Tharimmune Presents Favorable TH104 Phase 1 Clinical Data at the American College of Gastroenterology 2024 Annual Scientific Meeting
TH104, the Company's lead candidate for moderate-to-severe pruritus in chronic liver disease, showed no opioid withdrawal effects, reinforcing safety profile
TH104 was well tolerated with no unexpected treatment-emergent adverse events, supporting further clinical development
Company expects Phase 2 topline data for chronic pruritus in primary biliary cholangitis (PBC) patients in 2025
BRIDGEWATER, NJ / ACCESSWIRE / October 28, 2024 / Tharimmune, Inc. (NASDAQ:THAR) ("Tharimmune" or the "Company"), a clinical-stage biotechnology company developing a portfolio of therapeutic candidates in inflammation and immunology, presented new TH104 Phase 1 data at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting, underway in Philadelphia. The Phase 1 trial was a single-dose, single-center, open-label, randomized study of TH104 transmucosal buccal film conducted in two cohorts of patients with chronic liver disease (CLD). The primary outcome measure was to determine the safety and tolerability of a single buccal dose of TH104 in these patients.
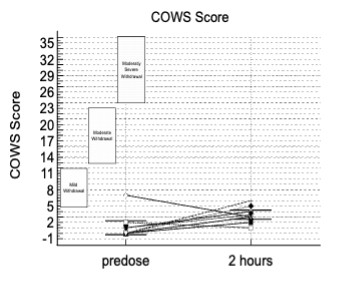
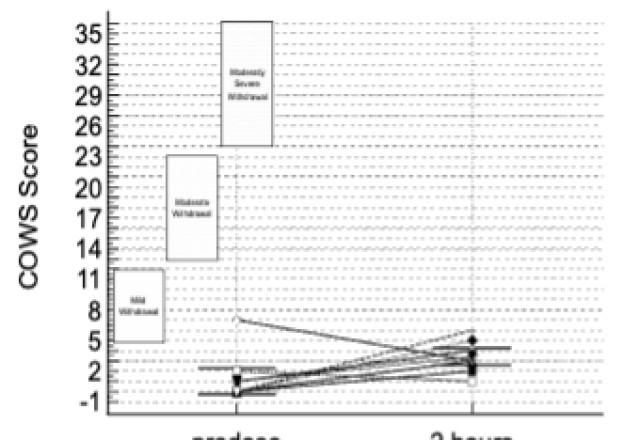
The data presented included adverse events (AEs) as well as assessment of patients using the Clinical Opiate Withdrawal Scale (COWS). The COWS is an 11-item clinician-administered scale that can be used in both inpatient and outpatient settings to reproducibly rate common signs and symptoms of opiate withdrawal and monitor them over time. The summed score for the complete scale can be used by clinicians to determine the stage or severity of withdrawal, and to assess the level of any physical dependence.
This study enrolled two types of CLD patients categorized as Child-Pugh A (cohort A) and Child-Pugh B (cohort B). The Child-Pugh score is a system for assessing the prognosis and necessity of transplant in CLD that provides a forecast of the increasing severity of a patient's liver disease and expected survival rate. The score is determined by scoring clinical measures of liver disease and the possibility of eventual liver failure, with Class A indicating mild liver disease and Class B indicating moderate liver disease with a one-to-five-year survival rates of 95% and 75%, respectively. There were no patients enrolled in this study with the most severe Child-Pugh C classification.
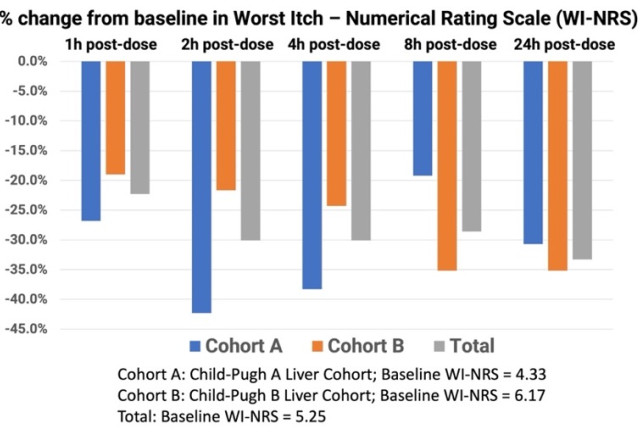
The mean baseline WI-NRS scores in Groups A and B were 4.33 and 6.17, respectively, translating to moderate-to-severe chronic pruritus at the start of the study. The mean baseline itch score for all 12 subjects was 5.25. At one hour post-dosing with TH104, Group A and Group B had a mean decline in WI-NRS scores of 26.8% and 19.0%, respectively, and continued to decline two hours post-dose by 42.3% and 21.7%, respectively. Both cohorts continued to improve in mean itch scores at the four-hour and eight-hour time points, including the combined total subjects. At 24-hours post dosing, Group A and Group B achieved a mean decline of 30.7% and 35.2%, respectively, in pruritus scores. The mean reduction in itch scores for all 12 subjects 24 hours after a single dose of TH104 was 33.3%.
There were no deaths, other serious adverse events or other significant adverse events reported during the entire study. There were no new adverse events during the entire study, with events correlated with previous studies and a safety profile consistent with the literature for the active ingredient in TH104.
"We are pleased with the totality of the Phase 1 data with TH104, which build upon previous studies demonstrating reliable and predictable delivery of nalmefene using our proprietary microparticle embedded transmucosal delivery system. This system is easily applied to the inside of the cheek within seconds in healthy volunteers. The mean 33.3% reduction in itch scores just 24 hours after a single dose of TH104 demonstrates the potential for rapid symptomatic relief," said Randy Milby, CEO of Tharimmune. "The data presented today in liver disease further strengthen our confidence in the potential of TH104 to address pruritis, a debilitating symptom for these patients."
The Company plans to initiate a Phase 2 multiple-ascending dose trial in the coming months to assess the safety and tolerability of TH104, which will assess the change from baseline in WI-NRS scores to evaluate chronic pruritus in PBC patients. The Company expects topline data in 2025 and is engaging with both U.S. and EU regulatory authorities.
About TH104
TH104 is embedded with nalmefene onto a proprietary transdermal buccal film that easily adheres to the inside of the mouth. This endows TH104 with key features making it an ideal product candidate for multiple liver-related and other pruritogenic inflammatory conditions. The molecule has a dual mechanism of action affecting both the µ-opioid receptor and the kappa-opioid receptor, as well as potentially inhibiting IL-17 inflammatory cytokine expression. These opioid receptors when stimulated and/or inhibited by the body's natural ligands have been known to be involved in the body's itch circuitry.
About Pruritus and Primary Biliary Cholangitis
According to the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), part of the National Institutes of Health, PBC is a chronic disease where the bile ducts in the liver eventually become dysfunctional and cause the buildup of bile, resulting in liver damage. The disease, believed to be an autoimmune condition, affects an estimated 58 out of every 100,000 U.S. women and about 15 out of every 100,000 U.S. men. Pruritus is one of the most common conditions associated with PBC, affecting up to 75% of individuals at some point during their disease course. It has a negative impact on health-related quality of life with limited treatment options. Published survey data of PBC respondents suffering from pruritus described their itch as "bugs crawling under the skin." More than 65% of patients reported that the itch was worse at night, known as nocturnal pruritus, a high unmet need.
About Tharimmune
Tharimmune, Inc. is a clinical-stage biotechnology company developing a diverse portfolio of therapeutic candidates in immunology and inflammation. The lead clinical asset, TH104, aims to suppress chronic pruritus associated with primary biliary cholangitis (PBC), a rare autoimmune liver disease with no known cure. The expanded pipeline includes TH023, an oral TNF-alpha inhibitor, offering a new approach to treating autoimmune diseases. Tharimmune is also advancing early-stage multi-specific biologics targeting unique epitopes against multiple solid tumors. The company has a license agreement with OmniAb, Inc. to access their antibody discovery technology for targeting specified disease markers. For more information, please visit: www.tharimmune.com.
Forward Looking Statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, contained in this press release, including statements regarding Tharimmune's or Intract's future financial or operating performance, the timing and design of Tharimmune's future Phase 2 trial, Tharimmune's and Intract's expectations with respect to the Merger, including the timing of entering into a definitive agreement, the timing of closing thereof, the pro forma ownership of the combined company, anticipated financing plans, the combined company's strategy, future operations, future financial position, projected costs, prospects, plans and objectives of management, are forward-looking statements. The words "anticipate," "believe," "continue," "could," "depends," "estimate," "expect," "intend," "may," "ongoing," "plan," "potential," "predict," "project," "target," "should," "will," "would," and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Such forward-looking statements are based on the beliefs of management, as well as assumptions made by, and information currently available to, Tharimmune and Intract's management. Tharimmune may not actually achieve the plans, intentions, or expectations disclosed in these forward-looking statements, and you should not place undue reliance on these forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in these forward-looking statements. Factors that may cause such differences include, but are not limited to, those discussed under Risk Factors set forth in our Annual Report on Form 10-K for the year ended December 31, 2023 and other periodic reports filed by Tharimmune from time to time with the Securities and Exchange Commission. In addition, the forward-looking statements included in this press release represent Tharimmune's and Intract's views as of the date of this release. Subsequent events and developments may cause Tharimmune's views to change; however, Tharimmune does not undertake and specifically disclaims any obligation to update or revise any forward-looking statements to reflect new information, future events or circumstances or to reflect the occurrences of unanticipated events, except as may be required by applicable law. These forward-looking statements should not be relied upon as representing Tharimmune's views as of any date subsequent to the date of this release.
Contacts:
Tharimmune, Inc.
[email protected]
Alliance Advisors IR
Tirth T. Patel
[email protected]
212-201-6614
Contact Information
Tirth Patel
LHA Investor Relations
[email protected]
1-212-201-6614
Related Images
|
SOURCE: Tharimmune, Inc.
View the original press release on accesswire.com