Cash and cash equivalents include all cash balances and highly liquid investments with original maturities of three months or less from the date of purchase. Cash equivalents consists primarily of amounts invested in money market funds held at financial institutions and corporate debt securities. The recorded carrying amount of cash equivalents approximates their fair value.
Debt Securities
All securities are short-term in nature and consist of debt securities classified as available-for-sale and have maturities greater than 90 days, but less than 365 days from the date of acquisition. The Company’s debt securities consist of available-for-sale securities that are classified as Level 2 because their value is based on valuations using significant inputs derived from, or corroborated by, observable market data. The Company evaluates, on a quarterly basis, its available-for-sale debt securities for potential impairment. For available-for-sale debt securities in an unrealized loss position, the Company assesses whether such declines are due to credit related factors such as changes to the rating of the security by a ratings agency, market conditions and supportable forecasts of economic and market conditions, among others. If the fair value of available-for-sale debt securities is less than the amortized cost basis, the Company assesses whether it has plans to sell the security or it is more likely than not it will be required to sell any available-for-sale debt security before recovery of its amortized cost basis. If either condition is met, the security’s amortized cost basis is written down to fair value and is recognized through other (expense) income, net. If neither condition is met, declines as a result of credit losses, if any, are recognized as an allowance for credit loss, limited to the amount of unrealized loss, through other (expense) income, net. Any portion of the unrealized loss that is not a result of a credit loss, is recognized in other comprehensive loss. The cost of available-for-sale securities sold is based on the specific-identification method. Realized gains and losses on the sale of debt securities are determined using the specific-identification method and recorded in other (expense) income, net.
Property and Equipment
Property and equipment are stated at cost, less accumulated depreciation. Depreciation expense is computed using the straight-line method over the estimated useful lives of the assets. Depreciation begins at the time the asset is placed into service. Maintenance and repairs are charged to operations as incurred. Property and equipment purchased for specific research and development projects with no alternative uses are expensed as incurred.
The useful lives of the property and equipment are as follows:
| | | | | |
| Lab equipment | 5 years |
| Furniture | 5 years |
| Leasehold improvements | Shorter of remaining lease term or 5 years |
| Computer equipment and software | 3 years |
Impairment of Long-Lived Assets
The Company evaluates its long-lived assets, including property and equipment, for impairment whenever events or changes in circumstances indicate that the carrying value of these assets may not be recoverable. The Company assesses the recoverability of long-lived assets by determining whether or not the carrying value of such assets will be recovered through undiscounted expected future cash flows. If the asset is considered to be impaired, the amount of any impairment is measured as the difference between the carrying value and the fair value of the impaired asset. Through December 31, 2023, the Company has not impaired any long-lived assets.
Accounts Receivable
Accounts receivable represent amounts billed to the Company’s customers, net of an allowance for credit losses. Trade accounts receivable are recorded at invoiced amounts and do not bear interest. The allowance for credit losses reflects the Company’s best estimate of probable losses inherent in the receivable portfolio determined based on various factors, including age of the outstanding invoice, credit quality of the customer, historical experience, current economic conditions, and management’s expectations of future economic conditions. The Company regularly reviews the adequacy of the allowance for credit losses by considering the age of each outstanding invoice and the collection history of each customer to determine the appropriate amount of allowance for credit losses.
The Company had no allowance as of December 31, 2023 and 2022. The Company had no credit losses for the years ended December 31, 2023 and 2022.
Inventories
Inventories are stated at the lower of cost, determined based on actual costs, or estimated net realizable value, on a first-in, first-out basis. Inventories consist of raw materials, work-in-process, and finished goods.
Prior to regulatory approval of the Company’s product candidates, expenses incurred to manufacture drug products are recorded as research and development expense. The Company begins capitalizing these expenses as inventory upon regulatory approval.
The Company periodically assesses the recoverability of its inventory and reduces the carrying value of the inventory when items are determined to be obsolete, defective or in excess of forecasted sales requirements. Inventory write-downs for excess, defective, and obsolete inventory are recorded as a cost of sales.
The Company wrote-down $8,000 and $1.0 million of inventories for the years ended December 31, 2023 and 2022, respectively.
Revenue Recognition
The Company recognizes revenue upon transfer of control of promised products to customers in an amount that reflects the consideration it expects to receive in exchange for those products. To determine revenue recognition for contracts with customers, the Company performs the following five-step approach: (i) identify the contract, or contracts, with a customer;
(ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when, or as, the performance obligation is satisfied. The five-step model is only applied to contracts when it is probable that the Company will collect substantially all of the consideration it is entitled to in exchange for the goods transferred to a customer.
Product Revenue
The Company’s product revenue consists of sales of Zokinvy, which received FDA approval in November 2020 and was launched commercially in the United States in January 2021 and in Europe in November 2022. Prior to 2021, the Company had no product revenue. In the United States, the Company sells Zokinvy to a single specialty pharmacy provider that subsequently dispenses the product directly to patients. The Company discloses revenue on a total basis without further disaggregation. Additionally, the Company does not have any contract assets or liabilities, other than accounts receivable, related to its product revenue.
In June 2021, the French National Agency for Medicines and Health Products Safety (ANSM) granted Zokinvy (lonafarnib) a Temporary Authorizations for Use (Autorisation Temporaire d'Utilisation or ATU) for an early access program for a term of one year. The Company has received a one year extension of the ATU program and expects the program to continue until commercial reimbursement of Zokinvy is approved in France. In the context of this program, the Company sells product to a distributor who in turn ships product to pharmacies after receiving requests from physicians for patients in France. In November 2021, the Company began distributing and recognizing revenue from sales of Zokinvy (lonafarnib) through a reimbursed early access program in France. The Company recorded revenue of $2.4 million and $0.2 million from sales of product under the ATU program for the years ended December 31, 2023 and 2022, respectively.
The Company recognizes product revenue when a customer obtains control of its product, which occurs at a point in time, typically upon delivery to a customer as the delivery of the product is the Company’s only performance obligation. Shipping and handling activities are fulfillment activities rather than a separate performance obligation and are recorded in cost of sales.
Product revenue is recorded at the net sales price (transaction price), which includes estimates of variable consideration resulting from rebates, prompt payment discounts, co-payment assistance, and returns. Amounts related to such items are estimated at contract inception and updated at the end of each reporting period as additional information becomes available. The amount of variable consideration may be constrained and is included in the transaction price only to the extent it is probable that a significant reversal of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is resolved. Product revenue is recorded after considering the impact of the following variable consideration amounts along with the constraint at the time of revenue recognition:
Rebates: The Company’s product is subject to government mandated rebates for Medicaid Drug Rebate Program, Medicare Part D Prescription Drug Benefit Program, and other government health care programs in the United States. Rebate amounts are based upon contractual agreements or legal requirements with public sector benefit providers. The Company uses the expected-value method for estimating these rebates based on statutory discount rates and expected utilization. The expected utilization of rebates is estimated based on expected coverage of identified patients. Estimates for these rebates are adjusted quarterly to reflect the most recent information. The Company records an accrued liability for unpaid rebates related to products for which control has been transferred to a customer.
Prompt payment discounts: The Company provides a discount to a customer if it pays for purchases within 30 days. The Company expects that its customers will earn prompt payment discounts and uses the most likely amount method for estimating such discounts. As a result, when revenues are recognized, the Company deducts the full amount of the prompt payment discounts from total product revenues and records these discounts as a reduction of accounts receivable.
Co-payment assistance: The Company provides co-payment assistance to patients who have commercial insurance and meet certain eligibility requirements. The Company uses the expected-value method for estimating co-payment assistance based on estimates of program redemption using data provided by third-party administrators. Estimates for the co-payment assistance are adjusted quarterly to reflect actual experience. The Company records an accrued liability for unredeemed co-payment assistance related to products for which control has been transferred to a customer.
Product returns: The Company offers limited product return rights and generally allows for the return of product that is damaged or defective, or within a few months prior to and up to a few months after the product expiration date. The Company considers several factors in the estimation of potential product returns, including expiration dates of the
product shipped, the limited product return rights, third-party data in monitoring channel inventory levels, shelf life of the product, and other relevant factors.
Other Revenue
Other revenue consists of milestone payments from the Marketing and Distribution Agreement (MDA) with AnGes, Inc., which was executed in May 2022. The MDA provides AnGes with a right to use the Company's intellectual property (IP) and seek regulatory approval for and commercialization of Zokinvy in Japan. The Company will receive additional payments upon achievement of certain regulatory milestones.
Cost of Sales
Cost of sales consists primarily of direct and indirect costs related to the manufacturing of Zokinvy for commercial sale, including third-party manufacturing costs, packaging services, freight, storage costs, and write down of inventories.
Accrued Research and Development Costs
The Company accrues for estimated costs of research and development activities conducted by third-party service providers, which include the conduct of preclinical and clinical studies, and contract manufacturing activities. The Company records the estimated costs of research and development activities based upon the estimated amount of services provided but not yet invoiced and includes these costs in accrued liabilities in the accompanying consolidated balance sheets and within research and development expenses in the accompanying consolidated statements of operations. The Company accrues for these costs based on factors such as estimates of the work completed made by its internal personnel and third-party service providers and in accordance with agreements established with its third-party service providers. The Company makes judgments and estimates in determining the accrued liabilities balance in each reporting period and although the Company does not expect its estimate to be materially different from amounts actually incurred, as actual costs become known the Company adjusts its accrued liabilities.
Leases
The Company determines if an arrangement is or contains a lease at inception. Material leases with a term greater than one year are recognized in right-of-use assets and current and noncurrent lease liabilities, as applicable, in the Company’s consolidated balance sheets.
The Company had a real estate lease for its office space in Palo Alto, California that expired on February 28, 2024. In February 2024, the Company amended the lease to extend the lease term for an additional month from March 1, 2024 to April 1, 2024 with a month to month renew option. The Company determines the initial classification and measurement of its right-of-use assets (ROU assets) and lease liabilities at the lease commencement date and thereafter if modified. The lease term includes any renewal options and termination options that the Company is reasonably assured to exercise. The present value of lease payments is determined by using the interest rate implicit in the lease, if that rate is readily determinable; otherwise, the Company uses its incremental borrowing rate. The incremental borrowing rate is determined by using the rate of interest that the Company would pay to borrow on a collateralized basis an amount equal to the lease payments for a similar term and in a similar economic environment.
Rent expense for operating leases is recognized on a straight-line basis, unless the operating lease ROU assets have been impaired, over the reasonably assured lease term based on the total lease payments and is included in operating expenses in the consolidated statements of operations. For operating leases that reflect impairment, the Company will recognize the amortization of the operating lease ROU assets on a straight-line basis over the remaining lease term with rent expense still included in selling, general and administrative expenses in the consolidated statements of operations.
The Company has elected the practical expedient to not separate lease and non-lease components. The Company’s non-lease components are primarily related to property maintenance and insurance, which varies based on future outcomes, and thus is recognized in selling, general and administrative expenses when incurred.
Debt Issuance Costs
Financing costs incurred with securing a term debt are recorded in the Company’s consolidated balance sheets as an offset to the term debt and amortized to interest expense in the Company’s consolidated statements of operations over the contractual life of the loan using the effective interest method.
Research and Development Costs
Research and development costs are expensed as incurred and consist of payroll expenses, stock-based compensation expense, lab supplies and allocated facility costs, as well as fees paid to third parties that conduct certain research and development and manufacturing activities on the Company’s behalf. Amounts incurred in connection with license and asset purchase agreements are also included in research and development expenses. Manufacturing costs related to products undergoing regulatory approval are expensed as research and development costs until receipt of such approval. Non-refundable advance payments for research and development activities to be received or incurred in the future are deferred and recorded in prepaid expenses and other current assets. The prepaid amounts are expensed as the related services are performed or goods are delivered.
Stock-Based Compensation
Stock-based awards to employees and directors, including stock options, are recorded at fair value as of the grant date using the Black-Scholes option pricing model and recognized as expense on a straight line-basis over the employee’s or director’s requisite service period (generally the vesting period). Stock-based awards to non-employees are recorded at their fair value as of the grant date, using the Black-Scholes option pricing model and recognized as expense over the period in which the related services are received. The determination of fair value for stock-based awards on the date of grant using an option pricing model requires management to make certain assumptions for Black-Scholes option pricing model inputs. The Company accounts for forfeitures of stock-based awards when they occur.
Income Taxes
The Company uses the asset and liability method of accounting for income taxes. Under this method, deferred tax assets and liabilities are determined based on the differences between the financial reporting and the tax bases of assets and liabilities and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. The Company must then assess the likelihood that the resulting deferred tax assets will be realized. A valuation allowance is provided when it is more likely than not that some portion or all of a deferred tax asset will not be realized. Due to the Company’s lack of earnings history, the net deferred tax assets have been fully offset by a valuation allowance.
The Company recognizes benefits of uncertain tax positions if it is more likely than not that such positions will be sustained upon examination based solely on their technical merits, as the largest amount of benefit that is more likely than not to be realized upon the ultimate settlement. The Company’s policy is to recognize interest and penalties related to the underpayment of income taxes as a component of income tax expense or benefit. To date, there have been no interest or penalties charged in relation to unrecognized tax benefits.
Internal Revenue Code Section 382 limits the use of net operating loss and tax credit carryforwards in certain situations where changes occur in the stock ownership of a company. In the event that the Company experiences a change of ownership, utilization of the net operating loss and tax credit carryforwards may be restricted.
Comprehensive Loss
Comprehensive loss represents all changes in stockholders' (deficit) equity except those resulting from and distributions to stockholders. The Company’s unrealized gains and losses on debt securities and foreign currency translation adjustments represent the components of other comprehensive loss that are excluded from the reported net loss and that are presented in the consolidated statements of comprehensive loss.
Net Loss per Share
Basic net loss per share of common stock is calculated by dividing the net loss by the weighted average number of shares of common stock outstanding during the period, without consideration for potentially dilutive securities. Since the Company was in a loss position for all periods presented, diluted net loss per share is the same as basic net loss per share for all periods as the inclusion of all potential common shares outstanding would have been anti-dilutive.
The following table sets forth the outstanding potentially dilutive securities which have been excluded in the calculation of diluted net loss per share because including such securities would be anti-dilutive (in common stock equivalent shares):
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Options to purchase common stock | 180,151 | | | 204,656 | |
| ESPP | 12,839 | | | 11,121 | |
| Restricted stock units (unvested) | 5,998 | | | 21,337 | |
| Total | 198,988 | | | 237,114 | |
Recent Adopted Accounting Pronouncements
In June 2016, the Financial Accounting Standards Board (FASB) issued Accounting Standards Updates (ASU) No. 2016-13, Financial Instruments—Credit Losses (Topic 326). ASU 2016-13 requires an entity to utilize a new impairment model that requires measurement and recognition of expected credit losses for most financial assets and certain other instruments, including but not limited to available-for-sale debt securities. Credit losses relating to available-for-sale debt securities will be recorded through an allowance for credit losses rather than as a direct write-down to the security. The new guidance requires the use of forward-looking expected credit loss models based on historical experience, current conditions, and reasonable and supportable forecasts that affect the collectability of the reported amount, which may result in earlier recognition of credit losses under the new guidance. In April 2019, the FASB issued ASU No. 2019-04, Codification Improvements to Topic 326, Financial Instruments – Credit Losses, Topic 815, Derivatives and Hedging, and Topic 825, Financial Instruments, which clarifies and corrects certain unintended applications of the guidance contained in each of the amended Topics. Additionally, in May 2019, the FASB issued ASU No. 2019-05, Financial Instruments – Credit Losses (Topic 326), which provides an option to irrevocably elect to measure certain individual financial assets at fair value instead of amortized cost. In November 2019, the FASB issued ASU No. 2019-10, Financial Instruments – Credit Losses (Topic 326), Derivatives and Hedging (Topic 815), and Leases (Topic 842), which deferred the effective date for ASU No. 2016-13 for smaller reporting companies to fiscal years beginning after December 15, 2022, including interim periods within those fiscal years. The Company adopted this guidance on a modified-retrospective basis effective January 1, 2023 and noted no material impact to the Company’s consolidated financial statements.
Recently Issued Accounting Pronouncements Not Yet Adopted
In November 2023, the FASB issued ASU No. 2023-07, Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures, which requires an enhanced disclosure of significant segment expenses on an annual and interim basis. This guidance will be effective for the annual periods beginning after December 15, 2023, and interim periods within fiscal years beginning after December 15, 2024. Early adoption is permitted. Upon adoption, the guidance should be applied retrospectively to all prior periods presented in the financial statements. The Company does not expect the adoption of this guidance to have a material impact on the consolidated financial statements.
In December 2023, the FASB issued ASU No. 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures, which improves the transparency of income tax disclosures by requiring consistent categories and greater disaggregation of information in the effective tax rate reconciliation and income taxes paid disaggregated by jurisdiction. It also includes certain other amendments to improve the effectiveness of income tax disclosures. This guidance will be effective for the annual periods beginning after December 15, 2024. Early adoption is permitted. Upon adoption, the guidance can be applied prospectively or retrospectively. The Company does not expect the adoption of this guidance to have a material impact on the consolidated financial statements.
3. Fair Value Measurements
Fair value accounting is applied for all financial assets and liabilities that are recognized or disclosed at fair value in the financial statements on a recurring basis (at least annually). As of December 31, 2023 and 2022, the carrying amount of cash equivalents, accounts receivable, prepaid expenses and other current assets, accounts payable and accrued liabilities approximated their estimated fair value due to their relatively short maturities. Management believes the terms of its long-term debt reflect current market conditions for an instrument with similar terms and maturity, therefore the carrying value of the Company’s debt approximated its fair value.
Assets and liabilities recorded at fair value on a recurring basis in the consolidated balance sheets are categorized based upon the level of judgment associated with the inputs used to measure their fair values. Fair value is defined as the exchange price that would be received for an asset or an exit price that would be paid to transfer a liability in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. The authoritative guidance on fair value measurements establishes a three-tier fair value hierarchy for disclosure of fair value measurements as follows:
Level 1: Inputs are unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date;
Level 2: Inputs are observable, unadjusted quoted prices in active markets for similar assets or liabilities, unadjusted quoted prices for identical or similar assets or liabilities in markets that are not active, or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the related assets or liabilities; and
Level 3: Unobservable inputs that are significant to the measurement of the fair value of the assets or liabilities that are supported by little or no market data.
The Company’s money market funds are classified as Level 1 because they are valued using quoted market prices. The Company’s debt securities and commercial paper consist of available-for-sale securities and are classified as Level 2 because their value is based on valuations using significant inputs derived from or corroborated by observable market data. There were no assets or liabilities classified as Level 3 as of December 31, 2023 and 2022.
There were no transfers into or out of Level 3 of the fair value hierarchy during the periods presented.
The following tables present the fair value hierarchy for assets measured at fair value, and summarize the estimated value of the Company’s cash equivalents and debt securities and the gross unrealized holding gains and losses (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2023 |
| Level | | Amortized cost | | Unrealized gain | | Unrealized loss | | Estimated Fair
Value |
| Cash equivalents: | | | | | | | | | |
| Money market funds | 1 | | $ | 18,653 | | | $ | — | | | $ | — | | | $ | 18,653 | |
| Total cash equivalents | | | $ | 18,653 | | | $ | — | | | $ | — | | | $ | 18,653 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2022 |
| Level | | Amortized cost | | Unrealized gain | | Unrealized loss | | Estimated Fair
Value |
| Cash equivalents: | | | | | | | | | |
| Money market funds | 1 | | $ | 11,546 | | | $ | — | | | $ | — | | | $ | 11,546 | |
| Commercial paper | 2 | | 3,968 | | | — | | | — | | | 3,968 | |
| Total cash equivalents | | | $ | 15,514 | | | $ | — | | | $ | — | | | $ | 15,514 | |
| Short-term debt securities: | | | | | | | | | |
| U.S. government bonds | 2 | | $ | 39,646 | | | $ | 3 | | | $ | (86) | | | $ | 39,563 | |
| Corporate debt securities | 2 | | 28,759 | | | — | | | (117) | | | 28,642 | |
| Commercial paper | 2 | | 4,945 | | | — | | | — | | | 4,945 | |
| Total debt securities | | | $ | 73,350 | | | $ | 3 | | | $ | (203) | | | $ | 73,150 | |
There were no financial liabilities to be remeasured on a recurring basis as of December 31, 2023 and 2022.
During the year ended December 31, 2023, the Company did not recognize any credit losses. The Company determined that the decline in fair value of debt securities was not due to credit-related factors, and no allowance for expected credit losses was recorded as of December 31, 2023. There were no impairment losses on investments during the years ended December 31, 2023 and 2022. The unrealized losses of $0.2 million as of December 31, 2022 include debt securities with unrealized losses of $21,000 that have been in the loss position for more than 12 months.
4. Balance Sheet Components
Inventories
Inventories consist of the following (in thousands):
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Work-in-progress | $ | 940 | | | $ | 884 | |
| Raw materials | 531 | | | 1,703 | |
| Finished goods | 85 | | | 266 | |
| Total inventories | $ | 1,556 | | | $ | 2,853 | |
Property and Equipment, Net
Property and equipment, net consist of the following (in thousands):
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Computer equipment and software | $ | 1,004 | | | $ | 736 | |
| Construction in progress | 305 | | | 367 | |
| Lab equipment | 271 | | | 271 | |
| Furniture | 116 | | | 116 | |
| Leasehold improvements | 101 | | | 101 | |
| Total property and equipment | 1,797 | | | 1,591 | |
| Less: accumulated depreciation | (1,170) | | | (895) | |
| Property and equipment, net | $ | 627 | | | $ | 696 | |
Depreciation expense for the years ended December 31, 2023 and 2022 was approximately $0.3 million.
Prepaid Expenses and Other Current Assets
Prepaid expenses and other current assets consist of the following (in thousands):
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Prepaid research costs | $ | 5,577 | | | $ | 2,822 | |
| Prepaid marketing | 911 | | | 753 | |
| Prepaid contract manufacturing costs | 577 | | | 3,542 | |
| Short term deposits | 404 | | | 4,542 | |
| Prepaid insurance | 403 | | | 586 | |
| Other | 1,289 | | | 1,740 | |
| Total prepaid expenses and other current assets | $ | 9,161 | | | $ | 13,985 | |
Accrued Liabilities
Accrued liabilities consist of the following (in thousands):
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Compensation and related benefits | $ | 3,046 | | | $ | 6,167 | |
| Contract research costs | 2,909 | | | 4,579 | |
| Product revenue reserves | 2,277 | | | 1,373 | |
| Contract manufacturing costs | 818 | | | 2,101 | |
| Legal fees | 438 | | | 562 | |
| Other | 478 | | | 873 | |
| Total accrued liabilities | $ | 9,966 | | | $ | 15,655 | |
5. License, Collaboration, and Product Development Agreements
License Agreement with the Trustees of the University of Pennsylvania and the Children’s Hospital of Philadelphia
In May 2019, the Company entered into a license agreement (the UPenn/CHOP Agreement) with the Trustees of the University of Pennsylvania (UPenn) and the Children’s Hospital of Philadelphia (CHOP), under which the Company obtained an exclusive, royalty-bearing, worldwide license to develop, manufacture and sell certain Glucagon Like Peptide-1 (GLP-1) receptor antagonist(s) products to treat all human and animal conditions. The Company also obtained an exclusive, royalty-bearing, sublicensable, worldwide license to certain data developed by CHOP. The Company is responsible for the development and commercialization of the licensed products at its sole cost and expense.
As part of the consideration for the rights granted to the Company under the UPenn/CHOP Agreement, the Company paid UPenn a one-time, non-refundable issue fee of $1.0 million, which is recorded in research and development expenses for the year ended December 31, 2019. In addition, the Company is obligated to pay UPenn a specified annual license maintenance fee, up to $2.5 million upon marketing authorization in one or more countries, and up to $18.0 million in commercial milestones. The Company will also be required to pay UPenn a flat royalty in the low-single digits on net sales of all licensed products by the Company, subject to specified reductions and offsets, and specified minimum annual royalty payments. The Company’s obligation to pay royalties expires on a product-by-product and country-by-country basis, on the later of: (i) the expiration of the last valid claim in the licensed patents in any country, or (ii) the tenth anniversary of the first commercial sale of the product in such country. No milestones have been achieved as of December 31, 2023.
The Company may terminate the UPenn/CHOP Agreement in its entirety for any reason by providing prior written notice to UPenn and CHOP. UPenn or CHOP may terminate the UPenn/CHOP Agreement, upon a written notice, for the Company’s failure to achieve the specified diligence milestones within the specified periods, subject to the Company’s extension rights.
Product Development Agreement
On August 11, 2018, the Company entered into a Product Development Agreement and a First Project Agreement (the Product Agreements), pursuant to which the Company will receive development program support services for its HDV program. The services are to be provided from July 1, 2018 through the new drug application (NDA) filing. As consideration, the Company has committed to pay fees of approximately $10.0 million in cash and stock over four years, including approximately $0.8 million for expert consultant fees and pass through costs to vendors, plus certain incentive-based regulatory milestone fees up to $1.0 million. As part of the aggregate payment, the Company issued 3,848 shares (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) of common stock subject to quarterly vesting requirements related to successful performance of the services and achievement of budget timeline set forth in the Product Agreements. The Product Agreements can be terminated by either party due to material breach or by the Company without cause with 90 days prior written notice. For the year ended December 31, 2022, the Company recognized research and development expense of $19,000 related to the shares issued under the Product Agreements. As of December 31, 2022, the restricted shares were fully vested. No milestones have been achieved as of December 31, 2023.
Progeria Research Foundation (PRF) Collaboration Agreement
On May 15, 2018, the Company entered into a Collaboration and Supply Agreement (the PRF Collaboration Agreement) with PRF. Under the PRF Collaboration Agreement, the parties agreed to collaborate with respect to the development and pursuit of regulatory approval of lonafarnib for the treatment of HGPS and processing-deficient progeroid laminopathies, collectively called progeria, in humans. PRF granted the Company a non-exclusive, world-wide, royalty-free, sub-licensable license pertaining to all intellectual property and data controlled by PRF to prepare and file any NDA for a product containing lonafarnib for progeria. The Company is obligated to: (i) exclusively supply lonafarnib to PRF for use in clinical trials and non-clinical research in progeria at the Company’s expense, (ii) prepare and be the sponsor of any NDA submission for lonafarnib for the treatment of progeria to the FDA, (iii) use commercially reasonable efforts to file a NDA for progeria by a specified date, (iv) submit a Rare Pediatric Disease designation and a request for expedited approval in connection with a NDA filing, (v) establish a patient support program in progeria, and (vi) use commercially reasonable efforts to develop a pediatric formulation of lonafarnib for use in progeria.
Under the PRF Collaboration Agreement, the Company is solely responsible for any additional studies necessary for obtaining an NDA for progeria and is also responsible for any additional costs for such studies up to $2.0 million. The PRF Collaboration Agreement continues for an initial term of ten years and automatically renews for subsequent renewal terms of two years each unless either party terminates earlier. No milestones have been achieved as of December 31, 2023.
Bristol-Meyers Squibb License Agreement
On April 20, 2016, the Company and Bristol-Myers Squibb Company (BMS) entered into a License Agreement (the BMS License Agreement) and a Common Stock Purchase Agreement (the BMS Purchase Agreement).
Under the BMS License Agreement, BMS granted the Company an exclusive, worldwide, license to research, develop, manufacture, and sell products containing PEG-interferon Lambda-1a (peginterferon lambda or the Licensed Product) for all therapeutic and diagnostic uses in humans and animals. The Company is responsible for the development and commercialization of the Licensed Product at its sole cost and expense. The Company paid BMS $2.0 million and issued 5,252 shares of its common stock (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) at an aggregate fair value of $3.2 million in April 2016. The BMS License Agreement also includes development and regulatory milestone payments totaling $61.0 million and commercial sales milestones of up to $128.0 million. The Company is obligated to pay BMS annual net sales royalties in the range of mid-single to mid-teens, depending on net sales levels. In fourth quarter of 2020, the Company recorded a $3.0 million milestone in research and development expense, triggered on successful demonstration of proof of concept, as defined by the BMS License Agreement, in a Phase 2 clinical trial. In March 2022, the Company recorded a $5.0 million milestone expense in research and development, which was related to the initiation of a Phase 3 clinical trial, as defined under the BMS License Agreement. No milestones have been achieved in 2023.
Merck License Agreement
In September 2010, the Company entered into an exclusive license agreement with Schering Corporation, subsequently acquired by Merck & Co., Inc. (Merck), which provides the Company with the exclusive right to develop, manufacture, and sell products containing the compounds lonafarnib for the treatment of all human viruses except certain specified viruses such as hepatitis B and hepatitis C alone. As part of consideration, the Company issued 911 shares of common stock (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) with a fair value of $0.5 million. The Company is obligated to pay Merck up to an aggregate of $27.0 million in development milestones and will be required to pay tiered royalties based on aggregate annual net sales of all licensed products ranging from mid-single to low double-digit royalties on net sales. In May 2015, the first regulatory milestone was achieved and the Company paid the related milestone payment of $1.0 million to Merck. In September 2023, the Phase 3 study was completed and the $1.0 million related milestone expense was recorded in accrued liabilities on the consolidated balance sheet.
On May 15, 2018, the Company entered into an amendment to the exclusive license agreement with Merck, which provides for expansion of the existing exclusively licensed field of use under the license agreement with Merck to include all uses of lonafarnib related to the treatment of Hutchinson-Gilford progeria syndrome (HGPS) in humans at no cost to the Company. On November 3, 2020, the Company entered into an amendment to the exclusive license agreement with Merck which expanded the indication to also include progeroid laminopathies, collectively called progeria. The Company has the sole responsibility and the continuing obligation for the manufacture and supply of lonafarnib to the PRF. Merck will not receive milestone payments in relation to lonafarnib for the treatment of progeria or any royalty payments for sales of a specified quantity of lonafarnib to treat the currently estimated progeria patient population worldwide.
6. Asset Purchase Agreements and Related License Agreements
Avexitide Purchase Agreement and Related Stanford License Agreement
In September 2015, the Company entered into an asset purchase agreement with two individuals, Dr. Tracey McLaughlin and Dr. Colleen Craig, (the Sellers), whereby the Company purchased all of the assets related to the compound avexitide (formerly known as exendin 9-39) including any related intellectual property from the Sellers (the Exendin APA). The Company also entered into a consulting agreement with the Sellers as part of the agreement. The Company issued 512 shares of common stock (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) that were valued at $0.2 million. In addition, the Company granted the Sellers options to purchase an aggregate 3,155 shares of common stock (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) at exercise prices of $61.80 and $517.80 that vest based on certain service and milestone-based performance vesting conditions. As of December 31, 2023, the service vesting conditions were met and the performance conditions were not deemed probable of achievement. No expense was recognized for the milestone-based options during the years ended December 31, 2023 and 2022.
The Company is also obligated to pay development milestone payments in an aggregate amount of up to $1.0 million to each Seller. Additionally, the Company is obligated to pay each Seller royalties of low single-digits based on aggregate annual net sales of all products developed based on avexitide, subject to certain reductions and exceptions. The Company’s obligation to pay royalties expires on the expiration of the last to expire valid claim as defined in the agreement. Additionally, the Company has assumed the license agreement the Sellers had previously entered into with the Board of Trustees of the Leland Stanford Junior University (Stanford). The Company is obligated to pay a royalty to Stanford in the low single-digits on annual net sales after the first commercial sale of any products developed based on avexitide. As of December 31, 2023, the Company has paid a total of $0.1 million in milestone payments to each of the Sellers related to the successful completion of the Phase 2 trials.
7. Debt
Innovatus Term Loan
On June 1, 2022 (Closing Date), the Company entered into a term loan and security agreement (Innovatus Loan) with Innovatus Life Sciences Lending Fund I, LP (Innovatus), providing for up to $75.0 million funded in three tranches with a maturity date of August 31, 2027. The floating per annum interest rate of the Innovatus Loan is equal to the sum of (a) the greater of (i) the Prime Rate published in the Money Rates section of the Wall Street Journal (or any successor thereto) and (ii) 3.5%, plus (b) 3.75%; provided that, at the election of the Borrower, up to 2.25% of such rate shall be payable in-kind until the third anniversary of the closing date. The Company is required to make monthly interest-only payments through July 1, 2027, after which the Company is required to make monthly amortizing payments, with the remaining balance of the principal plus accrued and unpaid interest due at maturity. 2.25% of the interest is payable in-kind for the first three years of the term by increasing the principal balance. Prepayments of the loan, in whole or in part, will be subject to an early prepayment fee which ranges between 3% and 0% of the principal amount repaid and declines each year until the third anniversary date of the Closing Date, after which no prepayment fee is required. The Company is also required to pay an exit fee upon any payment or prepayment equal to 6.5% of the aggregate principal amount of the tranches funded under the Innovatus Loan. The Innovatus Loan contains customary representations, warranties, events of default, including failure to pay amounts due, breaches of covenants and warranties, material adverse change events, certain cross defaults and judgements, and insolvency, and covenants of the Company and its subsidiaries, including a requirement to maintain a cash balance of not less than 5% of the aggregate principal amount of funded and outstanding loan terms at all times. Should the Company be unable to comply with these covenants or if the Company defaults on any portion of its outstanding borrowings, the lender can also impose a 5% interest rate penalty, restrict access to additional borrowings under the loan and security agreement, and accelerate the maturity of the debt to be immediately due and payable. The Innovatus Loan is secured by perfected first priority liens on the Company's assets, including a commitment by the Company to not allow any liens to be placed upon the Company's intellectual property.
The Company was funded $40.0 million in June 2022 on the Closing Date under Tranche A. The remaining $35.0 million is divided into two tranches (Tranche B and Tranche C). The $17.5 million under each of Tranche B and Tranche C was available for a period commencing on the later of (a) the first date that the Company achieves certain development and regulatory milestones applicable to each Tranche and (b) November 1, 2022. Both Tranche B and Tranche C draw periods
end on the earlier of (a) June 30, 2024 or (b) an event of default. Tranches B and C are no longer available to the Company following the filing of the Bankruptcy Petitions.
In connection with the issuance of the Innovatus Loan, the Company recorded a debt discount of $0.2 million and capitalized debt issuance costs of $1.1 million. The discount and issuance costs will be amortized over the life of the loan. Interest expense for the Innovatus Loan for the year ended December 31, 2023 was $5.5 million, and is inclusive of non-cash amortization of the debt discount and debt issuance costs and accretion of final payment. The carrying amount of the Innovatus Loan approximates fair value given the interest rate is based on the prime rate. The effective interest rate for the Innovatus Loan was 13.84% as of December 31, 2023.
Additionally, in connection with entering into the Innovatus Loan, the Company entered into a Stock Purchase Agreement with Innovatus for the sale of common stock with an aggregate value of $5.0 million. On June 1, 2022, the Company issued 24,967 shares of common stock (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) to Innovatus at a per share purchase price of $200.25, the preceding five-day volume weighted average price per share.
A portion of the loan proceeds was used to repay in full the approximately $33.5 million of aggregate principal amount, unpaid interest, and exit fees in connection with loans outstanding owed to Oxford Finance LLC by the Company.
The Company reclassified its long term debt to current liabilities on its consolidated balance sheet as of December 31, 2023, as there is substantial doubt regarding the Company's ability to continue as a going concern and in connection with its recent filing under Chapter 11, as discussed in Note 13. The Innovatus Loan provides that upon the filing of the Bankruptcy Petitions, the principal amount together with accrued and unpaid fees and interest thereon shall become immediately due and payable. However, any efforts to enforce such payment obligations under the Innovatus Loan are automatically stayed as a result of the Bankruptcy Petitions, and the creditors’ rights of enforcement in respect of the Innovatus Loan are subject to the applicable provisions of the Bankruptcy Code.
Oxford Term Loan
On June 1, 2022, upon entering into the Innovatus Loan, the Company repaid the loan from Oxford Finance LLC (Oxford Loan) including (i) the $30.0 million outstanding principal balances, (ii) $0.2 million in accrued and unpaid interest, and (iii) other final payments consisting of $3.3 million, for a total payment of $33.5 million. The Company recorded a loss of $1.1 million on early extinguishment of the debt related to the unamortized debt premium, discount, and cost of issuance, which was recognized as a component of other (expense) income, net in the consolidated statement of operations.
The Company is also required to pay a 5.0% success fee of the total amount drawn under the Amended Oxford Loan within 30 days following the FDA’s approval of the Company’s first product, excluding Zokinvy for treatment of Progeria processing-deficient progeroid laminopathies. This fee is enforceable through March 2029. The Company determined that the success fee met the scope exemption from derivative accounting and should be accounted for under the guidance for contingencies. Accordingly, the Company will record a liability for the success fee upon receipt of approval from the FDA. No success fee liability was recognized as of December 31, 2023 and 2022.
The Company accounts for the amortization of the debt discount utilizing the effective interest method. Debt and unamortized discount balances are as follows (in thousands):
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Face value of debt | $ | 41,452 | | | $ | 40,531 | |
| Exit fee | 2,600 | | | 2,600 | |
| Unamortized debt discount associated with exit fee, debt issuance costs and loan origination fees | (2,933) | | | (3,506) | |
| Current portion of long-term debt | $ | 41,119 | | | $ | 39,625 | |
As of December 31, 2023, future minimum payments of principal, paid in kind interest and exit fee under the Innovatus Loan were as follows (in thousands):
| | | | | |
| Year ending December 31, | |
| 2024 | 45,393 | |
| 2025 | — | |
| 2026 | — | |
2027 | — | |
| Total future payments | 45,393 | |
Less: paid in kind interest | (1,341) | |
Less: unamortized debt discount associated with exit fee, debt issuance costs and loan origination fees | (2,933) | |
Current portion of long-term debt | $ | 41,119 | |
8. Stockholders’ Equity
Common Stock
The holders of the Company’s common stock have one vote for each share of common stock. Common stockholders are entitled to dividends when, as, and if declared by the Board of Directors, subject to the prior rights of the convertible preferred stockholders. As of December 31, 2023, no dividends had been declared by the Board of Directors.
In December 2020, the Company filed a shelf registration statement on Form S-3 (File No. 333-251497) with the Securities and Exchange Commission, which permits the offering, issuance and sale by the Company up to a maximum aggregate offering price of $200.0 million of its common stock, preferred stock, debt securities and warrants. Up to a maximum of $50.0 million of the maximum aggregate offering price of $200.0 million may be issued and sold pursuant to an ATM financing facility (the 2020 ATM Facility) under a sales agreement with Jefferies. In January and March 2022, the Company completed ATM offerings for a total of 194,723 shares of common stock (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) under the 2020 ATM Facility. The offerings were made under the Company’s effective shelf registration statement and resulted in net proceeds to the Company of $45.6 million, after deducting commissions. As of December 31, 2022, the Company completed the sale of all shares available for sale under the 2020 ATM Facility, and the 2020 ATM Facility was terminated.
On March 25, 2022, the Company entered into a new Open Market Sale AgreementSM with Jefferies, pursuant to which the Company can sell up to a maximum of $50.0 million of our common stock in offerings that are deemed “at the market” offerings as defined in Rule 415 under the Securities Act, under the Company’s effective shelf registration statement (the 2022 ATM Facility). In April 2022, the Company completed offerings from the 2022 ATM facility for a total of 89,539 shares of its common stock (adjusted to reflect the 1-for-30 reverse stock split effected on January 5, 2024) resulting in net proceeds of $20.8 million, after deducting commissions costs. No additional offerings were completed since April 2022. The registration statement registering the offer and sale of shares pursuant to the 2022 ATM Facility expired in December 2023.
The Company had reserved shares of common stock for issuance as follows:
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Options issued and outstanding | 180,151 | | | 204,656 | |
| Awards available for future issuance | 203,891 | | | 65,882 | |
| Shares available for issuance under ESPP | 24,587 | | | 26,296 | |
| Restricted and performance stock units outstanding | 5,998 | | | 21,337 | |
| Total | 414,627 | | | 318,171 | |
9. Stock-Based Compensation
Restated 2013 Equity Incentive Plan
In June 2016, the Company’s Board of Directors adopted and in August 2016 the Company’s stockholders approved the Amended and Restated 2013 Equity Incentive Plan (the Restated 2013 Plan). Under the terms of the Restated 2013 Plan, the Company may grant stock options, stock appreciation rights, restricted stock, restricted stock units and other awards to individuals who are then employees, officers, non-employee directors or consultants of the Company. Under the terms of the Restated 2013 Plan, options may be granted at an exercise price not less than fair market value. For employees holding more than 10% of the voting rights of all classes of stock, the exercise prices for incentive and non-statutory stock options may not be less than 110% of fair market value, as determined by the Board of Directors. The terms of options granted under the Restated 2013 Plan may not exceed ten years. The vesting schedule of newly issued option grants is generally four years. As of December 31, 2023, the Company is authorized to issue up to 344,380 shares under the Restated 2013 Plan.
2021 Inducement Plan
During the second quarter of 2021, the Company approved the 2021 Inducement Plan to reserve 28,333 shares of its common stock to be used exclusively for grants of awards to individuals that were not previously employees or directors of the Company as a material inducement to such individuals’ entry into employment with the Company within the meaning of Rule 5635(c)(4) of the Nasdaq Listing Rules. As of December 31, 2023, there were 22,539 shares remaining and available to be issued under the 2021 Inducement Plan.
The following table summarizes stock option activity under the Company’s stock-based compensation plans during the year ended December 31, 2023 (in thousands, except option and share data):
| | | | | | | | | | | | | | | | | | | | | | | |
| Number of
Options | | Weighted-
Average
Exercise
Price | | Weighted-
Average
Remaining
Contractual
Life
(in Years) | | Aggregate
Intrinsic
Value |
| Outstanding as of December 31, 2022 | 204,656 | | | $ | 270.00 | | | 6.00 | | $ | — | |
| Granted | 119,623 | | | $ | 46.89 | | | | | |
| Forfeited | (144,128) | | | $ | 221.28 | | | | | |
| Outstanding as of December 31, 2023 | 180,151 | | | $ | 167.68 | | | 6.56 | | $ | — | |
| Vested and exercisable as of December 31, 2023 | 97,601 | | | $ | 248.27 | | | 5.36 | | $ | — | |
As of December 31, 2023, there were 203,891 shares remaining and available to be issued under the plans, including stock options, restricted stock units (RSUs) and performance stock units (PSUs).
The aggregate intrinsic values of options outstanding, exercisable, vested and expected to vest were calculated as the difference between the exercise price of the options and the closing price of the Company’s common stock of $6.75 as of December 31, 2023.
The aggregate intrinsic value of stock options exercised in 2023 and 2022 was $0 and $0.1 million, respectively.
During the years ended December 31, 2023 and 2022, the weighted-average grant date fair value of options granted were $28.97 and $109.53 per share, respectively. The total grant date fair value of options that vested during the years ended December 31, 2023 and 2022 was $5.8 million and $7.3 million, respectively.
The Company records stock-based compensation of stock options granted by estimating the fair value of stock-based awards using the Black-Scholes option pricing model and amortizes the fair value of the stock-based awards granted over the applicable vesting period of the awards on a straight-line basis. The fair value of stock options was estimated using the following weighted-average assumptions:
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Expected term (in years) | 0.86-6.08 | | 5.27-6.08 |
| Volatility | 90.21%-156.25% | | 68.71%-88.51% |
| Risk free interest rate | 3.50%-4.86% | | 1.76%-4.23% |
| Dividend yield | — | | — |
Expected Term: The expected term represents the number of years the Company estimates, based primarily on historical experience, that the options will be outstanding prior to exercise.
Expected Volatility: The expected volatility for stock options is based on the Company's historical stock price volatility.
Risk-Free Interest Rate: The risk-free interest rate is based on the U.S. Treasury zero coupon issues in effect at the time of grant for periods corresponding with the expected term of option.
Expected Dividend: The Company has never paid dividends on its common stock and has no plans to pay dividends on its common stock. Therefore, the Company used an expected dividend yield of zero.
2013 Employee Stock Purchase Plan
In 2013, the Company’s board of directors adopted and in 2016, upon the merger with Celladon, the Company amended and approved the 2013 Employee Stock Purchase Plan (2013 ESPP). The 2013 ESPP is intended to qualify as an employee stock purchase plan under Section 423 of the U.S. Internal Revenue Code and is administered by the Company’s board of directors.
The number of shares of common stock initially reserved for issuance under the 2013 ESPP was 3,333 shares with an automatic annual increase to the shares issuable under the 2013 ESPP to the lower of (i) 1% of the total number of shares of common stock outstanding on December 31 of the preceding calendar year, and (ii) 5,500 shares of common stock, unless a lower number determined by the board of directors. As of December 31, 2023, a total of 31,626 shares are reserved for issuance and 24,587 shares available for future issuance under the 2013 ESPP. During the years ended December 31, 2023 and 2022, employees purchased 1,709 shares for $0.1 million and 1,594 shares for $0.2 million, respectively, under the 2013 ESPP.
Restricted Stock Units and Performance Stock Units
In the first quarter of 2020, the Company revised its non-employee director compensation policy to approve the granting of RSUs and PSUs in accordance with the Restated 2013 Equity Incentive Plan (the Restated 2013 Plan). Each eligible director who has served for at least six months during the prior calendar year and continues to serve as a non-employee member of the board of Directors (the Board) is granted RSUs. Each eligible director who has served on the Board for less than six months during the prior calendar year and who continues to serve as a non-employee member of the Board, is granted RSUs which are pro-rated for the period served during the prior calendar year.
The RSUs granted to non-employee directors will vest on the one-year anniversary of the grant date, subject to the eligible director’s continuous services through the vesting date, and will vest in full upon a change in control, as defined under the Restated 2013 Plan. The RSUs granted to employees will vest annually on the one-year, two-year, and three-year anniversaries of the grant date. The fair value of all RSUs is measured at the grant date based on the closing market price of the Company’s common stock and is recognized as stock-based compensation expense on a straight-line basis over the vesting period.
Each PSU, which is a stock award, is earned through the achievement of performance-based metrics over a defined performance period. The length of the defined performance period, the performance-based metric to be achieved during the defined performance period, and the measure of whether and to what degree such performance-based metric has been
achieved will be conclusively determined by the compensation committee of the Company’s Board, in its sole discretion. The estimated fair value of equity awards that contain performance conditions is expensed over the term of the award once the Company has determined that it is probable that performance conditions will be satisfied.
During the years ended December 31, 2023 and 2022 the Company granted 500 and 1,000 PSUs, with a weighted-average grant date fair value of $67.80 and $206.10 per share, respectively. The performance-based metrics include the achievement of certain revenue targets and clinical and regulatory milestones. During the years ended December 31, 2023 and 2022, the Company recorded $0.4 million and $0.5 million, respectively, of stock-based compensation expense related to the PSUs as it is expected at the beginning of the period that all milestones would be achieved by the target dates. This expense is included in selling, general and administrative expenses. On June 30, 2023, the Company reassessed the probability of the performance-based metrics being achieved. The Company concluded that some of the performance-based metrics are no longer probable of being achieved but are still more than improbable. As such, starting July 2023 the Company discontinued recognizing the related expense on a prospective basis and will not reverse the previously recognized expenses. In May 2023, four employees were terminated who had outstanding PSU awards. As the performance-based metrics were not achieved at time of termination, the stock compensation expense related to these awards of $0.4 million was reversed. As of December 31, 2023, other than 500 PSUs for which vesting was accelerated in connection with the modification noted below, 1,349 PSUs have vested as the performance-based metrics of the PSUs were met upon the achievement of $35 million in cumulative net sales of Zokinvy.
During the years ended December 31, 2023 and 2022, the Company granted 1,305 and 9,925 RSUs, respectively, with a weighted-average grant date fair value of $67.80 and $156.58 per share, respectively. In relation to the RSUs granted, the Company recognized $0.7 million and $1.2 million in stock-based compensation expense for the years ended December 31, 2023 and 2022, respectively, which were included in selling, general and administrative expenses.
The following table summarizes RSU and PSU activity and weighted average grant date fair value for the year ended December 31, 2023: | | | | | | | | | | | |
| Shares | | Weighted-
Average
Grant Date
Fair Value |
| Unvested shares as of December 31, 2022 | 21,337 | | | $ | 220.40 | |
| Granted | 1,805 | | | $ | 67.80 | |
| Vested | (8,625) | | | $ | 176.10 | |
| Forfeited | (8,519) | | | $ | 236.55 | |
| Unvested shares as of December 31, 2023 | 5,998 | | | $ | 215.24 | |
Awards Modification
On February 6, 2023, the Company entered into a separation agreement and general release with David Cory, the Company's former President and CEO. Pursuant to the separation agreement, 50% of Mr. Cory's unvested equity awards were accelerated to vest on the date the separation agreement was executed. Additionally, the exercise period for Mr. Cory's vested awards was extended, including his accelerated awards. The stock compensation recognized related to these modifications was $0.9 million for the year ended December 31, 2023, which is reflected in selling, general and administrative expenses.
Stock-Based Compensation Expense
Total stock-based compensation expense recognized was as follows (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Research and development | $ | 2,391 | | | $ | 3,159 | |
| Selling, general and administrative | 2,617 | | | 5,158 | |
| Total | $ | 5,008 | | | $ | 8,317 | |
As of December 31, 2023, the total unrecognized compensation expense related to unvested awards was $10.9 million, which the Company expects to recognize over an estimated weighted average period of 2.6 years.
10. Income Taxes
The Company’s provision for income taxes was approximately $6,000 and $23,000 for the years ended December 31, 2023 and 2022, respectively, with an effective tax rate of 0.01% and (0.02)% for the years ended December 31, 2023 and 2022, respectively. The effective tax rate in each period differs from the U.S. statutory tax rate primarily due to the valuation allowance on the Company’s deferred tax assets as it is more likely than not that some or all of the Company’s deferred tax assets will not be realized. The tax expense recorded for the year ended December 31, 2023 relates to foreign taxes.
The following table presents loss before provision for income taxes as follows (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Domestic | $ | (75,002) | | | $ | (96,769) | |
| Foreign | 45 | | | 16 | |
| Total loss before provision for income taxes | $ | (74,957) | | | $ | (96,753) | |
The state and foreign income tax provisions for the years ended December 31, 2023 and 2022 are summarized as follows (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Current: | | | |
| State | $ | — | | | $ | 21 | |
| Foreign | 6 | | | 2 | |
Total current | $ | 6 | | | $ | 23 | |
| | | |
| Deferred: | | | |
| State | $ | — | | | $ | — | |
| Foreign | — | | | — | |
Total deferred | $ | — | | | $ | — | |
| Total provision for income taxes | $ | 6 | | | $ | 23 | |
The effective tax rate of the provision for income taxes differs from the federal statutory rate as follows:
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Federal statutory income tax rate | 21.00 | % | | 21.00 | % |
| Change in valuation allowance | (27.82) | | | (25.99) | |
| Tax credits | 6.24 | | | 4.15 | |
| State income taxes, net of federal benefit | 4.12 | | | 2.39 | |
| Stock-based compensation | (3.47) | | | (0.98) | |
| Other, net | (0.06) | | | (0.59) | |
| Provision for income taxes | 0.01 | % | | (0.02) | % |
The components of the deferred tax assets and liabilities are as follows (in thousands):
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Deferred tax assets: | | | |
| Net operating loss carryforwards | $ | 69,417 | | | $ | 63,069 | |
| Tax credits | 15,194 | | | 9,985 | |
| Depreciation and amortization | 2,775 | | | 2,920 | |
| Stock-based compensation | 1,905 | | | 3,429 | |
Section 174 capitalization | 26,668 | | | 15,581 | |
| Accruals and reserves | 884 | | | 1,431 | |
| Lease liabilities | 20 | | | 131 | |
| Gross deferred tax assets | 116,863 | | | 96,546 | |
| Valuation allowance | (116,842) | | | (96,418) | |
| Total deferred tax assets | 21 | | | 128 | |
| Deferred tax liabilities: | | | |
| Right-of-use assets | (21) | | | (128) | |
| Total deferred tax liabilities | (21) | | | (128) | |
| Net deferred tax assets | $ | — | | | $ | — | |
Due to the Company’s lack of earnings history, the deferred tax assets have been fully offset by a valuation allowance as of December 31, 2023 and 2022. The net change in the valuation allowance for the years ended December 31, 2023 and 2022 was an increase of $20.4 million and $25.1 million, respectively.
As of December 31, 2023, the Company had approximately $322.5 million and $47.1 million, respectively of federal and state operating loss carryforwards available to reduce future taxable income that will begin to expire in 2030 and 2028, respectively, for federal and state tax purposes. The Company had $15.5 million of federal Orphan Drug credit carryforwards available to reduce future taxable income that will begin to expire in 2041.
As of December 31, 2023, the Company also had research and development tax credit carryforwards of approximately $1.6 million and $4.0 million for federal and state purposes available to offset future taxable income tax, respectively. If not utilized, the federal carryforwards will expire beginning in 2043, and the state credits can be carried forward indefinitely.
The Company’s ability to utilize NOL carryforwards or other tax attributes, such as research tax credits, in any taxable year may be limited if the Company has experienced an ownership change under Section 382 of the Internal Revenue Code (the Code) of 1986, as amended. The Company’s merger with Celladon resulted in such an ownership change and, accordingly, Celladon’s NOL carryforwards and certain other tax attributes will be subject to further limitations on their use. The Company assessed whether it had an ownership change, as defined by Section 382 of the Code from its formation through December 31, 2023. Based upon this assessment, the Company experienced ownership changes on April 20, 2016, October 18, 2018 and December 31, 2020. Due to the April 20, 2016 and October 18, 2018 ownership changes, reductions were made to the Company’s NOL and tax credit carryforwards under these rules. Additional ownership changes could result in additional limitations on the Company’s NOL and tax credit carryforwards.
Tax benefits from uncertain tax positions are recognized when it is more likely than not that the position will be sustained upon examination, including resolutions of any related appeals or litigation processes, based on the technical merits. The amount recognized is measured as the largest amount of tax benefit that is greater than 50 percent likely of being realized upon effective settlement.
Uncertain Tax Positions
A reconciliation of the Company’s unrecognized tax benefits for the years ended December 31, 2023 and 2022 is as follows (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Balance at beginning of year | $ | 3,492 | | | $ | 1,950 | |
Additions based on tax positions related to current year | 1,784 | | | 1,586 | |
Reductions based on tax positions related to prior year | — | | | (44) | |
| Balance at end of year | $ | 5,276 | | | $ | 3,492 | |
If the $5.3 million of unrecognized tax benefits is recognized, there would not be an effect on the effective tax rate. The Company does not expect the unrecognized tax benefits to change significantly over the next 12 months. As of December 31, 2023, the unrecognized tax benefits for uncertain tax positions were offset against deferred tax assets and would not affect the income tax rate if recognized due to the Company being in a full valuation allowance position.
The Company’s policy is to account for interest and penalties in tax expense on the consolidated statements of operations. The Company files income tax returns in the U.S. federal and state jurisdictions. All periods since inception are subject to examination by U.S. federal and state jurisdictions. There were no such interest or penalties during the years ended December 31, 2023 and 2022.
11. Legal Matters
From time to time in the ordinary course of business, the Company may be involved in various legal claims and litigation, including those pertaining to the defense and enforcement of the Company's patent or other intellectual property rights and contractual rights. The Company is defending all current litigation matters, including the litigation matters discussed below. Although there can be no assurances and the outcome of these matters is currently not determinable, the Company currently believes that none of these claims or proceedings are likely to have a material adverse effect on the Company's financial position.
The Company records accruals for outstanding legal proceedings, claims or investigations when it is probable that a liability will be incurred, and the amount of loss can be reasonably estimated. The Company evaluated developments in legal proceedings, claims or investigations that could affect the amount of any accrual, as well as any developments that would result in a loss contingency to become both probable and reasonably estimable. The Company has not recorded any accrual for loss contingencies associated with such legal claims or litigation discussed herein as they are in the early stages and an estimate of loss, if any, cannot be made. Legal fees and other costs associated with such actions will be expensed as incurred.
Schoen v. Eiger BioPharmaceuticals, Inc., et al., Case No. 22-cv-06985
On November 8, 2022 a putative securities class action complaint was filed in the United States District Court for the Northern District of California alleging that the company and two former executives violated Sections 10(b) and 20(a) of the Securities Exchange Act and SEC Rule 10b-5. The complaint alleged generally that between March 2021 and October 2022, material misstatements and omissions were made to shareholders regarding the TOGETHER study of peginterferon lambda for the treatment of COVID-19 as well as the likelihood of FDA approval of an Emergency Use Authorization for peginterferon lambda. The Court appointed a lead plaintiff on March 2, 2023. On April 10, 2023, the lead plaintiff filed a notice of voluntary dismissal without prejudice.
The Progeria Research Foundation, Inc. v. Eiger BioPharmaceuticals, Inc. Arbitration
On November 15, 2022, the Company received a demand for arbitration (Demand) from claimant The Progeria Research Foundation, Inc. (PRF) asserting two claims under a May 15, 2018 Collaboration and Supply Agreement (the PRF Collaboration Agreement) between the parties. The terms of the PRF Collaboration Agreement are discussed in Note 5 of our consolidated financial statements. PRF has alleged that the Company breached an obligation to supply quantities of a drug as requested by PRF. PRF also has a claim for declaratory relief regarding the grant of licenses under the PRF Collaboration Agreement. On January 18, 2023, the Company filed a response to the Demand denying PRF’s claims, contesting the arbitrability of PRF’s claim for declaratory relief, and asserting a counterclaim for declaratory relief related
to the contractual provision underlying PRF’s original drug supply claim. To give the parties an opportunity to discuss a potential negotiated resolution of their dispute, the arbitration was suspended through the end of 2023. As a result, all arbitration activities were on hold, and the final hearing, originally scheduled for May 9 – 12, 2023, in Boston, Massachusetts, was cancelled. On February 29, 2024 the Company and PRF negotiated and entered into an amended and restated agreement. The amended and restated collaboration agreement with PRF confirms the Company's exclusive right to commercialize Zokinvy subject to terms and conditions of the Agreement. On March 1, 2024, the arbitration was dismissed with prejudice by both parties.
12. Commitments and Contingencies
Indemnification Agreements for Directors and Officers
In the ordinary course of business, the Company enters into agreements that may include indemnification provisions. Pursuant to such agreements, the Company may indemnify, hold harmless and defend an indemnified party for losses suffered or incurred by the indemnified party. Some of the provisions will limit losses to those arising from third-party actions. In some cases, the indemnification will continue after the termination of the agreement. The maximum potential amount of future payments the Company could be required to make under these provisions is not determinable. The Company has never incurred material costs to defend lawsuits or settle claims related to these indemnification provisions and has never accrued any liabilities related to such obligations in its consolidated financial statements. The Company has also entered into indemnification agreements with its directors and officers that may require the Company to indemnify its directors and officers against liabilities that may arise by reason of their status or service as directors or officers to the fullest extent permitted by Delaware corporate law. The Company currently has directors’ and officers’ insurance.
Other
The Company enters into contracts in the normal course of business with various third parties for clinical trials, preclinical research studies and testing, manufacturing and other services and products for operating purposes. These contracts generally provide for termination upon notice. Payments due upon cancellation consist only of payments for services provided or expenses incurred, including non-cancellable obligations of our service providers, up to the date of cancellation.
13. Subsequent Events
Voluntary Filing Under Chapter 11
As previously reported in the Company's Current Report on Form 8-K, filed with the U.S. Securities and Exchange Commission on April 1, 2024, the Company and its wholly owned subsidiaries commenced voluntary proceedings under Chapter 11 of the United States Bankruptcy Code in the United States Bankruptcy Court for the Northern District of Texas (Bankruptcy Court). The Chapter 11 proceedings are jointly administered under the caption In re Eiger BioPharmaceuticals, Inc., et al, Case No. 24-80040 . (Chapter 11 Cases). The Company continues to operate its business in accordance with the applicable provisions of the Bankruptcy Code and orders of the Bankruptcy Court.
On March 31, 2024, we entered into a “stalking horse” asset purchase agreement (Asset Purchase Agreement) with Sentynl Therapeutics, Inc. (Purchaser), pursuant to which the Purchaser has agreed to acquire substantially all of the rights of the Company to its Zokinvy® program, including the Company’s in-license from Merck Sharp & Dohme Corp. (successor-in-interest of Schering Corporation) (Transferred Assets). The acquisition of the Transferred Assets by the Purchaser pursuant to the Asset Purchase Agreement is subject to approval of the Bankruptcy Court and one or more auctions, if necessary, to solicit higher or otherwise better bids. On April 1, 2024, we filed a motion (Bidding Procedures Motion) seeking approval of, among other things, certain bidding procedures (Bidding Procedures), which will establish procedures for the selection of the highest or otherwise best bids for the sale of the Transfer Assets and other assets. Other interested bidders would be permitted to participate in the auction if they submit qualifying bids that are higher or otherwise better than the Asset Purchase Agreement. The Asset Purchase Agreement acts as a baseline for competitive bids for the acquisition of the Transferred Assets. The Bidding Procedures Motion additionally seeks the Bankruptcy Court’s approval of the Asset Purchase Agreement and designation of the Purchaser as the “stalking horse” bidder for the Transferred Assets.
Under the Asset Purchase Agreement, the Purchaser has agreed, subject to the Bankruptcy Court’s approval and absent any higher or otherwise better bid, to acquire the Transferred Assets from the Debtors for $26 million, subject to certain adjustments, including per diem reductions if the sale closes after April 24, 2024, in accordance with the terms and conditions of the Asset Purchase Agreement, plus the assumption of specified liabilities related to the Transferred Assets.
The Asset Purchase Agreement includes customary representations and warranties and various customary covenants under the circumstances that are subject to certain limitations, including, without limitation, a termination fee, expense reimbursement and the right to designate executory contracts and unexpired leases to assume or reject. On April 3, 2024, the Purchaser increased the purchase price to $30.0 million, subject to certain adjustments, including per diem reductions if the sale closes after April 24, 2024.
Subject to certain specific exceptions under the Bankruptcy Code, the Chapter 11 Cases automatically stayed most judicial or administrative actions against the Company and its direct subsidiaries and efforts by creditors to collect on or otherwise exercise rights or remedies with respect to obligations of the Company and its direct subsidiaries incurred prior to the petition date.
Nasdaq Listing
On April 2, 2024, the Company received written notice (Delisting Notice) from the Listing Qualifications Department of the Nasdaq Stock Market LLC (Nasdaq) notifying the Company that, as a result of the Chapter 11 Cases and in accordance with Nasdaq Listing Rules 5101, 5110(b) and IM-5101-1, Nasdaq had determined that the Company’s common stock will be delisted from Nasdaq. The Delisting Notice also advises the Company of its right to appeal Nasdaq’s determination pursuant to procedures set forth in Nasdaq Listing Rule 5800 Series. The Company does not intend to pursue an appeal.
Trading of the Company’s common stock will be suspended at the opening of business on April 11, 2024. Nasdaq will file a Form 25-NSE with the Securities and Exchange Commission, which will remove the Company’s common stock from listing and registration on Nasdaq. As a result, the Company’s common stock is expected to begin trading on the over-the-counter (“OTC”) market on April 11, 2024. On the OTC market, shares of the Company’s common stock, which previously traded on the Nasdaq under the symbol EIGR, are expected to trade under the symbol EIGRQ.
ITEM 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
ITEM 9A. Controls and Procedures
(a)Evaluation of Disclosure Controls and Procedures.
We are responsible for maintaining disclosure controls and procedures, as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act. Disclosure controls and procedures are controls and other procedures designed to ensure that the information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is accumulated and communicated to our management, including our principal executive officer and our principal financial officer, as appropriate to allow timely decisions regarding required disclosure.
Based on our management’s evaluation (with the participation of our principal executive officer and our principal financial officer) of our disclosure controls and procedures as required by Rule 13a-15 under the Exchange Act, our principal executive officer and our principal financial officer have concluded that our disclosure controls and procedures were effective to achieve their stated purpose as of December 31, 2023, the end of the period covered by this report.
(b)Management’s Report on Internal Control over Financial Reporting.
Our management is responsible for establishing and maintaining adequate internal control over financial reporting as such term is defined in Exchange Act Rule 13a-15(f). Internal control over financial reporting is a process designed under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, to provide reasonable assurance, not absolute assurance, regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America.
There are inherent limitations to the effectiveness of any system of internal control over financial reporting. These limitations include the possibility of human error, the circumvention or overriding of the system and reasonable resource constraints. Because of its inherent limitations, our internal control over financial reporting may not prevent or detect
misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. Under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission.
As of December 31, 2023, our management assessed the effectiveness of our internal control over financial reporting using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control-Integrated Framework (2013 Framework). Based on this assessment, our management concluded that, as of December 31, 2023, our internal control over financial reporting was effective based on those criteria.
(c)Changes in Internal Control over Financial Reporting.
There were no changes in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) that occurred during the fiscal year ended December 31, 2023, that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. Other Information
During the three months ended December 31, 2023, none of our directors or executive officers has adopted or terminated a Rule 10b5-1 trading arrangement or a non-Rule 10b5–1 trading arrangement (each as defined in Item 408 of Regulation S-K under the Securities Exchange Act of 1934, as amended).
ITEM 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
PART III. OTHER INFORMATION
ITEM 10. Directors, Executive Officers and Corporate Governance
Except as set forth below, the information required by this item is incorporated by reference from our definitive Proxy Statement to be filed with the SEC in connection with our 2024 Annual Meeting of Stockholders or an amendment to this Form 10-K within 120 days after the end of the fiscal year ended December 31, 2023.
We have adopted a Code of Business Conduct and Ethics that applies to all of our directors, officers and employees, including our principal executive officer and principal financial officer. A current copy of the code is posted on the Investors Corporate Governance section of our website, which is located at www.eigerbio.com.
We intend to satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding an amendment to, or waiver from, a provision of this Code of Business Conduct and Ethics by posting such information on our website, at the address and location specified above and, to the extent required by the listing standards of The Nasdaq Global Market, by filing a Current Report on Form 8-K with the SEC, disclosing such information.
ITEM 11. Executive Compensation
The information required by this item is incorporated by reference from our definitive Proxy Statement to be filed with the SEC in connection with our 2024 Annual Meeting of Stockholders or an amendment to this Form 10-K within 120 days after the end of the fiscal year ended December 31, 2023.
ITEM 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this item is incorporated by reference from our definitive Proxy Statement to be filed with the SEC in connection with our 2024 Annual Meeting of Stockholders or an amendment to this Form 10-K within 120 days after the end of the fiscal year ended December 31, 2023.
ITEM 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this item is incorporated by reference from our definitive Proxy Statement to be filed with the SEC in connection with our 2024 Annual Meeting of Stockholders or an amendment to this Form 10-K within 120 days after the end of the fiscal year ended December 31, 2023.
ITEM 14. Principal Accountant Fees and Services
Our independent registered public accounting firm is KPMG LLP, San Francisco, CA, Auditor ID: 185
The information required by this item is incorporated by reference from our definitive Proxy Statement to be filed with the SEC in connection with our 2024 Annual Meeting of Stockholders or an amendment to this Form 10-K within 120 days after the end of the fiscal year ended December 31, 2023.
PART IV
ITEM 15. Exhibits and Financial Statement Schedules
(a)Financial Statements and Financial Statement Schedules
1.Financial Statements
See Index to Financial Statements at Item 8 herein.
2.Financial Statement Schedules
All other schedules are omitted because they are not applicable, or the required information is shown in the financial statements or notes thereto.
3.Exhibits
| | | | | | | | |
Exhibit
Number | | Description of Document |
| | |
| 2.1 | | |
| | |
| 3.1 | | |
| | |
| 3.2 | | |
| | |
| 3.3 | | |
| | |
| 3.4 | | |
| | |
| 3.5 | | |
| | |
| 4.1 | | |
| | |
| 4.2 | | |
| | |
| 4.3 | | |
| | |
| 10.1+ | | |
| | |
| | | | | | | | |
| 10.2+ | | |
| | |
| 10.3+ | | |
| | |
| 10.4+ | | |
| | |
| 10.5+ | | |
| | |
| 10.6+ | | |
| | |
| 10.7+ | | |
| | |
| 10.8 | | |
| | |
| 10.9* | | |
| | |
| 10.10* | | |
| | |
| 10.11 | | |
| | |
| 10.12* | | |
| | |
| 10.13 | | |
| | |
| | | | | | | | |
| 10.14 | | |
| | |
| 10.15 | | |
| | |
10.16* | | |
| | |
10.17* | | |
| | |
| 10.18 | | |
| | |
10.19+ | | |
| | |
10.20+ | | |
| | |
10.21+ | | |
| | |
10.22+ | | |
| | |
10.23* | | |
| | |
10.24+ | | |
| | |
10.25+ | | |
| | |
10.26* | | |
| | |
| | | | | | | | |
| 10.27 | | |
| | |
| 10.28 | | |
| | |
10.29+ | | |
| | |
10.30+ | | |
| | |
10.31+ | | |
| | |
10.32* | | |
| | |
| 10.33 | | Common Stock Purchase Agreement, dated June 1, 2022, among Eiger BioPharmaceuticals, Inc., Innovatus Life Sciences Lending Fund I, LP, Innovatus Life Sciences Offshore Fund I-A, LP, Innovatus Flagship Fund I, LP, and Innovatus Flagship Offshore Fund I, LP (incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K filed with the SEC on June 7, 2022). |
| | |
10.34+ | | |
| | |
10.35+ | | |
| | |
| 10.4 | | |
| | |
10.37+ | | |
| | |
10.38+
| | |
| | |
10.39+ | | |
| | |
10.40+ | | |
| | |
| | | | | | | | |
10.41+ | | |
| | |
| 21.1** | | |
| | |
| 24.1 | | |
| | |
| 31.1** | | |
| | |
| 31.2** | | |
| | |
| 32.1** | | |
| | |
97.1** | | |
| | |
| 101.INS** | | Inline XBRL Instance Document- the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document. |
| | |
| 101.SCH** | | Inline XBRL Taxonomy Extension Schema Document |
| | |
| 101.CAL** | | Inline XBRL Taxonomy Extension Calculation Linkbase Document |
| | |
| 101.DEF** | | Inline XBRL Taxonomy Extension Definition Linkbase Document |
| | |
| 101.LAB** | | Inline XBRL Taxonomy Extension Label Linkbase Document |
| | |
| 101.PRE** | | Inline XBRL Taxonomy Extension Presentation Linkbase Document |
| | |
| 104 | | The cover page from the Company’s Annual Report on Form 10-K for the year ended December 31, 2023, has been formatted in Inline XBRL and is contained in Exhibit 101. |
__________________________
+ Indicates management contract or compensatory plan.
* Portions of this exhibit have been omitted as being immaterial and would be competitively harmful if publicly disclosed.
** Filed herewith.
ITEM 16. Form 10-K Summary
None.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | |
| Eiger BioPharmaceuticals, Inc. |
| | |
Date: April 8, 2024 | By: | /s/ David Apelian |
| | David Apelian |
| | Director, Chief Executive Officer |
| | (Principal Executive Officer) |
| | |
| Eiger BioPharmaceuticals, Inc. |
| | |
Date: April 8, 2024 | By: | /s/ William G. Kachioff |
| | William G. Kachioff |
| | Chief Financial Officer |
| | (Principal Financial and Accounting Officer) |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints David Apelian and William G. Kachioff, and each of them, as his or her attorneys-in-fact, with the power of substitution, for him or her in any and all capacities, to sign any amendments to this report, and to file the same, with exhibits thereto and other documents in connection therewith with the Securities and Exchange Commission, hereby ratifying and confirming all that said attorneys-in-fact, and each of them, or his or her substitute or substitutes may do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant in the capacities and on the dates indicated:
| | | | | | | | | | | | | | |
| Signature | | Title | | Date |
| | | | |
| /s/ David Apelian | | Member of the Board of Directors, Chief Executive Officer (Principal Executive Officer) | | April 8, 2024 |
| David Apelian | | | |
| | | | |
/s/ William G. Kachioff | | Chief Financial Officer (Principal Financial and Accounting Officer) | | April 8, 2024 |
William G. Kachioff | | | |
| | | | |
| /s/ Thomas J. Dietz | | Chairman of the Board of Directors | | April 8, 2024 |
| Thomas J. Dietz | | | | |
| | | | |
| /s/ Evan Loh | | Member of the Board of Directors | | April 8, 2024 |
| Evan Loh | | | | |
| | | | |
| /s/ Amit K. Sachdev | | Member of the Board of Directors | | April 8, 2024 |
| Amit K. Sachdev | | | | |
| | | | |
| /s/ Kim Sablich | | Member of the Board of Directors | | April 8, 2024 |
| | | | | | | | | | | | | | |
| Kim Sablich | | | | |
| | | | |
| /s/ Lisa Kelly-Croswell | | Member of the Board of Directors | | April 8, 2024 |
| Lisa Kelly-Croswell | | | | |